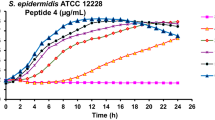
Abstract
Bacterial biofilms are associated with a large number of infections. Biofilm-dwelling bacteria are particularly resistant to antibiotics, making it hard to eradicate biofilm-associated infections. Here, we use a novel cross-disciplinary approach combining microbiology and chemoinformatics to identify new and efficient anti-biofilm drugs. We found that ellagic acid (present in green tea) significantly inhibited biofilm formation of Streptococcus dysgalactiae. Based on ellagic acid, we performed in silico screening of the Chinese Natural Product Database to predict a 2nd-generation list of compounds with similar characteristics. One of these, esculetin, proved to be more efficient in preventing biofilm formation by Staphylococcus aureus. From esculetin a 3rd-generation list of compounds was predicted. One of them, fisetin, was even better to abolish biofilm formation than the two parent compounds. Fisetin dramatically inhibited biofilm formation of both S. aureus and S. dysgalactiae. The compounds did not affect planktonic growth in concentrations where they affected biofilm formation and appeared to be specific antagonists of biofilms. Arguably, since all three compounds are natural ingredients of dietary plants, they should be well-tolerated by humans. Our results indicate that such small plant components, with bacterial lifestyle altering properties are promising candidates for novel generations of antimicrobial drugs. The study underlines the potential in combining chemoinformatics and biofilm research.




Similar content being viewed by others
References
Ahn YJ, Kawamura T, Kim M, Yamamoto T, Mitsuoka T (1991) Tea polyphenols: selective growth inhibitors of Clostridium spp. Agric Biol Chem 55:1425–1426
Akiyama H, Fujii K, Yamasaki O, Oono T, Iwatsuki K (2001) "Antibacterial action of several tannins against Staphylococcus aureus" J. Antimicrob Chemother 48:487–491
Anderl JN, Franklin MJ, Stewart PS (2000) Role of antibiotic penetration limitation in Klebsiella pneumoniae biofilm resistance to ampicillin and ciprofloxacin. Antimicrob Agents Chemother 44:1818–1824
Arai Y, Watanabe S, Kimira M, Shimoi K, Mochizuki R, Kinae N (2000) Dietary intakes of flavonols, flavones and isoflavones by Japanese women and the inverse correlation between quercetin intake and plasma LDL cholesterol concentration. J Nutr 130:2243–2250
Bradley A (2002) Bovine mastitis: an evolving disease. Vet J 164:116–128
Cespedes CL, Avila JG, Martinez A, Serrato B, Calderon-Mugica JC, Salgado-Garciglia R (2006) Antifungal and antibacterial activities of Mexican tarragon (Tagetes lucida). J Agric Food Chem 54:3521–3527
Chemical Computing Group I. (2007) Molecular operating environment, 2007.09 ed. Montreal Quebec
Christensen BB, Sternberg C, Andersen JB, Palmer RJ Jr, Nielsen AT, Givskov M, Molin S (1999) Molecular tools for study of biofilm physiology. Methods Enzymol 310:20–42
Coetser SE, Cloete TE (2005) Biofouling and biocorrosion in industrial water systems. Crit Rev Microbiol 31:213–232
Costerton JW, Lewandowski Z, Caldwell DE, Korber DR, Lappin-Scott HM (1995) Microbial biofilms. Annu Rev Microbiol 49:711–745
Costerton JW, Stewart PS, Greenberg EP (1999) Bacterial biofilms: a common cause of persistent infections. Science 284:1318–1322
Donlan RM (2002) Biofilms: microbial life on surfaces. Emerg Infect Dis 8:881–890
Duncan SH, Flint HJ, Stewart CS (1998) Inhibitory activity of gut bacteria against Escherichia coli O157 mediated by dietary plant metabolites. FEMS Microbiol Lett 164:283–288
Duncan SH, Leitch EC, Stanley KN, Richardson AJ, Laven RA, Flint HJ, Stewart CS (2004) Effects of esculin and esculetin on the survival of Escherichia coli O157 in human faecal slurries, continuous-flow simulations of the rumen and colon and in calves. Br J Nutr 91:749–755
Durant JL, Leland BA, Henry DR, Nourse JG (2002) Reoptimization of MDL keys for use in drug discovery. J Chem Inf Comput Sci 42:1273–1280
Ehrman TM, Barlow DJ, Hylands PJ (2007) Phytochemical informatics of traditional Chinese medicine and therapeutic relevance. J Chem Inf Model 47:2316–2334
Ferrières L, Hancock V, Klemm P (2007) Biofilm exclusion of uropathogenic bacteria by selected asymptomatic bacteriuria Escherichia coli strains. Microbiology 153:1711–1719
Frees D, Chastanet A, Qazi S, Sorensen K, Hill P, Msadek T, Ingmer H (2004) Clp ATPases are required for stress tolerance, intracellular replication and biofilm formation in Staphylococcus aureus. Mol Microbiol 54:1445–1462
Fylaktakidou KC, Hadjipavlou-Litina DJ, Litinas KE, Nicolaides DN (2004) Natural and synthetic coumarin derivatives with anti-inflammatory/antioxidant activities. Curr Pharm Des 10:3813–3833
Hamilton-Miller JMT (1995) Antimicrobial properties of tea (Camellia sinensis L.). Antimicrob Agents Chemother 39:2375–2377
Hausen BM, Schmieder M (1986) The sensitizing capacity of coumarins (I). Contact Dermatitis 15:157–163
Heydorn A, Nielsen AT, Hentzer M, Sternberg C, Givskov M, Ersboll BK, Molin S (2000) Quantification of biofilm structures by the novel computer program COMSTAT. Microbiology 146:2395–2407
Ishige K, Schubert D, Sagara Y (2001) Flavonoids protect neuronal cells from oxidative stress by three distinct mechanisms. Free Radic Biol Med 30:433–446
Jones SM, Morgan M, Humphrey TJ, Lappin-Scott H (2001) Effect of vancomycin and rifampicin on methicillin-resistant Staphylococcus aureus biofilms. Lancet 357:40–41
Kawai M, Hirano T, Higa S, Arimitsu J, Maruta M, Kuwahara Y, Ohkawara T, Hagihara K, Yamadori T, Shima Y, Ogata A, Kawase I, Tanaka T (2007) Flavonoids and related compounds as anti-allergic substances. Allergol Int 56:113–123
Kim SH, Kang KA, Zhang R, Piao MJ, Ko DO, Wang ZH, Chae SW, Kang SS, Lee KH, Kang HK, Kang HW, Hyun JW (2008) Protective effect of esculetin against oxidative stress-induced cell damage via scavenging reactive oxygen species. Acta Pharmacol Sin 29:1319–1326
Klemm P, Hancock V, Kvist M, Schembri M (2007) Candidate targets for new antivirulence drugs: selected cases of bacterial adhesion and biofilm formation. Future Microbiol 2:643–653
Lewis K (2001) Riddle of biofilm resistance. Antimicrob Agents Chemother 45:999–1007
Li M, Ni N, Chou HT, Lu CD, Tai PC, Wang B (2008) Structure-based discovery and experimental verification of novel AI-2 quorum sensing inhibitors against Vibrio harveyi. ChemMedChem 3:1242–1249
Martin-Aragon S, Benedi JM, Villar AM (1998) Effects of the antioxidant (6, 7-dihydroxycoumarin) esculetin on the glutathione system and lipid peroxidation in mice. Gerontology 44:21–25
Melchior MB, Vaarkamp H, Fink-Gremmels J (2006) Biofilms: a role in recurrent mastitis infections? Vet J 171:398–407
National Committee for Clinical Laboratory Standards (1997) Methods for dilution antimicrobial susceptility tests for bacteria that grow aerobically, 4th edn. NCCLS document M7-A4. National Committeefor Clinical Laboratory Standards, Wayne, PA
Otto M (2008) Staphylococcal biofilms. Curr Top Microbiol Immunol 322:207–228
Qin Z, Zhang J, Xu B, Chen L, Wu Y, Yang X, Shen X, Molin S, Danchin A, Jiang H, Qu D (2006) Structure-based discovery of inhibitors of the YycG histidine kinase: new chemical leads to combat Staphylococcus epidermidis infections. BMC Microbiol 6:96
Sagara Y, Vanhnasy J, Maher P (2004) Induction of PC12 cell differentiation by flavonoids is dependent upon extracellular signal-regulated kinase activation. J Neurochem 90:1144–1155
Shen J, Xu X, Cheng F, Liu H, Luo X, Shen J, Chen K, Zhao W, Shen X, Jiang H (2003) Virtual screening on natural products for discovering active compounds and target information. Curr Med Chem 10:2327–2342
Silván A, Abad M, Bermejo P, Sollhuber M, Villar A (1996) Antiinflammatory activity of coumarins from Santolina oblongifolia. J Nat Prod 59:1183–1185
Speziale P, Visai L, Rindi S, Pietrocola G, Provenza G, Provenzano M (2008) Prevention and treatment of Staphylococcus biofilms. Curr Med Chem 15:3185–3195
Stagg GV, Millin DJ (1975) The nutritional and therapeutic value of tea—a review. J Sci Food Agric 26:1439–1459
Syed DN, Suh Y, Afaq F, Mukhtar H (2008) Dietary agents for chemoprevention of prostate cancer. Cancer Lett 265:167–176
Tanimoto T (1957) IBM internal report. Technical report, IBM
Warren JW (2001) Catheter-associated urinary tract infections. Int J Antimicrob Agents 17:299–303
Yam TS, Shah S, Hamilton-Miller JMT (1997) Microbiological activity of whole and fractionated crude extracts of tea (Camellia sinensis), and of tea components. FEMS Microbiol Lett 152:169–174
Zeng Z, Qian L, Cao L, Tan H, Huang Y, Xue X, Shen Y, Zhou S (2008) Virtual screening for novel quorum sensing inhibitors to eradicate biofilm formation of Pseudomonas aeruginosa. Appl Microbiol Biotechnol 79:119–126
Acknowledgements
We thank Birthe Jul Jørgensen for expert technical assistance and Olivier Taboureau for fruitful discussions and critical comments. This work was supported by grants from the Danish Medical Research Council (Grant 271-07-0291), the Danish Research Council for Technology and Production Sciences (Grant 274-06-0301), and Lundbeckfonden (Grant R19-A2191).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Fig. S1
1st-generation hits from CNPD with a higher than 90% similarity to query natural compound ellagic acid (Tanimoto coefficient ≥0.9).CAS registry numbers in brackets [] correspond to hits with chemical structures that are close analogs to the structure depicted. (GIF 263 kb)
Fig. S2
2nd-generation hits from CNPD with a higher than 90% similarity to query natural compound esculetin (Tanimoto coefficient ≥0.9). CAS registry numbers in brackets [] correspond to chemical structures that are close analogs to the structure depicted. (When more than two analog compounds are found, their CAS registry numbers are given as footnote.) (GIF 296 kb)
Rights and permissions
About this article
Cite this article
Dürig, A., Kouskoumvekaki, I., Vejborg, R.M. et al. Chemoinformatics-assisted development of new anti-biofilm compounds. Appl Microbiol Biotechnol 87, 309–317 (2010). https://doi.org/10.1007/s00253-010-2471-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-010-2471-0