Abstract
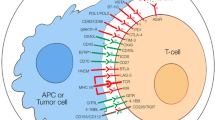
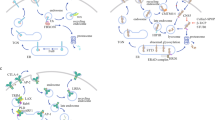
Avoiding immune surveillance and inducing a tumor-promoting inflammatory milieu found entry into the new generation of the hallmarks of cancer. Cancer cells hijack immune mechanisms which physiologically protect the body from the development of autoimmune diseases and excessive tissue damage during inflammation by downregulating immune responses. This is frequently achieved by upregulation of immune checkpoints. Therefore, the blocking of immune checkpoint ligand–receptor interactions can reinstall the immune systems capability to fight cancer cells as shown for CTLA4 and PD-1 inhibitors in a clinical setting. Newly described checkpoint antigens are currently under investigation in cancer immunotherapy. Preclinical data emphasize the immune checkpoint axis TIGIT–PVR/PVRL2 as very promising target. This axis includes additional receptors such as DNAM-1, CD96, and CD112R. In this review, we discuss the recent findings of the relevance of this complex receptor ligand system in hematologic and solid cancers. Emphasis is also laid on the discussion of potential combinations with other immunotherapeutic approaches.
Similar content being viewed by others
References
Ansell SM, Lesokhin AM, Borrello I et al (2015) PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma. N Engl J Med 372:311–319. https://doi.org/10.1056/NEJMoa1411087
Ardolino M, Zingoni A, Cerboni C et al (2011) DNAM-1 ligand expression on Ag-stimulated T lymphocytes is mediated by ROS-dependent activation of DNA-damage response: relevance for NK-T cell interaction. Blood 117:4778–4786. https://doi.org/10.1182/blood-2010-08-300954
Bevelacqua V, Bevelacqua Y, Candido S et al (2012) Nectin like-5 overexpression correlates with the malignant phenotype in cutaneous melanoma. Oncotarget 3:882–892. https://doi.org/10.18632/oncotarget.594
Blake SJ, Stannard K, Liu J et al (2016) Suppression of metastases using a new lymphocyte checkpoint target for cancer immunotherapy. Cancer Discov 6:446–459. https://doi.org/10.1158/2159-8290.CD-15-0944
Boles KS, Vermi W, Facchetti F et al (2009) A novel molecular interaction for the adhesion of follicular CD4 T cells to follicular DC. Eur J Immunol 39:695–703. https://doi.org/10.1002/eji.200839116
Bottino C, Castriconi R, Pende D et al (2003) Identification of PVR (CD155) and nectin-2 (CD112) as cell surface ligands for the human DNAM-1 (CD226) activating molecule. J Exp Med 198:557–567. https://doi.org/10.1084/jem.20030788
Burnet FM (1967) Immunological aspects of malignant disease. Lancet 289:1171–1174. https://doi.org/10.1016/S0140-6736(67)92837-1
Burns GF, Triglia T, Werkmeister JA et al (1985) TLiSA1, a human T lineage-specific activation antigen involved in the differentiation of cytotoxic T lymphocytes and anomalous killer cells from their precursors. J Exp Med 161:1063–1078
Carlsten M, Björkström NK, Norell H et al (2007) DNAX accessory molecule-1 mediated recognition of freshly isolated ovarian carcinoma by resting natural killer cells. Cancer Res 67:1317–1325. https://doi.org/10.1158/0008-5472.CAN-06-2264
Carlsten M, Norell H, Bryceson YT et al (2009) Primary human tumor cells expressing CD155 impair tumor targeting by down-regulating DNAM-1 on NK cells. J Immunol 183:4921–4930. https://doi.org/10.4049/jimmunol.0901226
Castriconi R, Dondero A, Corrias MV et al (2004) Natural killer cell-mediated killing of freshly isolated neuroblastoma cells: critical role of DNAX accessory molecule-1–poliovirus receptor interaction. Cancer Res 64:9180–9184. https://doi.org/10.1158/0008-5472.CAN-04-2682
Cerboni C, Fionda C, Soriani A et al (2014) The DNA damage response: a common pathway in the regulation of NKG2D and DNAM-1 ligand expression in normal, infected, and cancer cells. Front Immunol 4:1–7. https://doi.org/10.3389/fimmu.2013.00508
Chan CJ, Andrews DM, McLaughlin NM et al (2010) DNAM-1/CD155 interactions promote cytokine and NK cell-mediated suppression of poorly immunogenic melanoma metastases. J Immunol 184:902–911. https://doi.org/10.4049/jimmunol.0903225
Chan CJ, Martinet L, Gilfillan S et al (2014) The receptors CD96 and CD226 oppose each other in the regulation of natural killer cell functions. Nat Immunol 15:431–438. https://doi.org/10.1038/ni.2850
Chauvin J, Pagliano O, Fourcade J et al (2015) TIGIT and PD-1 impair tumor antigen-specific CD8+ T cells in melanoma patients. J Clin Invest 125:2046–2058. https://doi.org/10.1172/JCI80445
Cocchi F, Menotti L, Dubreuil P et al (2000) Cell-to-cell spread of wild-type herpes simplex virus type 1, but not of syncytial strains, is mediated by the immunoglobulin-like receptors that mediate virion entry, nectin1 (PRR1/HveC/HIgR) and nectin2 (PRR2/HveB). J Virol 74:3909–3917
Coley WB (1891) II. Contribution to the knowledge of sarcoma. Ann Surg 14:199–220
Cortez VS, Ulland TK, Cervantes-Barragan L et al (2017) SMAD4 impedes the conversion of NK cells into ILC1-like cells by curtailing non-canonical TGF-β signaling. Nat Immunol 18:995–1003. https://doi.org/10.1038/ni.3809
Dixon KO, Schorer M, Nevin J et al (2018) Functional anti-TIGIT antibodies regulate development of autoimmunity and antitumor immunity. J Immunol. https://doi.org/10.4049/jimmunol.1700407
Ehrlich P (1899) Croonian lecture: on immunity with special reference to cell life. Proc R Soc London 66:424–448
El-Sherbiny YM, Meade JL, Holmes TD et al (2007) The requirement for DNAM-1, NKG2D, and NKp46 in the natural killer cell-mediated killing of myeloma cells. Cancer Res 67:8444–8449. https://doi.org/10.1158/0008-5472.CAN-06-4230
Fabre S, Reymond N, Cocchi F et al (2002) Prominent role of the Ig-like V domain in trans-interactions of nectins. Nectin3 and nectin 4 bind to the predicted C-C′-C″-D beta-strands of the nectin1 V domain. J Biol Chem 277:27006–27013. https://doi.org/10.1074/jbc.M203228200
Feucht J, Kayser S, Gorodezki D et al (2016) T-cell responses against CD19+ pediatric acute lymphoblastic leukemia mediated by bispecific T-cell engager (BiTE) are regulated contrarily by PD-L1 and CD80/CD86 on leukemic blasts. Oncotarget. https://doi.org/10.18632/oncotarget.12357
Fionda C, Abruzzese M, Zingoni A et al (2015) Nitric oxide donors increase PVR/CD155 DNAM-1 ligand expression in multiple myeloma cells: role of DNA damage response activation. BMC Cancer 15:17. https://doi.org/10.1186/s12885-015-1023-5
Fuchs A, Cella M, Giurisato E et al (2004) Cutting edge: CD96 (tactile) promotes NK cell-target cell adhesion by interacting with the poliovirus receptor (CD155). J Immunol 172:3994–3998. https://doi.org/10.4049/jimmunol.172.7.3994
Fuhrman C, Yeh W-I, Seay HR et al (2015) Divergent phenotypes of human regulatory T cells expressing the receptors TIGIT and CD226. J Immunol 195:145–155. https://doi.org/10.4049/jimmunol.1402381
Gao Y, Souza-Fonseca-Guimaraes F, Bald T et al (2017) Tumor immunoevasion by the conversion of effector NK cells into type 1 innate lymphoid cells. Nat Immunol 18:1004–1015. https://doi.org/10.1038/ni.3800
Gerhardt T, Ley K (2015) Monocyte trafficking across the vessel wall. Cardiovasc Res 107:321–330. https://doi.org/10.1093/cvr/cvv147
Gopalakrishnan V, Spencer CN, Nezi L et al (2018) Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 359:97–103. https://doi.org/10.1126/science.aan4236
Guevarra LA, Afable ACF, Belza PJO et al (2018) Immunogenicity of a Fap2 peptide mimotope of Fusobacterium nucleatum and its potential use in the diagnosis of colorectal cancer. Infect Agent Cancer 13:1–6. https://doi.org/10.1186/s13027-018-0184-7
Gur C, Ibrahim Y, Isaacson B et al (2015) Binding of the Fap2 protein of Fusobacterium nucleatum to human inhibitory receptor TIGIT protects tumors from immune cell attack. Immunity 42:344–355. https://doi.org/10.1016/j.immuni.2015.01.010
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. https://doi.org/10.1016/j.cell.2011.02.013
He W, Zhang H, Han F et al (2017) CD155T/TIGIT signaling regulates CD8+ T-cell metabolism and promotes tumor progression in human gastric cancer. Cancer Res 77:6375–6388. https://doi.org/10.1158/0008-5472.CAN-17-0381
Hirota T, Irie K, Okamoto R et al (2005) Transcriptional activation of the mouse Necl-5/Tage4/PVR/CD155 gene by fibroblast growth factor or oncogenic Ras through the Raf-MEK-ERK-AP-1 pathway. Oncogene 24:2229–2235. https://doi.org/10.1038/sj.onc.1208409
Hodi FS, O’Day SJ, McDermott DF et al (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363:711–723. https://doi.org/10.1056/NEJMoa1003466
Hosen N, Park CY, Tatsumi N et al (2007) CD96 is a leukemic stem cell-specific marker in human acute myeloid leukemia. Proc Natl Acad Sci USA 104:11008–11013. https://doi.org/10.1073/pnas.0704271104
Hou S, Zheng X, Wei H et al (2014) Recombinant soluble CD226 protein directly inhibits cancer cell proliferation in vitro. Int Immunopharmacol 19:119–126. https://doi.org/10.1016/j.intimp.2014.01.012
Hutten TJA, Norde WJ, Woestenenk R et al (2017) Increased coexpression of PD-1, TIGIT, and KLRG-1 on tumor-reactive CD8+ T cells during relapse after allogeneic stem cell transplantation. Biol Blood Marrow Transplant. https://doi.org/10.1016/j.bbmt.2017.11.027
Iguchi-Manaka A, Okumura G, Kojima H et al (2016) Increased soluble CD155 in the serum of cancer patients. PLoS ONE 11:1–12. https://doi.org/10.1371/journal.pone.0152982
Inozume T, Yaguchi T, Furuta J et al (2016) Melanoma cells control antimelanoma CTL responses via interaction between TIGIT and CD155 in the effector phase. J Invest Dermatol 136:255–263. https://doi.org/10.1038/JID.2015.404
Johnston RJ, Comps-Agrar L, Hackney J et al (2014) The immunoreceptor TIGIT regulates antitumor and antiviral CD8(+) T cell effector function. Cancer Cell 26:923–937. https://doi.org/10.1016/j.ccell.2014.10.018
Joller N, Hafler JP, Brynedal B et al (2011) Cutting edge: TIGIT has T cell-intrinsic inhibitory functions. J Immunol 186:1338–1342. https://doi.org/10.4049/jimmunol.1003081
Joller N, Lozano E, Burkett PR et al (2014) Treg cells expressing the coinhibitory molecule TIGIT selectively inhibit proinflammatory Th1 and Th17 cell responses. Immunity 40:569–581. https://doi.org/10.1016/j.immuni.2014.02.012
Kamran N, Takai Y, Miyoshi J et al (2013) Toll-like receptor ligands induce expression of the costimulatory molecule CD155 on antigen-presenting cells. PLoS ONE 8:e54406. https://doi.org/10.1371/journal.pone.0054406
Karabulut M, Gunaldi M, Alis H et al (2016) Serum nectin-2 levels are diagnostic and prognostic in patients with colorectal carcinoma. Clin Transl Oncol 18:160–171. https://doi.org/10.1007/s12094-015-1348-1
Kellner C, Peipp M, Schub N et al (2013) Targeting CD96 for antibody based elimination of leukemic stem cells in AML: a new strategy in stem cell transplantation. Blood 122:3972 LP–L3972
Köhnke T, Krupka C, Tischer J et al (2015) Increase of PD-L1 expressing B-precursor ALL cells in a patient resistant to the CD19/CD3-bispecific T cell engager antibody blinatumomab. J Hematol Oncol 8:111. https://doi.org/10.1186/s13045-015-0213-6
Kong Y, Zhu L, Schell TD et al (2016) T-cell immunoglobulin and ITIM domain (TIGIT) associates with CD8+ T-cell exhaustion and poor clinical outcome in AML patients. Clin Cancer Res 22:3057–3066. https://doi.org/10.1158/1078-0432.CCR-15-2626
Krupka C, Kufer P, Kischel R et al (2015) Blockade of the PD-1/PD-L1 axis augments lysis of AML cells by the CD33/CD3 BiTE antibody construct AMG 330: reversing a T-cell-induced immune escape mechanism. Leukemia. https://doi.org/10.1038/leu.2015.214
Kuball J, de Boer K, Wagner E et al (2011) Pitfalls of vaccinations with WT1-, proteinase3- and MUC1-derived peptides in combination with MontanideISA51 and CpG7909. Cancer Immunol Immunother 60:161–171. https://doi.org/10.1007/s00262-010-0929-7
Kurtulus S, Sakuishi K, Ngiow S et al (2015) TIGIT predominantly regulates the immune response via regulatory T cells. J Clin Invest 125:4053–4062. https://doi.org/10.1172/JCI81187
Levin SD, Taft DW, Brandt CS et al (2011) Vstm3 is a member of the CD28 family and an important modulator of T-cell function. Eur J Immunol 41:902–915. https://doi.org/10.1002/eji.201041136
Li M, Xia P, Du Y et al (2014) T-cell immunoglobulin and ITIM domain (TIGIT) receptor/poliovirus receptor (PVR) ligand engagement suppresses interferon-γ production of natural killer cells via β-arrestin 2-mediated negative signaling. J Biol Chem 289:17647–17657. https://doi.org/10.1074/jbc.M114.572420
Liu S, Zhang H, Li M et al (2013) Recruitment of Grb2 and SHIP1 by the ITT-like motif of TIGIT suppresses granule polarization and cytotoxicity of NK cells. Cell Death Differ 20:456–464. https://doi.org/10.1038/cdd.2012.141
Lopez M, Aoubala M, Jordier F et al (1998) The human poliovirus receptor related 2 protein is a new hematopoietic/endothelial homophilic adhesion molecule. Blood 92:4602–4611
Lozano E, Dominguez-Villar M, Kuchroo V, Hafler D (2012) The TIGIT/CD226 axis regulates human T cell function. J Immunol 188:3869–3875. https://doi.org/10.4049/jimmunol.1103627
Mahoney KM, Rennert PD, Freeman GJ (2015) Combination cancer immunotherapy and new immunomodulatory targets. Nat Rev Drug Discov 14:561–584. https://doi.org/10.1038/nrd4591
Mamessier E, Sylvain A, Thibult M-L et al (2011) Human breast cancer cells enhance self tolerance by promoting evasion from NK cell antitumor immunity. J Clin Invest 121:3609–3622. https://doi.org/10.1172/JCI45816
Mariathasan S, Turley SJ, Nickles D et al (2018) TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 554:544–548. https://doi.org/10.1038/nature25501
Masson D, Jarry A, Baury B et al (2001) Overexpression of the CD155 gene in human colorectal carcinoma. Gut 49:236–240. https://doi.org/10.1136/gut.49.2.236
Matson V, Fessler J, Bao R et al (2018) The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science 359:104–108. https://doi.org/10.1126/science.aao3290
Melero I, Berman DM, Aznar MA et al (2015) Evolving synergistic combinations of targeted immunotherapies to combat cancer. Nat Rev Cancer 15:457–472. https://doi.org/10.1038/nrc3973
Nakai R, Maniwa Y, Tanaka Y et al (2010) Overexpression of Necl-5 correlates with unfavorable prognosis in patients with lung adenocarcinoma. Cancer Sci 101:1326–1330. https://doi.org/10.1111/j.1349-7006.2010.01530.x
Nishiwada S, Sho M, Yasuda S et al (2015) Clinical significance of CD155 expression in human pancreatic cancer. Anticancer Res 35:2287–2297
Nobis P, Zibirre R, Meyer G et al (1985) Production of a monoclonal antibody against an epitope on HeLa cells that is the functional poliovirus binding site. J Gen Virol 66:2563–2569. https://doi.org/10.1099/0022-1317-66-12-2563
Oshima T, Sato S, Kato J et al (2013) Nectin-2 is a potential target for antibody therapy of breast and ovarian cancers. Mol Cancer 12:60. https://doi.org/10.1186/1476-4598-12-60
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264. https://doi.org/10.1038/nrc3239
Pauken KE, Wherry EJ (2014) TIGIT and CD226: tipping the balance between costimulatory and coinhibitory molecules to augment the cancer immunotherapy toolkit. Cancer Cell 26:785–787. https://doi.org/10.1016/j.ccell.2014.11.016
Pende D, Bottino C, Castriconi R et al (2005a) PVR (CD155) and nectin-2 (CD112) as ligands of the human DNAM-1 (CD226) activating receptor: involvement in tumor cell lysis. Mol Immunol 42:463–469. https://doi.org/10.1016/j.molimm.2004.07.028
Pende D, Spaggiari GM, Marcenaro S et al (2005b) Analysis of the receptor-ligand interactions in the natural killer—mediated lysis of freshly isolated myeloid or lymphoblastic leukemias: evidence for the involvement of the poliovirus receptor (CD155) and nectin-2 (CD112). Blood 105:2066–2073. https://doi.org/10.1182/blood-2004-09-3548
Pende D, Castriconi R, Romagnani P et al (2006) Expression of the DNAM-1 ligands, nectin-2 (CD112) and poliovirus receptor (CD155), on dendritic cells: relevance for natural killer-dendritic cell interaction. Blood 107:2030–2036. https://doi.org/10.1182/blood-2005-07-2696
Peng Y, Xi C, Zhu Y et al (2016) Altered expression of CD226 and CD96 on natural killer cells in patients with pancreatic cancer. Oncotarget. https://doi.org/10.18632/oncotarget.11953
Plimack ER, Bellmunt J, Gupta S et al (2017) Safety and activity of pembrolizumab in patients with locally advanced or metastatic urothelial cancer (KEYNOTE-012): a non-randomised, open-label, phase 1b study. Lancet Oncol 18:212–220. https://doi.org/10.1016/S1470-2045(17)30007-4
Routy B, Le Chatelier E, Derosa L et al (2018) Gut microbiome influences efficacy of PD-1–based immunotherapy against epithelial tumors. Science 359:91–97. https://doi.org/10.1126/science.aan3706
Sakisaka T, Takai Y (2004) Biology and pathology of nectins and nectin-like molecules. Curr Opin Cell Biol 16:513–521. https://doi.org/10.1016/j.ceb.2004.07.007
Samanta D, Ramagopal UA, Rubinstein R et al (2012) Structure of nectin-2 reveals determinants of homophilic and heterophilic interactions that control cell-cell adhesion. Proc Natl Acad Sci USA 109:14836–14840. https://doi.org/10.1073/pnas.1212912109
Samanta D, Guo H, Rubinstein R et al (2017) Structural, mutational and biophysical studies reveal a canonical mode of molecular recognition between immune receptor TIGIT and nectin-2. Mol Immunol 81:151–159. https://doi.org/10.1016/j.molimm.2016.12.003
Sanchez-Correa B, Gayoso I, Bergua JM et al (2012) Decreased expression of DNAM-1 on NK cells from acute myeloid leukemia patients. Immunol Cell Biol 90:109–115. https://doi.org/10.1038/icb.2011.15
Schadendorf D, Hodi FS, Robert C et al (2015) Pooled analysis of long-term survival data from phase II and phase III trials of ipilimumab in unresectable or metastatic melanoma. J Clin Oncol 33:1889–1894. https://doi.org/10.1200/JCO.2014.56.2736
Sharma P, Callahan MK, Bono P et al (2016) Nivolumab monotherapy in recurrent metastatic urothelial carcinoma (CheckMate 032): a multicentre, open-label, two-stage, multi-arm, phase 1/2 trial. Lancet Oncol 17:1590–1598. https://doi.org/10.1016/S1470-2045(16)30496-X
Shibuya A, Campbell D, Hannum C et al (1996) DNAM-1, a novel adhesion molecule involved in the cytolytic function of T lymphocytes. Immunity 4:573–581. https://doi.org/10.1016/S1074-7613(00)70060-4
Sivan A, Corrales L, Hubert N et al (2015) Commensal bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science 350:1084–1089. https://doi.org/10.1126/science.aac4255
Solecki DJ, Gromeier M, Mueller S et al (2002) Expression of the human poliovirus receptor/CD155 gene is activated by sonic hedgehog. J Biol Chem 277:25697–25702. https://doi.org/10.1074/jbc.M201378200
Soriani A, Zingoni A, Cerboni C et al (2009) ATM-ATR-dependent up-regulation of DNAM-1 and NKG2D ligands on multiple myeloma cells by therapeutic agents results in enhanced NK-cell susceptibility and is associated with a senescent phenotype. Blood 113:3503–3511. https://doi.org/10.1182/blood-2008-08-173914
Stamm H, Klingler F, Grossjohann E-M et al (2018) Immune checkpoints PVR and PVRL2 are prognostic markers in AML and their blockade represents a new therapeutic option. Oncogene. https://doi.org/10.1038/s41388-018-0288-y
Stanietsky N, Simic H, Arapovic J et al (2009) The interaction of TIGIT with PVR and PVRL2 inhibits human NK cell cytotoxicity. Proc Natl Acad Sci 106:17858–17863. https://doi.org/10.1073/pnas.0903474106
Stanietsky N, Rovis TL, Glasner A et al (2013) Mouse TIGIT inhibits NK-cell cytotoxicity upon interaction with PVR. Eur J Immunol 43:2138–2150. https://doi.org/10.1002/eji.201243072
Stengel KF, Harden-Bowles K, Yu X et al (2012) Structure of TIGIT immunoreceptor bound to poliovirus receptor reveals a cell-cell adhesion and signaling mechanism that requires cis-trans receptor clustering. Proc Natl Acad Sci 109:5399–5404. https://doi.org/10.1073/pnas.1120606109
Tahara-Hanaoka S, Shibuya K, Onoda Y et al (2004) Functional characterization of DNAM-1 (CD226) interaction with its ligands PVR (CD155) and nectin-2 (PRR-2/CD112). Int Immunol 16:533–538
Tahara-Hanaoka S, Shibuya K, Kai H et al (2006) Tumor rejection by the poliovirus receptor family ligands of the DNAM-1 (CD226) receptor. Blood 107:1491–1496. https://doi.org/10.1182/blood-2005-04-1684
Takahashi N, Sugaya M, Suga H et al (2017) Increased soluble CD226 in sera of patients with cutaneous T cell lymphoma mediating cytotoxic activity against tumor cells via CD155. J Invest Dermatol. https://doi.org/10.1016/j.jid.2017.03.025
Tauriainen J, Scharf L, Frederiksen J et al (2017) Perturbed CD8+ T cell TIGIT/CD226/PVR axis despite early initiation of antiretroviral treatment in HIV infected individuals. Sci Rep 7:40354. https://doi.org/10.1038/srep40354
Tauriello DVF, Palomo-ponce S, Iglesias M et al (2018) TGF-beta drives immune evasion in genetically reconstituted colon cancer metastasis. Nat Publ Gr 554:538–543. https://doi.org/10.1038/nature25492
Topalian SL, Hodi FS, Brahmer JR et al (2012) Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 366:2443–2454. https://doi.org/10.1056/NEJMoa1200690
Uttenthal B, Martinez-Davila I, Ivey A et al (2014) Wilms’ Tumour 1 (WT1) peptide vaccination in patients with acute myeloid leukaemia induces short-lived WT1-specific immune responses. Br J Haematol 164:366–375. https://doi.org/10.1111/bjh.12637
Wang PL, O’Farrell S, Clayberger C, Krensky AM (1992) Identification and molecular cloning of tactile. A novel human T cell activation antigen that is a member of the Ig gene superfamily. J Immunol 148:2600–2608
Wang H, Zhang Z, Fan M et al (2010) Expression of CD112 in colon carcinoma tissues and cell lines and their clinical significance. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 26:477–479
Wang F, Hou H, Wu S et al (2015) TIGIT expression levels on human NK cells correlate with functional heterogeneity among healthy individuals. Eur J Immunol 45:2886–2897. https://doi.org/10.1002/eji.201545480
Wellbrock J, Latuske E, Kohler J et al (2015) Expression of hedgehog pathway mediator GLI represents a negative prognostic marker in human acute myeloid leukemia and its inhibition exerts antileukemic effects. Clin Cancer Res 21:2388–2398. https://doi.org/10.1158/1078-0432.CCR-14-1059
Xu F, Sunderland A, Zhou Y et al (2017) Blockade of CD112R and TIGIT signaling sensitizes human natural killer cell functions. Cancer Immunol Immunother. https://doi.org/10.1007/s00262-017-2031-x
Yi M, Yu S, Qin S et al (2018) Gut microbiome modulates efficacy of immune checkpoint inhibitors. J Hematol Oncol 11:47. https://doi.org/10.1186/s13045-018-0592-6
Yu X, Harden K, Gonzalez C L, et al (2009) The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat Immunol 10:48–57. https://doi.org/10.1038/ni.1674
Zhang B, Zhao W, Li H et al (2016) Immunoreceptor TIGIT inhibits the cytotoxicity of human cytokine-induced killer cells by interacting with CD155. Cancer Immunol Immunother 65:305–314. https://doi.org/10.1007/s00262-016-1799-4
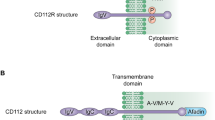
Zhu Y, Paniccia A, Schulick AC et al (2016) Identification of CD112R as a novel checkpoint for human T cells. J Exp Med 213:167–176. https://doi.org/10.1084/jem.20150785
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
H.S., J.W., and W.F.: inventorship on patents held by Amgen Research (Munich) GmbH/Amgen Inc. WF: advisory and consulting role for Amgen; advisory role for ARIAD/Incyte, Novartis, Pfizer; royalties from Amgen; support for meeting attendance from Amgen, Gilead, GSO, Teva, JAZZ Pharmaceuticals, Daiichi Sankyo; research funding from Amgen, Pfizer.
Rights and permissions
About this article
Cite this article
Stamm, H., Wellbrock, J. & Fiedler, W. Interaction of PVR/PVRL2 with TIGIT/DNAM-1 as a novel immune checkpoint axis and therapeutic target in cancer. Mamm Genome 29, 694–702 (2018). https://doi.org/10.1007/s00335-018-9770-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-018-9770-7