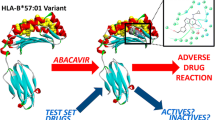
Abstract
Genetic polymorphism in the genes encoding the human leukocyte antigen (HLA) molecules enables presentation of a wide range peptide ligands thus maximising immune surveillance of pathogens. A consequence of the diversification of the HLA Ag-binding pocket is the enhanced opportunity for off-target binding of small drugs by HLA molecules, with subsequent immune reactivity. These potential off-target interactions are ‘set up’ to generate T cell-mediated adverse drug reactions even though the precise mechanisms of most HLA-drug interactions are still poorly understood. The association between abacavir hypersensitivity syndrome and HLA-B*57:01 is one exception that has been resolved at a molecular and mechanistic level. Here, we explore the road to understanding the interaction between abacavir and the HLA-B*57:01 molecule and review the current state of understanding of interactions between other drugs and HLA molecules implicated in adverse drug reactions, which appear to involve multiple mechanisms. The continued expansion of the pharmacopoeia generates an imperative to understand these interactions at the molecular level in order to prevent the continued burden on individuals and the health care system.



Similar content being viewed by others
References
Adam J, Eriksson KK, Schnyder B, Fontana S, Pichler WJ, Yerly D (2012) Avidity determines T-cell reactivity in abacavir hypersensitivity. Eur J Immunol 42:1706–1716. doi:10.1002/eji.201142159
Adam J et al (2014) Abacavir induced T cell reactivity from drug naïve individuals shares features of allo-immune responses. PLoS One 9:e95339. doi:10.1371/journal.pone.0095339
Akiba H et al (2002) Skin inflammation during contact hypersensitivity is mediated by early recruitment of CD8+T cytotoxic 1 cells inducing keratinocyte apoptosis. J Immunol 168:3079–3087. doi:10.4049/jimmunol.168.6.3079
Arroyo S, de la Morena A (2001) Life-threatening adverse events of antiepileptic drugs. Epilepsy res 47:155–174. doi:10.1016/S0920-1211(01)00306-0
Bates DW et al (1997) The costs of adverse drug events in hospitalized patients. Adverse drug events prevention study group. Jama 277:307–311. doi:10.1001/jama.1997.03540280045032
Bharadwaj M, Illing P, Theodossis A, Purcell AW, Rossjohn J, McCluskey J (2012) Drug hypersensitivity and human leukocyte antigens of the major histocompatibility complex. Annu rev Pharmacol Toxicol 52:401–431. doi:10.1146/annurev-pharmtox-010611-134701
Brander C, Maurihellweg D, Bettens F, Rolli H, Goldman M, Pichler WJ (1995) Heterogeneous T cell responses to beta-lactam-modified self-structures are observed in penicillin-allergic individuals. J Immunol 155:2670–2678
Burrows SR, Khanna R, Burrows JM, Moss DJ (1994) An alloresponse in humans is dominated by cytotoxic T lymphocytes (CTL) cross-reactive with a single Epstein-Barr virus CTL epitope: implications for graft-versus-host disease. J Exp med 179:1155–1161. doi:10.1084/jem.179.4.1155
Cao ZH et al (2012) HLA-B*58:01 allele is associated with augmented risk for both mild and severe cutaneous adverse reactions induced by allopurinol in Han Chinese. Pharmacogenomics 13:1193–1201. doi:10.2217/pgs.12.89
Carolino F, Santos N, Piñeiro C, Santos AS, Soares P, Sarmento A, Rodrigues Cernadas J (2017) Prevalence of abacavir-associated hypersensitivity syndrome and HLA-B*5701 allele in a Portuguese HIV-positive population. Porto Biomedical Journal 2:59–62. doi:10.1016/j.pbj.2016.12.004
Chan AL, Lee HY, Ho CH, Cham TM, Lin SJ (2008) Cost evaluation of adverse drug reactions in hospitalized patients in Taiwan: a prospective, descriptive, observational study. Curr Ther res Clin Exp 69:118–129. doi:10.1016/j.curtheres.2008.04.005
Chang CC, Too CL, Murad S, Hussein SH (2011) Association of HLA-B*1502 allele with carbamazepine-induced toxic epidermal necrolysis and Stevens-Johnson syndrome in the multi-ethnic Malaysian population. Int J Dermatol 50:221–224. doi:10.1111/j.1365-4632.2010.04745.x
Chang CC et al (2017) Association of HLA-B*15:13 and HLA-B*15:02 with phenytoin-induced severe cutaneous adverse reactions in a Malay population. Pharmacogenom J 17:170–173. doi:10.1038/tpj.2016.10
Chen Z, Liew D, Kwan P (2014) Effects of a HLA-B*15:02 screening policy on antiepileptic drug use and severe skin reactions. Neurology 83:2077–2084. doi:10.1212/WNL.0000000000001034
Chen Z, Liew D, Kwan P (2016) Real-world cost-effectiveness of pharmacogenetic screening for epilepsy treatment. Neurology 86:1086–1094. doi:10.1212/wnl.0000000000002484
Chen CB et al (2017) Risk and association of HLA with oxcarbazepine-induced cutaneous adverse reactions in Asians. Neurology 88:78–86. doi:10.1212/WNL.0000000000003453
Chessman D et al (2008) Human leukocyte antigen class I-restricted activation of CD8+ T cells provides the immunogenetic basis of a systemic drug hypersensitivity. Immunity 28:822–832. doi:10.1016/j.immuni.2008.04.020
Chiu ML et al (2012) Association between HLA-B*58:01 allele and severe cutaneous adverse reactions with allopurinol in Han Chinese in Hong Kong. Br J Dermatol 167:44–49. doi:10.1111/j.1365-2133.2012.10894.x
Chung WH et al (2004) Medical genetics: a marker for Stevens-Johnson syndrome. Nature 428:486. doi:10.1038/428486a
Chung WH et al (2008) Granulysin is a key mediator for disseminated keratinocyte death in Stevens-Johnson syndrome and toxic epidermal necrolysis. Nat Med 14:1343–1350. doi:10.1038/nm.1884
Chung WH et al (2015a) Insights into the poor prognosis of allopurinol-induced severe cutaneous adverse reactions: the impact of renal insufficiency, high plasma levels of oxypurinol and granulysin. Ann Rheum dis 74:2157–2164. doi:10.1136/annrheumdis-2014-205577
Chung WH et al (2015b) Oxypurinol-specific T cells possess preferential TCR clonotypes and express granulysin in allopurinol-induced severe cutaneous adverse reactions. J Invest Dermatol 135:2237–2248. doi:10.1038/jid.2015.165
Clayton GM et al (2014) Structural basis of chronic beryllium disease: linking allergic hypersensitivity and autoimmunity. Cell 158:132–142. doi:10.1016/j.cell.2014.04.048
Clemens EB et al (2016) Towards identification of immune and genetic correlates of severe influenza disease in indigenous Australians. Immunol Cell Biol 94:367–377. doi:10.1038/icb.2015.93
Dainichi T, Uchi H, Moroi Y, Furue M (2007) Stevens-Johnson syndrome, drug-induced hypersensitivity syndrome and toxic epidermal necrolysis caused by allopurinol in patients with a common HLA allele: what causes the diversity? Dermatology 215:86–88. doi:10.1159/000102045
Daly AK et al (2009) HLA-B*5701 genotype is a major determinant of drug-induced liver injury due to flucloxacillin. Nat Genet 41:816–819. doi:10.1038/ng.379
Easterbrook PJ et al (2003) Epidemiological risk factors for hypersensitivity reactions to abacavir. HIV Med 4:321–324. doi:10.1046/j.1468-1293.2003.00166.x
F.D.A (2008) Information for healthcare professionals: abacavir (marketed as Ziagen) and abacavir-containing medications. U.S. Food and Drug Administration, U.S. Department of Health and Human Services. http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm123927.htm. Accessed 29.12.2010 2010
Falta MT et al (2016) Metal-specific CD4+ T-cell responses induced by beryllium exposure in HLA-DP2 transgenic mice. Mucosal Immunol 9:218–228. doi:10.1038/mi.2015.54
Faulkner L, Meng X, Naisbitt DJ, Spraggs CF, Park BK (2016) No evidence for drug-specific activation of circulating T cells from patients with HLA-DRB1∗07:01-restricted lapatinib-induced liver injury. Chem res Toxicol 29:2111–2113. doi:10.1021/acs.chemrestox.6b00400
Genin E et al (2011) Genome-wide association study of Stevens-Johnson syndrome and toxic epidermal necrolysis in Europe. Orphanet J Rare dis 6:52. doi:10.1186/1750-1172-6-52
Genin E et al (2014) HLA-A*31:01 and different types of carbamazepine-induced severe cutaneous adverse reactions: an international study and meta-analysis. Pharmacogenomics J 14:281–288. doi:10.1038/tpj.2013.40
Günthard HF et al (2016) Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2016 recommendations of the International Antiviral Society-USA Panel. J Am Med Assoc 316:191–210. doi:10.1001/jama.2016.8900
Harding DJ, Subramaniam K, MacQuillan G, Davis J, Nolan D (2012) Severe drug-induced hypersensitivity syndrome with a shared HLA-B allele. Med J Aust 197:411–413. doi:10.5694/mja12.10477
Hari Y et al (2001) T cell involvement in cutaneous drug eruptions. Clin Exp Allergy 31:1398–1408. doi:10.1046/j.1365-2222.2001.01164.x
Hautekeete ML et al (1999) HLA association of amoxicillin-clavulanate-induced hepatitis. Gastroenterology 117:1181–1186. doi:10.1016/S0016-5085(99)70404-X
Hetherington S et al (2001) Hypersensitivity reactions during therapy with the nucleoside reverse transcriptase inhibitor abacavir. Clin Ther 23:1603–1614. doi:10.1016/S0149-2918(01)80132-6
Hetherington S et al (2002) Genetic variations in HLA-B region and hypersensitivity reactions to abacavir. Lancet 359:1121–1122. doi:10.1016/S0140-6736(02)08158-8
Hung SI et al (2005) HLA-B*5801 allele as a genetic marker for severe cutaneous adverse reactions caused by allopurinol. Proc Natl Acad Sci U S A 102:4134–4139. doi:10.1073/pnas.0409500102
Hung SI et al (2006) Genetic susceptibility to carbamazepine-induced cutaneous adverse drug reactions. Pharmacogenet Genom 16:297–306. doi:10.1097/01.fpc.0000199500.46842.4a
Hung SI et al (2010) Common risk allele in aromatic antiepileptic-drug induced Stevens-Johnson syndrome and toxic epidermal necrolysis in Han Chinese. Pharmacogenomics 11:349–356. doi:10.2217/pgs.09.162
Hunziker T, Bruppacher R, Kuenzi UP, Maibach R, Braunschweig S, Halter F, Hoigne RV (2002) Classification of ADRs: a proposal for harmonization and differentiation based on the experience of the Comprehensive Hospital Drug Monitoring Bern St. Gallen, 1974-1993. Pharmacoepidemiol Drug Saf 11:159–163. doi:10.1002/pds.669
Illing PT et al (2012) Immune self-reactivity triggered by drug-modified HLA-peptide repertoire. Nature 486:554–558. doi:10.1038/nature11147
Jaruthamsophon K, Tipmanee V, Sangiemchoey A, Sukasem C, Limprasert P (2017) HLA-B∗15:21 and carbamazepine-induced Stevens-Johnson syndrome: pooled-data and in silico analysis. Sci rep 7. doi:10.1038/srep45553
Jenkins RE, Meng X, Elliott VL, Kitteringham NR, Pirmohamed M, Park BK (2009) Characterisation of flucloxacillin and 5-hydroxymethyl flucloxacillin haptenated HSA in vitro and in vivo. Proteomics Clin Appl 3:720–729. doi:10.1002/prca.200800222
Johnson JA, Bootman JL (1995) Drug-related morbidity and mortality. A cost-of-illness model. Arch Intern Med 155:1949–1956. doi:10.1001/archinte.1995.00430180043006
Kang HR et al (2011) Positive and negative associations of HLA class I alleles with allopurinol-induced SCARs in Koreans. Pharmacogenet Genomics 21:303–307. doi:10.1097/FPC.0b013e32834282b8
Kaniwa N et al (2008) HLA-B locus in Japanese patients with anti-epileptics and allopurinol-related Stevens-Johnson syndrome and toxic epidermal necrolysis. Pharmacogenomics 9:1617–1622. doi:10.2217/14622416.9.11.1617
Kaniwa N et al (2010) HLA-B*1511 is a risk factor for carbamazepine-induced Stevens-Johnson syndrome and toxic epidermal necrolysis in Japanese patients. Epilepsia 51:2461–2465. doi:10.1111/j.1528-1167.2010.02766.x
Kim SH et al (2011) Carbamazepine-induced severe cutaneous adverse reactions and HLA genotypes in Koreans. Epilepsy res 97:190–197. doi:10.1016/j.eplepsyres.2011.08.010
Kim SH et al (2015) Characterization of amoxicillin- and clavulanic acid-specific T cells in patients with amoxicillin-clavulanate-induced liver injury. Hepatology 62:887–899. doi:10.1002/hep.27912
Ko TM et al (2011) Shared and restricted T-cell receptor use is crucial for carbamazepine-induced Stevens-Johnson syndrome. J Allergy Clin Immunol 128:1266–U1624. doi:10.1016/j.jaci.2011.08.013
Kulkantrakorn K et al (2012) HLA-B*1502 strongly predicts carbamazepine-induced Stevens-Johnson syndrome and toxic epidermal necrolysis in Thai patients with neuropathic pain. Pain Pract 12:202–208. doi:10.1111/j.1533-2500.2011.00479.x
Lazarou J, Pomeranz BH, Corey PN (1998) Incidence of adverse drug reactions in hospitalized patients—a meta-analysis of prospective studies. J am med Assoc 279:1200–1205. doi:10.1001/jama.279.15.1200
Lichtenfels M et al (2014) HLA restriction of carbamazepine-specific T-cell clones from an HLA-A*31:01-positive hypersensitive patient. Chem res Toxicol 27:175–177. doi:10.1021/tx400460w
Lin CH et al (2015) Immunologic basis for allopurinol-induced severe cutaneous adverse reactions: HLA-B∗58:01-restricted activation of drug-specific T cells and molecular interaction: to the editor. J Allergy Clin Immunol 135:1063–1065.e1065. doi:10.1016/j.jaci.2014.09.041
Locharernkul C et al (2008) Carbamazepine and phenytoin induced Stevens-Johnson syndrome is associated with HLA-B*1502 allele in Thai population. Epilepsia 49:2087–2091. doi:10.1111/j.1528-1167.2008.01719.x
Lonjou C et al (2008) A European study of HLA-B in Stevens-Johnson syndrome and toxic epidermal necrolysis related to five high-risk drugs. Pharmacogenet Genomics 18:99–107. doi:10.1097/FPC.0b013e3282f3ef9c
Lucena MI et al (2011) Susceptibility to amoxicillin-clavulanate-induced liver injury is influenced by multiple HLA class I and II alleles. Gastroenterology 141:338–347. doi:10.1053/j.gastro.2011.04.001
Macdonald WA et al (2009) T cell allorecognition via molecular mimicry. Immunity 31:897–908. doi:10.1016/j.immuni.2009.09.025
Mallal S et al (2002) Association between presence of HLA-B*5701, HLA-DR7, and HLA-DQ3 and hypersensitivity to HIV-1 reverse-transcriptase inhibitor abacavir. Lancet 359:727–732. doi:10.1016/S0140-6736(02)07873-X
Mallal S et al (2008) HLA-B*5701 screening for hypersensitivity to abacavir. New Engl J Med 358:568–579. doi:10.1056/NEJMoa0706135
Man CB, Kwan P, Baum L, Yu E, Lau KM, Cheng AS, Ng MH (2007) Association between HLA-B*1502 allele and antiepileptic drug-induced cutaneous reactions in Han Chinese. Epilepsia 48:1015–1018. doi:10.1111/j.1528-1167.2007.01022.x
Martin S, Weltzien HU (1994) T cell recognition of haptens, a molecular view. Int Arch Allergy Immunol 104:10–16. doi:10.1159/000236703
Martin AM et al (2004) Predisposition to abacavir hypersensitivity conferred by HLA-B*5701 and a haplotypic Hsp70-Hom variant. Proc Natl Acad Sci U S A 101:4180–4185. doi:10.1073/pnas.0307067101
McCormack M et al (2011) HLA-A*3101 and carbamazepine-induced hypersensitivity reactions in Europeans. New Engl J Med 364:1134–1143. doi:10.1056/NEJMoa1013297
McCormack M et al (2012) Genome-wide mapping for clinically relevant predictors of lamotrigine- and phenytoin-induced hypersensitivity reactions. Pharmacogenomics 13:399–405. doi:10.2217/pgs.11.165
Mehta TY et al (2009) Association of HLA-B*1502 allele and carbamazepine-induced Stevens-Johnson syndrome among Indians. Indian J Dermatol Venereol Leprol 75:579–582. doi:10.4103/0378-6323.57718
Meng X et al (2011) Direct evidence for the formation of diastereoisomeric benzylpenicilloyl haptens from benzylpenicillin and benzylpenicillenic acid in patients. J Pharmacol Exp Ther 338:841–849. doi:10.1124/jpet.111.183871
Meng X et al (2017) Definition of the nature and hapten threshold of the β-lactam antigen required for t cell activation in vitro and in patients. J Immunol. doi:10.4049/jimmunol.1700209
Monshi M et al (2012) HLA-B*57:01-restricted activation of drug-specific T-cells provides the immunological basis for flucloxacillin-induced liver injury. Hepatology 57:727–739. doi:10.1002/hep.26077
Naisbitt DJ et al (2015) Towards depersonalized abacavir therapy: chemical modification eliminates HLA-B*57: 01-restricted CD8+ T-cell activation. Aids 29:2385–2395. doi:10.1097/qad.0000000000000867
Nassif A et al (2004) Toxic epidermal necrolysis: effector cells are drug-specific cytotoxic T cells. J Allergy Clin Immunol 114:1209–1215. doi:10.1016/j.jaci.2004.07.047
Ng CY et al (2016) Impact of the HLA-B*58:01 allele and renal impairment on allopurinol-induced cutaneous adverse reactions. J Invest Dermatol 136:1373–1381. doi:10.1016/j.jid.2016.02.808
Norcross MA et al (2012) Abacavir induces loading of novel self-peptides into HLA-B*57: 01: an autoimmune model for HLA-associated drug hypersensitivity. Aids 26:F21–F29. doi:10.1097/QAD.0b013e328355fe8f
Ostrov DA et al (2012) Drug hypersensitivity caused by alteration of the MHC-presented self-peptide repertoire. Proc Natl Acad Sci U S A 109:9959–9964. doi:10.1073/pnas.1207934109
Ozeki T et al (2011) Genome-wide association study identifies HLA-A*3101 allele as a genetic risk factor for carbamazepine-induced cutaneous adverse drug reactions in Japanese population. Hum Mol Genet 20:1034–1041. doi:10.1093/hmg/ddq537
Padovan E, Bauer T, Tongio MM, Kalbacher H, Weltzien HU (1997) Penicilloyl peptides are recognized as T cell antigenic determinants in penicillin allergy. Eur J Immunol 27:1303–1307. doi:10.1002/eji.1830270602
Phillips EJ, Sandra RS, Sullivan NH, John RK (2002) Utility of patch testing in patients with hypersensitivity syndromes associated with abacavir. Aids 16:2223–2225. doi:10.1097/00002030-200211080-00017
Pichler WJ (2003) Delayed drug hypersensitivity reactions. Ann Intern Med 139:683–693. doi:10.7326/0003-4819-139-8-200310210-00012
Pichler W, Yawalkar N, Schmid S, Helbling A (2002) Pathogenesis of drug-induced exanthems. Allergy 57:884–893. doi:10.1034/j.1398-9995.2002.02161.x
Plumpton CO, Yip VLM, Alfirevic A, Marson AG, Pirmohamed M, Hughes DA (2015) Cost-effectiveness of screening for HLA-A∗31:01 prior to initiation of carbamazepine in epilepsy. Epilepsia 56:556–563. doi:10.1111/epi.12937
Pymm P et al (2017) MHC-I peptides get out of the groove and enable a novel mechanism of HIV-1 escape. Nat Struct Mol Biol 24:387–394. doi:10.1038/nsmb.3381
Reche PA, Reinherz EL (2003) Sequence variability analysis of human class I and class II MHC molecules: functional and structural correlates of amino acid polymorphisms. J Mol Biol 331:623–641. doi:10.1016/S0022-2836(03)00750-2
Robinson J, Halliwell JA, Hayhurst JD, Flicek P, Parham P, Marsh SG (2015) The IPD and IMGT/HLA database: allele variant databases. Nucleic Acids res 43:D423–D431. doi:10.1093/nar/gku1161
Roediger B, Weninger W (2011) How nickel turns on innate immune cells. Immunol Cell Biol 89:1–2. doi:10.1038/icb.2010.114
Roughead EE, Semple SJ (2009) Medication safety in acute care in Australia: where are we now? Part 1: a review of the extent and causes of medication problems 2002-2008. Aust New Zealand Health Policy 6. doi:10.1186/1743-8462-6-18
Saag M et al (2008) High sensitivity of human leukocyte antigen-B*5701 as a marker for immunologically confirmed abacavir hypersensitivity in white and black patients. Clin Infect dis 46:1111–1118. doi:10.1086/529382
Schaid DJ et al (2014) Prospective validation of HLA-DRB1*07:01 allele carriage as a predictive risk factor for lapatinib-induced liver injury. J Clin Oncol 32:2296–2303. doi:10.1200/JCO.2013.52.9867
Schnyder B, MauriHellweg D, Zanni M, Bettens F, Pichler WJ (1997) Direct, MHC-dependent presentation of the drug sulfamethoxazole to human alpha beta T cell clones. J Clin Invest 100:136–141. doi:10.1172/JCI119505
Schrijvers R, Gilissen L, Chiriac AM, Demoly P (2015) Pathogenesis and diagnosis of delayed-type drug hypersensitivity reactions, from bedside to bench and back. Clinical and Translational Allergy 5:31. doi:10.1186/s13601-015-0073-8
Shi YW et al (2012) Association between HLA and Stevens-Johnson syndrome induced by carbamazepine in Southern Han Chinese: genetic markers besides B*1502? Basic Clin Pharmacol Toxicol 111:58–64. doi:10.1111/j.1742-7843.2012.00868.x
Small CB, Margolis DA, Shaefer MS, Ross LL (2017) HLA-B*57:01 allele prevalence in HIV-infected North American subjects and the impact of allele testing on the incidence of abacavir-associated hypersensitivity reaction in HLA-B*57:01-negative subjects. BMC Infect Dis 17:256. doi:10.1186/s12879-017-2331-y
Somkrua R, Eickman EE, Saokaew S, Lohitnavy M, Chaiyakunapruk N (2011) Association of HLA-B*5801 allele and allopurinol-induced Stevens Johnson syndrome and toxic epidermal necrolysis: a systematic review and meta-analysis. BMC Medical Genetics 12:118. doi:10.1186/1471-2350-12-118
Spraggs CF et al (2011) HLA-DQA1*02:01 is a major risk factor for lapatinib-induced hepatotoxicity in women with advanced breast cancer. J Clin Oncol 29:667–673. doi:10.1200/jco.2010.31.3197
Stephens C et al (2013) HLA alleles influence the clinical signature of amoxicillin-clavulanate hepatotoxicity. PLoS One 8:e68111. doi:10.1371/journal.pone.0068111
Tangamornsuksan W, Chaiyakunapruk N, Somkrua R, Lohitnavy M, Tassaneeyakul W (2013) Relationship between the HLA-B*1502 allele and carbamazepine-induced Stevens-Johnson syndrome and toxic epidermal necrolysis: a systematic review and meta-analysis. JAMA Dermatology 149:1025–1032. doi:10.1001/jamadermatol.2013.4114
Tassaneeyakul W et al (2009) Strong association between HLA-B*5801 and allopurinol-induced Stevens-Johnson syndrome and toxic epidermal necrolysis in a Thai population. Pharmacogenet Genom 19:704–709. doi:10.1097/FPC.0b013e328330a3b8
Tassaneeyakul W et al (2010) Association between HLA-B*1502 and carbamazepine-induced severe cutaneous adverse drug reactions in a Thai population. Epilepsia 51:926–930
Tassaneeyakul W et al (2016) Associations between HLA class I and cytochrome P450 2C9 genetic polymorphisms and phenytoin-related severe cutaneous adverse reactions in a Thai population. Pharmacogenet Genomics 26:225–234. doi:10.1097/fpc.0000000000000211
Then SM, Rani ZZ, Raymond AA, Ratnaningrum S, Jamal R (2011) Frequency of the HLA-B*1502 allele contributing to carbamazepine-induced hypersensitivity reactions in a cohort of Malaysian epilepsy patients. Asian Pac J Allergy Immunol 29:290–293
Tohkin M et al (2013) A whole-genome association study of major determinants for allopurinol-related Stevens-Johnson syndrome and toxic epidermal necrolysis in Japanese patients. Pharmacogenomics J 13:60–69. doi:10.1038/tpj.2011.41
Toksoy A, Sennefelder H, Adam C, Hofmann S, Trautmann A, Goebeler M, Schmidt M (2017) Potent NLRP3 inflammasome activation by the HIV reverse transcriptase inhibitor abacavir. J Biol Chem 292:2805–2814. doi:10.1074/jbc.M116.749473
Wang Q et al (2011) Association between HLA-B*1502 allele and carbamazepine-induced severe cutaneous adverse reactions in Han people of southern China mainland. Seizure 20:446–448. doi:10.1016/j.seizure.2011.02.003
Watkins S, Pichler WJ (2013) Sulfamethoxazole induces a switch mechanism in T cell receptors containing TCRVβ20-1, altering pHLA recognition. PLoS One 8:e76211. doi:10.1371/journal.pone.0076211
Wei C-Y, Chung W-H, Huang H-W, Chen Y-T, Hung S-I (2012) Direct interaction between HLA-B and carbamazepine activates T cells in patients with Stevens-Johnson syndrome. J Allergy Clin Immunol 129:1562–1569. doi:10.1016/j.jaci.2011.12.990
Whitaker P et al (2011) Mass spectrometric characterization of circulating and functional antigens derived from piperacillin in patients with cystic fibrosis. J Immunol 187:200–211. doi:10.4049/jimmunol.1100647
WHO (1972) International drug monitoring: the role of national centres. Report of a WHO meeting vol 498
Wu XT et al (2010) Association between carbamazepine-induced cutaneous adverse drug reactions and the HLA-B*1502 allele among patients in central China. Epilepsy Behav 19:405–408. doi:10.1016/j.yebeh.2010.08.007
Wuillemin N, Adam J, Fontana S, Krähenbühl S, Pichler WJ, Yerly D (2013) HLA haplotype determines hapten or p-i T cell reactivity to flucloxacillin. J Immunol 190:4956–4964. doi:10.4049/jimmunol.1202949
Wuillemin N et al (2014) T cells infiltrate the liver and kill hepatocytes in HLA-B∗57:01-associated floxacillin-induced liver injury. Am J Pathol 184:1677–1682. doi:10.1016/j.ajpath.2014.02.018
Xu CF et al (2016) HLA-B∗57:01 confers susceptibility to pazopanib-associated liver injury in patients with cancer. Clin Cancer res 22:1371–1377. doi:10.1158/1078-0432.CCR-15-2044
Yang C-WO et al (2007) HLA-B*1502-bound peptides: implications for the pathogenesis of carbamazepine-induced Stevens-Johnson syndrome. J Allergy Clin Immunol 120:870–877. doi:10.1016/j.jaci.2007.06.017
Yaseen FS et al (2015) Promiscuous T-cell responses to drugs and drug-haptens. J Allergy Clin Immunol 136:474–476. doi:10.1016/j.jaci.2015.02.036
Yip VL, Pirmohamed M (2017) The HLA-A*31:01 allele: influence on carbamazepine treatment. Pharmgenomics Pers Med 10:29–38. doi:10.2147/pgpm.s108598
Yun J, Marcaida MJ, Eriksson KK, Jamin H, Fontana S, Pichler WJ, Yerly D (2014) Oxypurinol directly and immediately activates the drug-specific T cells via the preferential use of HLA-B*58:01. J Immunol 197:2984–2993. doi:10.4049/jimmunol.1302306
Yun J, Mattsson J, Schnyder K, Fontana S, Largiadèr CR, Pichler WJ, Yerly D (2013) Allopurinol hypersensitivity is primarily mediated by dose dependent oxypurinol-specific T cell response. Clin Exp Allergy 43:1246–1255. doi:10.1111/cea.12184
Zanni MP et al (1998) HLA-restricted, processing- and metabolism-independent pathway of drug recognition by human alpha beta T lymphocytes. J Clin Invest 102:1591–1598. doi:10.1172/JCI3544
Zhang Y et al (2011) Strong association between HLA-B*1502 and carbamazepine-induced Stevens-Johnson syndrome and toxic epidermal necrolysis in mainland Han Chinese patients. Eur J Clin Pharmacol 67:885–887. doi:10.1007/s00228-011-1009-4
Zhang FR et al (2013) HLA-B*13:01 and the dapsone hypersensitivity syndrome. New Engl J med 369:1620–1628. doi:10.1056/NEJMoa1213096
Zhou P, Zhang S, Wang Y, Yang C, Huang J (2016) Structural modeling of HLA-B*1502/peptide/carbamazepine/T-cell receptor complex architecture: implication for the molecular mechanism of carbamazepine-induced Stevens-Johnson syndrome/toxic epidermal necrolysis. J Biomol Struct Dyn 34:1806–1817. doi:10.1080/07391102.2015.1092476
Acknowledgments
PTI is supported by an Australian National Health and Medical Research Council (NHMRC) Early Career Fellowship (1072159). AWP is supported by a NHMRC Senior Research Fellowship (1044215) and a NHMRC project grant (1122099). JMc was supported by a NHMRC Project Grant (1120467) and Program Grant (1113293). Thank you to Julian Vivian and Jamie Rossjohn for assistance with structural figures.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published in the Special Issue MHC Genes and Their Ligands in Health and Disease with Editor Prof. Ronald Bontrop.
Rights and permissions
About this article
Cite this article
Illing, P.T., Purcell, A.W. & McCluskey, J. The role of HLA genes in pharmacogenomics: unravelling HLA associated adverse drug reactions. Immunogenetics 69, 617–630 (2017). https://doi.org/10.1007/s00251-017-1007-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-017-1007-5