Abstract
Purpose
CD44 is overexpressed in various tumors including hepatocellular carcinoma (HCC). The purpose of this study was to examine the effects of CD44 antisense oligonucleotide (ASO) alone or combination with doxorubicin on HCC cells in vitro.
Methods
Cytotoxicity was measured by use of a cell viability assay in HCC cell line SNU-449. Tumorigenesis and invasion were accessed by colony formation, growth in soft agar and ECMatrix invasion assay. Apoptosis and necrosis were evaluated by using double staining with Hoechst 33342 and propidium iodide. Protein expression and mRNA level were detected by Western blot and RT-PCR.
Results
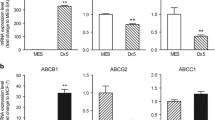
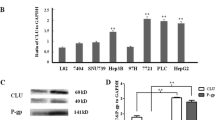
We have designed novel CD44 ASO, which can effectively down-regulate CD44 expression in SNU-449. Colony formation, growth in soft agar and invasion were significantly impaired after CD44 ASO treatment in SNU-499. In company with CD44 down-regulated by CD44 ASO, MDR-1 and Bcl-2 expression were also greatly reduced. CD44 ASO also increased chemosensitivity to doxorubicin significantly, lowered IC50 by one order of magnitude. Apoptosis and necrosis were also induced by CD44 ASO alone or in combination treatment with doxorubicin.
Conclusions
Inhibition of CD44 expression by CD44 ASO significantly induced apoptosis, decreased tumorigenesis and invasion, and increased chemosensitivity. Thus, CD44 ASO is potentially a therapy that is worth investigating in the clinical setting.






Similar content being viewed by others
References
Llovet JM, Beaugrand M (2003) Hepatocellular carcinoma: present status and future prospects. J Hepatol 38:S136–S149
Bruix J, Boix L, Sala M, Llovet JM (2004) Focus on hepatocellular carcinoma. Cancer Cell 5:215–219
Greten TF, Papendorf F, Bleck JS, Kirchhoff T, Wohlberedt T, Kubicka S et al (2005) Survival rate in patients with hepatocellular carcinoma: a retrospective analysis of 389 patients. Br J Cancer 92:1862–1868
Avila MA, Berasain C, Sangro B, Prieto J (2006) New therapies for hepatocellular carcinoma. Oncogene 25:3866–3884
Greten TF, Manns MP, Korangy F (2006) Immunotherapy of hepatocellular carcinoma. J Hepatol 45:868–878
Abou-Alfa GK (2006) Hepatocellular carcinoma: molecular biology and therapy. Semin Oncol 33:S79–S83
Zhu AX (2007) Development of sorafenib and other molecularly targeted agents in hepatocellular carcinoma. Cancer 112:250–259
Bendall LJ, Nilsson SK, Khan NI, James A, Bonnet C, Lock RB et al (2004) Role of CD44 variant exon 6 in acute lymphoblastic leukemia: association with altered bone marrow localisation and increased tumour burden. Leukemia 18:1308–1311
Ponta H, Sherman L, Herrlich PA (2003) CD44: from adhesion molecules to signalling regulators. Nat Rev Mol Cell Biol 4:33–45
Cooper DL, Dougherty GJ (1995) To metastasize or not? Selection of CD44 splice sites. Nat Med 1:635–637
Liu J, Jiang G (2006) CD44 and hematologic malignancies. Cell Mol Immunol 3:359–365
Naor D, Nedvetzki S, Golan I, Melnik L, Faitelson Y (2002) CD44 in cancer. Crit Rev Clin Lab Sci 39:527–579
Jothy S (2003) CD44 and its partners in metastasis. Clin Exp Metastasis 20:195–201
Seiter S, Schadendorf D, Herrmann K, Schneider M, Rosel M, Arch R et al (1996) Expression of CD44 variant isoforms in malignant melanoma. Clin Cancer Res 2:447–456
Akisik E, Bavbek S, Dalay N (2002) CD44 variant exons in leukemia and lymphoma. Pathol Oncol Res 8:36–40
Endo K, Terada T (2000) Protein expression of CD44 (standard and variant isoforms) in hepatocellular carcinoma: relationships with tumor grade, clinicopathologic parameters, p53 expression, and patient survival. J Hepatol 32:78–84
Jin L, Hope KJ, Zhai Q, Smadja-Joffe F, Dick JE (2006) Targeting of CD44 eradicates human acute myeloid leukemic stem cells. Nat Med 12:1167–1174
Song G, Liao X, Zhou L, Wu L, Feng Y, Han ZC (2004) HI44a, an anti-CD44 monoclonal antibody, induces differentiation and apoptosis of human acute myeloid leukemia cells. Leuk Res 28:1089–1096
Gadhoum Z, Delaunay J, Maquarre E, Durand L, Lancereaux V, Qi J et al (2004) The effect of anti-CD44 monoclonal antibodies on differentiation and proliferation of human acute myeloid leukemia cells. Leuk Lymphoma 45:1501–1510
Charrad RS, Gadhoum Z, Qi J, Glachant A, Allouche M, Jasmin C et al (2002) Effects of anti-CD44 monoclonal antibodies on differentiation and apoptosis of human myeloid leukemia cell lines. Blood 99:290–299
Tamm I, Dörken B, Hartmann G (2001) Antisense therapy in oncology: new hope for an old idea? Lancet 358:489–497
Spitaler M, Wiesenhofer B, Biedermann V, Seppi T, Zimmermann J, Grunicke H et al (1999) The involvement of protein kinase C isoenzymes alpha, epsilon and zeta in the sensitivity to antitumor treatment and apoptosis induction. Anticancer Res 19:3969–3976
Beeram M, Patnaik A, Rowinsky EK (2005) Raf: a strategic target for therapeutic development against cancer. J Clin Oncol 23:6771–6790
Bartholomeusz C, Itamochi H, Yuan LX, Esteva FJ, Wood CG, Terakawa N et al (2005) Bcl-2 antisense oligonucleotide overcomes resistance to E1A gene therapy in a low HER2-expressing ovarian cancer xenograft model. Cancer Res 65:8406–8413
Lacy J, Loomis R, Grill S, Srimatkandada P, Carbone R, Cheng YC (2006) Systemic Bcl-2 antisense oligodeoxynucleotide in combination with cisplatin cures EBV+ nasopharyngeal carcinoma xenografts in SCID mice. Int J Cancer 119:309–316
De Cesare M, Perego P, Righetti SC, Pratesi G, Carenini N, Rivoltini L et al (2005) Enhanced antitumour efficacy of gimatecan in combination with Bcl-2 antisense oligonucleotide in human melanoma xenografts. Eur J Cancer 41:1213–1222
July LV, Beraldi E, So A, Fazli L, Evans K, English JC, Gleave ME (2004) Nucleotide-based therapies targeting clusterin chemosensitize human lung adenocarcinoma cells both in vitro and in vivo. Mol Cancer Ther 3:223–232
Kim R, Emi M, Tanabe K, Toge T (2004) Therapeutic potential of antisense Bcl-2 as a chemosensitizer for cancer therapy. Cancer 101:2491–2502
O’Connor OA, Smith EA, Toner LE, Teruya-Feldstein J, Frankel S, Rolfe M et al (2006) The combination of the proteasome inhibitor bortezomib and the bcl-2 antisense molecule oblimersen sensitizes human B-cell lymphomas to cyclophosphamide. Clin Cancer Res 12:2902–2911
Bullard KM, Kim HR, Wheeler MA, Wilson CM, Neudauer CL, Simpson MA et al (2003) Hyaluronan synthase-3 is upregulated in metastatic colon carcinoma cells and manipulation of expression alters matrix retention and cellular growth. Int J Cancer 107:739–746
Dartsch DC, Schaefer A, Boldt S, Kolch W, Marquardt H (2002) Comparison of anthracycline-induced death of human leukemia cells: programmed cell death versus necrosis. Apoptosis 7:537–548
Koceva-Chyla A, Jedrzejczak M, Skierski J, Kania K, Jozwiak Z (2005) Mechanisms of induction of apoptosis by anthraquinone anticancer drugs aclarubicin and mitoxantrone in comparison with doxorubicin: relation to drug cytotoxicity and caspase-3 activation. Apoptosis 10:1497–1514
Gupta GP, Massague J (2006) Cancer metastasis: building a framework. Cell 127:679–695
Radotra B, McCormick D (1997) Glioma invasion in vitro is mediated by CD44-hyaluronan interactions. J Pathol 181:434–438
Knutson JR, Iida J, Fields GB, McCarthy JB (1996) CD44/chondroitin sulfate proteoglycan and alpha 2 beta 1 integrin mediate human melanoma cell migration on type IV collagen and invasion of basement membranes. Mol Biol Cell 7:383–396
Ambudkar SV, Dey S, Hrycyna CA, Ramachandra M, Pastan I, Gottesman MM (1999) Biochemical, cellular, and pharmacological aspects of the multidrug transporter. Annu Rev Pharmacol Toxicol 39:361–398
Toole BP (2004) Hyaluronan: from extracellular glue to pericellular cue. Nat Rev Cancer 4:528–539
Miyake H, Hara I, Kamidono S, Gleave ME (2001) Synergistic chemsensitization and inhibition of tumor growth and metastasis by the antisense oligodeoxynucleotide targeting clusterin gene in a human bladder cancer model. Clin Cancer Res 7:4245–4252
Yang X, Zheng F, Xing H, Gao Q, Wei W, Lu Y et al (2004) Resistance to chemotherapy-induced apoptosis via decreased caspase-3 activity and overexpression of antiapoptotic proteins in ovarian cancer. J Cancer Res Clin Oncol 130:423–428
Mita MM, Ochoa L, Rowinsky EK, Kuhn J, Schwartz G, Hammond LA et al (2006) A phase I, pharmacokinetic and biologic correlative study of oblimersen sodium (Genasense, G3139) and irinotecan in patients with metastatic colorectal cancer. Ann Oncol 17:313–321
Bourguignon LY, Gilad E, Brightman A, Diedrich F, Singleton P (2006) Hyaluronan–CD44 interaction with leukemia-associated RhoGEF and epidermal growth factor receptor promotes Rho/Ras co-activation, phospholipase C epsilon-Ca2+ signaling, and cytoskeleton modification in head and neck squamous cell carcinoma cells. J Biol Chem 281:14026–14040
Bourguignon LY, Gilad E, Rothman K, Peyrollier K (2005) Hyaluronan–CD44 interaction with IQGAP1 promotes Cdc42 and ERK signaling, leading to actin binding, Elk-1/estrogen receptor transcriptional activation, and ovarian cancer progression. J Biol Chem 280:11961–11972
Miletti-Gonzalez KE, Chen S, Muthukumaran N, Saglimbeni GN, Wu X, Yang J, Apolito K et al (2005) The CD44 receptor interacts with P-glycoprotein to promote cell migration and invasion in cancer. Cancer Res 65:6660–6667
Acknowledgments
This study was supported by Singapore Cancer Syndicate Grant—TN0031, AN0038 and Terry Fox Run Cancer Research Grant 2004 (Chien-Shing Chen).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xie, Z., Choong, P.F., Poon, L.F. et al. Inhibition of CD44 expression in hepatocellular carcinoma cells enhances apoptosis, chemosensitivity, and reduces tumorigenesis and invasion. Cancer Chemother Pharmacol 62, 949–957 (2008). https://doi.org/10.1007/s00280-008-0684-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-008-0684-z