Abstract.
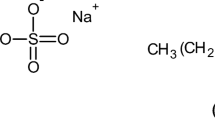
Complexes between lysozyme and sodium alkyl sulfates (decyl, dodecyl, tetradecyl and hexadecyl) in the solid state were prepared by mixing aqueous solutions of lysozyme and of the surfactant, separating the precipitated complex and purifying it. The stoichiometry of the complexes was investigated by elemental analysis and was found to correspond to about 8±1 alkyl sulfate ions per lysozyme. Low- and wide-angle X-ray scattering were used to investigate the structure of the complexes. The scattering curves showed one fairly large scattering maximum, revealing a low state of organization of the complexes. The characteristic length characterizing the complexes was calculated from the value of the wave vector corresponding to the maximum of the scattered intensity. This length increased by about 0.233 nm per additional methylene group in the surfactant alkyl chain. A model where spherical aggregates of alkyl sulfate ions are arranged in a disordered simple cubic structure, dispersed in a matrix of lysozyme, provides a possible explanation of the results.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Mathis, A., Zana, R. Study of complexes between lysozyme and sodium alkyl sulfates in the solid state. Colloid Polym Sci 280, 968–971 (2002). https://doi.org/10.1007/s00396-002-0721-0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00396-002-0721-0