Abstract
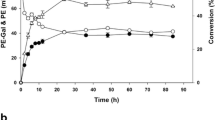
In this work, the β-galactosidase from Enterobacter cloacae B5 (BgaB5) exhibited excellent transglycosylation activity toward tyrosol (p-hydroxyphenethyl alcohol) when using lactose as the glycosyl donor, generating a series of tyrosol glycosides with potential pharmacological properties. The effects of substrate concentration, temperature, pH, and reaction time on the transglycosylation reaction catalyzed by the enzyme BgaB5 were studied in detail. Three tyrosol derivatives were produced in a total high yield of 50.0% when incubating the enzyme with 250 mM tyrosol and 1000 mM lactose (pH 7.5) at 50 °C for 5 min. These derivatives were subsequently purified by column chromatography and preparative thin-layer chromatography. MS analysis of the purified compounds suggested one monogalactoside (M r 300) and two digalactoside derivatives (M r 462). The following NMR analysis further identified them to be p-hydroxyphenethyl β-D-galactopyranoside, p-hydroxyphenethyl β-D- galactopyranosyl-(1 → 3′)-β-D-galactopyranoside, and p-hydroxyphenethyl β-D- galactopyranosyl-(1 → 6′)-β-D-galactopyranoside, respectively. The yield of the tyrosol monogalactoside which was known to possess potent bioactivities reached 39.4%, higher than other enzymatic yields reported so far. The two digalactosides, which were expected to have potential applications for novel drug screening and discovery, were artificially obtained with 10.6% yield for the first time.





Similar content being viewed by others
References
Amsterdam JD, Panossian AG (2016) Rhodiola rosea L. as a putative botanical antidepressant. Phytomedicine 23:770–783. doi:10.1016/j.phymed.2016.02.009
Bojarová P, Kren V (2011) Glycosidases in carbohydrate synthesis: when organic chemistry falls short. Chimia (Aarau) 65:65–70. doi:10.2533/chimia.2011.65
Chang X, Zhang K, Zhou R, Luo F, Zhu L, Gao J, He H, Wei T, Yan T, Ma C (2016) Cardioprotective effects of salidroside on myocardial ischemia-reperfusion injury in coronary artery occlusion-induced rats and Langendorff-perfused rat hearts. Int J Cardiol 215:532–544. doi:10.1016/j.ijcard.2016.04.108
Cieśla Ł, Moaddel R (2016) Comparison of analytical techniques for the identification of bioactive compounds from natural products. Nat Prod Rep 33:1131–1145. doi:10.1039/c6np00016a
Díaz Lanza AM, Abad Martínez MJ, Fernández Matellano L, Recuero Carretero C, Villaescusa Castillo L, Silván Sen AM, Bermejo Benito P (2001) Lignan and phenylpropanoid glycosides from Phillyrea latifolia and their in vitro anti-inflammatory activity. Planta Med 67:219–223. doi:10.1055/s-2001-12004
Gosling A, Stevens GW, Barber AR, Kentish SE, Gras SL (2010) Recent advances refining galactooligosaccharide production from lactose. Food Chem 121:307–318. doi:10.1016/j.foodchem.2009.12.063
Griffith BR, Langenhan JM, Thorson JS (2005) ‘Sweetening’ natural products via glycorandomization. Curr Opin Biotechnol 16:622–630. doi:10.1016/j.copbio.2005.10.002
Jin H, Pei L, Shu X, Yang X, Yan T, Wu Y, Wei N, Yan H, Wang S, Yao C, Liu D, Tian Q, Wang L, Lu Y (2016) Therapeutic intervention of learning and memory decays by salidroside stimulation of neurogenesis in aging. Mol Neurobiol 53:851–866. doi:10.1007/s12035-014-9045-6
Kim GE, Lee JH, Jung SH, Seo ES, Jin SD, Kim GJ, Cha J, Kim EJ, Park KD, Kim D (2010) Enzymatic synthesis and characterization of hydroquinone galactoside using Kluyveromyces lactis lactase. J Agric Food Chem 58:9492–9497. doi:10.1021/jf101748j
Langenhan JM, Griffith BR, Thorson JS (2005) Neoglycorandomization and chemoenzymatic glycorandomization: two complementary tools for natural product diversification. J Nat Prod 68:1696–1711. doi:10.1021/np0502084
Liu H, Lv P, Zhu Y, Wu H, Zhang XF, Zheng L, Zhao J (2017) Salidroside promotes peripheral nerve regeneration based on tissue engineering strategy using Schwann cells and PLGA: in vitro and in vivo. Sci Rep 7:39869. doi:10.1038/srep39869
Lu L, Xiao M, Li ZY, Li YM, Wang F (2009) A novel transglycosylating β-galactosidase from Enterobacter cloacae B5. Process Biochem 44:232–236. doi:10.1016/j.biortech.2010.03.106
Lu L, Xu X, Gu G, Jin L, Xiao M, Wang F (2010) Synthesis of novel galactose containing chemicals by beta-galactosidase from Enterobacter cloacae B5. Bioresour Technol 101:6868–6872. doi:10.1016/j.biortech.2010.03.106
Lu L, Xu S, Jin L, Zhang D, Li Y, Xiao M (2012) Synthesis of galactosyl sucralose by β-galactosidase from Lactobacillus bulgaricus L3. Food Chem 134:269–275. doi:10.1016/j.foodchem.2012.02.134
Lu L, Xu LJ, Guo Y, Zhang D, Qi T, Jin L, Gu G, Xu L, Xiao M (2015) Glycosylation of phenolic compounds by the site-mutated β-galactosidase from Lactobacillus bulgaricus L3. PLoS One 10:e0121445. doi:10.1371/journal.pone.0121445
Makowski K, Białkowskaa A, Olczakb J, Kurc J, Turkiewicza M (2009) Antarctic, cold-adapted beta-galactosidase of Pseudoalteromonas sp. 22b as an effective tool for alkyl galactopyranosides synthesis. Enzym Microb Technol 44:59–64. doi:10.1016/j.enzmictec.2008.09.010
Nabavi SF, Braidy N, Orhan IE, Badiee A, Daglia M, Nabavi SM (2016) Rhodiola rosea L. and Alzheimer’s disease: from farm to pharmacy. Phytother Res 30:532–539. doi:10.1002/ptr.5569
Oliveira C, Guimarães PM, Domingues L (2011) Recombinant microbial systems for improved β-galactosidase production and biotechnological applications. Biotechnol Adv 29:600–609. doi:10.1016/j.biotechadv.2011.03.008
Panossian A, Wikman G, Sarris J (2010) Rosenroot (Rhodiola rosea): traditional use, chemical composition, pharmacology and clinical efficacy. Phytomedicine 17:481–493. doi:10.1016/j.phymed.2010.02.002
Park AR, Oh DK (2010) Galacto-oligosaccharide production using microbial beta-galactosidase: current state and perspectives. Appl Microbiol Biotechnol 85:1279–1286. doi:10.1007/s00253-009-2356-2
Porciúncula González C, Castillaa A, Garófalob L, Souleb S, Irazoquia G, Giacominia C (2013) Enzymatic synthesis of 2-aminoethyl β-D-galactopyranoside catalyzed by Aspergillus oryzae β-galactosidase. arbohyd Res 368:104–110. doi:10.1016/j.carres.2012.12.009
Potocká E, Mastihubová M, Mastihuba V (2015) Enzymatic synthesis of tyrosol glycosides. J Mol Catal B-Enzym 113:23–28. doi:10.1016/j.biortech.2011.10.073
Qi Z, Qi S, Ling L, Lv J, Feng Z (2016) Salidroside attenuates inflammatory response via suppressing JAK2-STAT3 pathway activation and preventing STAT3 transfer into nucleus. Int Immunopharmacol 35:265–271. doi:10.1016/j.intimp.2016.04.004
Qian EW, Ge DT, Kong SK (2012) Salidroside protects human erythrocytes against hydrogen peroxide-induced apoptosis. J Nat Prod 75:531–537. doi:10.1021/np200555s
Sears P, Wong CH (2001) Toward automated synthesis of oligosaccharides and glycoproteins. Science 291:2344–2350. doi:10.1126/science.1058899
Seo S, Rebehmed J, de Brevern AG, Karboune S (2015) Enzymatic synthesis of galactosylated serine/threonine derivatives by β-galactosidase from Escherichia coli. Int J Mol Sci 16:13714–13728. doi:10.3390/ijms160613714
Shi T, Chen H, Jing L, Liu X, Sun X, Jiang R (2011) Development of a kilogram-scale synthesis of salidroside and its analogues. Synth Commun 41:2594–2600. doi:10.1080/00397911.2010.515332
Shi TY, Feng SF, Xing JH, Wu YM, Li XQ, Zhang N, Tian Z, Liu SB, Zhao MG (2012) Neuroprotective effects of Salidroside and its analogue tyrosol galactoside against focal cerebral ischemia in vivo and H2O2-induced neurotoxicity in vitro. Neurotox Res 21:358–367. doi:10.1007/s12640-011-9290-7
Shimizu R, Shimabayashi H, Moriwaki M (2006) Enzymatic production of highly soluble myricitrin glycosides using beta-galactosidase. Biosci Biotechnol Biochem 70:940–948. doi:10.1271/bbb.70.940
Si PP, Zhen JL, Cai YL, Wang WJ, Wang WP (2016) Salidroside protects against kainic acid-induced status epilepticus via suppressing oxidative stress. Neurosci Lett 618:19–24. doi:10.1016/j.neulet.2016.02.056
Zhang J, Lu L, Lu L, Zhao Y, Kang L, Pang X, Liu J, Jiang T, Xiao M, Ma B (2016) Galactosylation of steroidal saponins by β-galactosidase from Lactobacillus bulgaricus L3. Glycoconj J33:53–62. doi:10.1007/s10719-015-9632-4
Zhu L, Chen T, Chang X, Zhou R, Luo F, Liu J, Zhang K, Wang Y, Yang Y, Long H, Liu Y, Yan T, Ma C (2016) Salidroside ameliorates arthritis-induced brain cognition deficits by regulating rho/ROCK/NF-κB pathway. Neuropharmacology 103:134–142. doi:10.1016/j.neuropharm.2015.12.007
Acknowledgments
Science and Technology Development Project of Shandong Province (No. 2015GSF121004, 2014GSF121006), National Natural Science Foundation of China (No. 31670062, No. 31000035), and Fundamental Research Funds of Shandong University (No. 2016JC028).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 544 kb)
Rights and permissions
About this article
Cite this article
Qi, T., Gu, G., Xu, L. et al. Efficient synthesis of tyrosol galactosides by the β-galactosidase from Enterobacter cloacae B5. Appl Microbiol Biotechnol 101, 4995–5003 (2017). https://doi.org/10.1007/s00253-017-8249-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8249-x