Abstract
Purpose
Postnatal surge of gonadotrophins, Luteinizing hormone (LH) and Follicle-Stimulating hormone (FSH) known as minipuberty, is critical for gonocyte maturation into spermatogonial stem cells (SSC) in the testis. Gonadotrophins are essential for optimum fertility in men, but very little is known how they regulate germ cells during minipuberty. This study examined whether gonadotrophins play a role on gonocyte transformation in vivo.
Methods
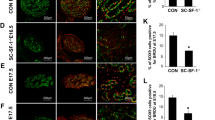
Testes from hypogonadal (hpg) mice and their wild type (WT) littermates (n = 6/group) were weighed, and processed in paraffin at postnatal days (D) 0, 3, 6 and 9. Mouse VASA homologue (germ cell marker), anti-Müllerian hormone (Sertoli cell marker) antibodies and DAPI (nuclei marker) were used for immunofluorescence followed by confocal imaging. Germ cells on or off basement membrane (BM) and Sertoli cells/tubule were counted using Image J and analyzed with GraphPad.
Results
Comparing to WT littermates, there were significantly fewer germ cells on BM/tubule (p < 0.05) in D9 hpg mice, whereas there was no significant difference for germ cells off BM/tubule and Sertoli cells/tubule between littermates. However, testicular weight was significantly reduced in D3-D9 hpg mice comparing to WT littermates.
Conclusion
Gonadotrophin deficiency reduced D9 germ cells on BM indicating impaired gonocyte transformation into SSC. This suggests that gonadotrophins may mediate gonocyte transformation during minipuberty.



Similar content being viewed by others
References
Hutson JM, Southwell BR, Li R, Lie G, Ismail K, Harisis G, Chen N (2013) The regulation of testicular descent and the effects of cryptorchidism. Endocr Rev 34(5):725–752
Hadziselimovic F, Huff D, Duckett J, Herzog B, Elder J, Snyder H, Buser M (1987) Treatment of cryptorchidism with low doses of buserelin over a 6-months period. Eur J Pediatr 146(2):S56–S58
Li R, Vannitamby A, Yue SSK, Handelsman D, Hutson J (2017) Mouse minipuberty coincides with gonocyte transformation into spermatogonial stem cells: a model for human minipuberty. Reprod Fertil Dev 29(12):2430–2436
Hadziselimovic F, Zivkovic D, Bica DT, Emmons LR (2005) The importance of mini-puberty for fertility in cryptorchidism. J Urol 174(4):1536–1539
Cortes D, Thorup J, Beck BL (1995) Quantitative histology of germ cells in the undescended testes of human fetuses, neonates and infants. J Urol 154(3):1188–1192
Li R, Vannitamby A, Meijer J, Southwell B, Hutson J (2015) Postnatal germ cell development during mini-puberty in the mouse does not require androgen receptor: implications for managing cryptorchidism. J Urol 193(4):1361–1367. https://doi.org/10.1016/j.juro.2014.10.024
Su S, Szarek M, Vooght A, Hutson J, Li R (2014) Gonocyte transformation to spermatogonial stem cells occurs earlier in patients with undervirilisation syndromes. J Pediatr Surg 49(2):323–327. https://doi.org/10.1016/j.jpedsurg.2013.11.047
Li R, Thorup J, Sun C, Cortes D, Southwell B, Hutson J (2014) Immunofluorescent analysis of testicular biopsies with germ cell and sertoli cell markers shows significant MVH negative germ cell depletion with older age at orchiopexy. J Urol 191(2):458–464
Loebenstein M, Thorup J, Cortes D, Clasen-Linde E, Hutson JM, Li R (2019) Cryptorchidism, gonocyte development, and the risks of germ cell malignancy and infertility: a systematic review. J Pediatr Surg. https://doi.org/10.1016/j.jpedsurg.2019.06.023
Singh J, Handelsman D (1996) Neonatal administration of FSH increases Sertoli cell numbers and spermatogenesis in gonadotropin-deficient (hpg) mice. J Endocrinol 151(1):37–48
O’shaughnessy P, Monteiro A, Verhoeven G, De Gendt K, Abel M (2010) Effect of FSH on testicular morphology and spermatogenesis in gonadotrophin-deficient hypogonadal mice lacking androgen receptors. Reproduction 139(1):177–184
Singh J, Handelsman DJ (1996) The effects of recombinant FSH on testosterone-induced spermatogenesis in gonadotrophin-deficient (hpg) mice. J Androl 17(4):382–393
Abel MH, Wootton AN, Wilkins V, Huhtaniemi I, Knight PG, Charlton HM (2000) The effect of a null mutation in the follicle-stimulating hormone receptor gene on mouse reproduction. Endocrinology 141(5):1795–1803
Kumar TR, Wang Y, Lu N, Matzuk MM (1997) Follicle stimulating hormone is required for ovarian follicle maturation but not male fertility. Nat Genet 15(2):201
Dierich A, Sairam MR, Monaco L, Fimia GM, Gansmuller A, LeMeur M, Sassone-Corsi P (1998) Impairing follicle-stimulating hormone (FSH) signaling in vivo: targeted disruption of the FSH receptor leads to aberrant gametogenesis and hormonal imbalance. Proc Natl Acad Sci 95(23):13612–13617
Singh J, O’Neill C, Handelsman DJ (1995) Induction of spermatogenesis by androgens in gonadotropin-deficient (hpg) mice. Endocrinology 136(12):5311–5321
Cattanach B, IDDON CA, Charlton H, CHIAPPA SA, Fink G (1977) Gonadotrophin-releasing hormone deficiency in a mutant mouse with hypogonadism. Nature 269(5626):338
Allan CM, Garcia A, Spaliviero J, Zhang F-P, Jimenez M, Huhtaniemi I, Handelsman DJ (2004) Complete Sertoli cell proliferation induced by follicle-stimulating hormone (FSH) independently of luteinizing hormone activity: evidence from genetic models of isolated FSH action. Endocrinology 145(4):1587–1593
Plant T (2008) Hypothalamic control of the pituitary-gonadal axis in higher primates: key advances over the last two decades. J Neuroendocrinol 20(6):719–726
Li R, Vannitamby A, Zhang JG, Fehmel EL, Southwell BR, Hutson JM (2015) Oct4-GFP expression during transformation of gonocytes into spermatogonial stem cells in the perinatal mouse testis. J Pediatr Surg 50(12):2084–2089. https://doi.org/10.1016/j.jpedsurg.2015.08.031
Li R, Azzollini D, Shen R, Thorup J, Clasen-Linde E, Cortes D, Hutson JM (2019) Postnatal germ cell development during first 18 months of life in testes from boys with non-syndromic cryptorchidism and complete or partial androgen insensitivity syndrome. J Pediatr Surg 54(8):1654–1659. https://doi.org/10.1016/j.jpedsurg.2018.12.011
Baker PJ, O’Shaughnessy PJ (2001) Role of gonadotrophins in regulating numbers of Leydig and Sertoli cells during fetal and postnatal development in mice. Reproduction 122(2):227–234
Myers M, Ebling FJP, Nwagwu M, Boulton R, Wadhwa K, Stewart J, Kerr JB (2005) Atypical development of Sertoli cells and impairment of spermatogenesis in the hypogonadal (hpg) mouse. J Anat 207(6):797–811. https://doi.org/10.1111/j.1469-7580.2005.00493.x
Heckert LL, Griswold MD (2002) The expression of the follicle-stimulating hormone receptor in spermatogenesis. Recent Prog Horm Res 57:129
Abel M, Baker P, Charlton H, Monteiro A, Verhoeven G, De Gendt K, Guillou F, O’Shaughnessy P (2008) Spermatogenesis and sertoli cell activity in mice lacking sertoli cell receptors for follicle-stimulating hormone and androgen. Endocrinology 149(7):3279–3285
Acknowledgements
We sincerely thank Prof. David Handelsman from Andrology Laboratory, ANZAC Research Institute, Concord Hospital, University of Sydney, Australia, for kind supply of the initial frozen sperm of hpg mice and other technical assistance to enable the establishment of this strain at our institute.
Funding
This study was funded by the Australian National Health and Medical Research Council (Grant no. APP1127109) and the Victorian Government’s Operational Infrastructure Support Program.
Author information
Authors and Affiliations
Contributions
EB performed experiments, analyzed data and wrote the first draft, HJC created the macro for cell counting, SAA and SW collected testes and prepared the paraffin blocks, GS and MYT provided assistance on data analysis and figure preparation. RL conceived the study and did the initial critical edition followed by JH’s critical revision. None of the authors has any potential conflict of interest to disclose.
Corresponding author
Ethics declarations
Conflict of interest
Author RL and JH have received the above mentioned research grant. Authors EB, SAA, GS, SW, MYT, JH, and RL declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Burton, E., Abeydeera, S.A., Sarila, G. et al. The role of gonadotrophins in gonocyte transformation during minipuberty. Pediatr Surg Int 36, 1379–1385 (2020). https://doi.org/10.1007/s00383-020-04737-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-020-04737-6