Abstract
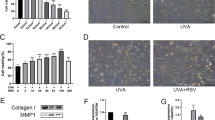
Lipid peroxidation caused by oxidative stress within the tissue leads to destruction and dysfunction of cellular membranes. Human dermal fibroblasts in the skin are subject to constant photooxidative stress caused mainly by deeply penetrating UVA irradiation. Therefore, the membrane damage caused by this photooxidative stress may be a major promoter of photoaging and photocarcinogenic processes initiated and promoted by long-term UVA exposure of the skin. The oxidative destruction is counterbalanced by a complex network of enzymatic and nonenzymatic antioxidants creating the skin’s line of defence against UVA-induced reactive oxygen species. The lazaroid tirilazad represents a new synthetic group of antioxidants with structural molecular similarity to glucocorticosteroids. We investigated the antioxidative capacity of tirilazad by determining its effects on the levels of malondialdehyde (MDA), as a marker of lipid peroxidation, induced directly or indirectly by UVA in human dermal fibroblasts. In a time- and dose-dependent kinetic, we demonstrated that fibroblasts incubated with tirilazad are well protected against subsequent UVA irradiation and show no increase in MDA levels similar to the unirradiated controls. This was also observed when lipid peroxidation was caused chemically by incubation of human dermal fibroblasts with 200 μM Fe3+-citrate and 1 mM ascorbyl phosphate as a model of indirect UVA-induced skin damage. Lysates of fibroblasts treated this way showed a tenfold increase in MDA levels, whereas preincubation with tirilazad resulted in a significantly lower increase in MDA levels. Furthermore, in a comparison with the well-established radical scavenger Trolox, an α-tocopherol analogue, tirilazad offered better protection to the membranes. Our results demonstrate for the first time that the lazaroid tirilazad is an effective inhibitor of direct and indirect UVA-induced increases in MDA as a marker of lipid peroxidation in human dermal fibroblasts.



Similar content being viewed by others
References
Anderson DK, Braughler JM, Hall ED, Waters TR, McCall JM, Means ED (1988) Effects of treatment with U-74006F on neurological outcome following experimental spinal cord injury. J Neurosurg 69:562–567
Aubailly M, Santus R, Salmon S (1991) Ferrous iron release from ferritin by ultraviolet-A radiations. Photochem Photobiol 54:769–773
Barja de Quiroga G, Lopez-Torres M, Perez-Campo R (1992) Relationship between antioxidants, lipid peroxidation and aging. In: Emerit I, Chance B (eds) Free radical aging. Birkhäuser, Basel
Bash DE (1997) Sunlight and the onset of skin cancer. Trends Genet 13:410–414
Bath PM, Iddenden R, Bath FJ, Orgogozo JM (2001) Tirilazad for acute ischaemic stroke (Cochrane Library no. CD002087). Cochrane Library Review
Bayreuther K, Rodemann HP, Hommel R, Dittmann K, Albiez M, Francz PI (1988) Human skin fibroblasts in vitro differentiate along a terminal cell lineage. Proc Natl Acad Sci U S A 85:5112–5116
Biemond P, Swaak AJ, van Eijk HG, Koster JF (1988) Superoxide dependent iron release from ferritin in inflammatory diseases. Free Radic Biol Med 4:185–198
Bissett DL, McBride JF (1996) Synergistic topical photoprotection by a combination of the iron chelator 2-furildioxime and sunscreen. J Am Acad Dermatol 35:546–549
Bissett DL, Chatterjee R, Hannon DP (1991) Chronic ultraviolet radiation-induced increase in skin iron and the photoprotective effect of topically applied iron chelators. Photochem Photobiol 54:215–223
Blattner C, Bender K, Herrlich P, Rahmsdorf HJ (1998) Photoproducts in transcriptionally active DNA induce signal transduction to the delayed UV-responsive genes for collagenase and metallothionein. Oncogene 16:2827–2834
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Braughler JM, Pregenzer JF (1989) The 21-aminosteroid inhibitors of lipid peroxidation: reactions with lipid peroxyl and phenoxy radicals. Free Radic Biol Med 7:125–130
Braughler JM, Pregenzer JF, Chase RL, Ducan LA, Jacobsen EJ, McCall JM (1987) Novel 21-amino steroids as potent inhibitors of iron dependent lipid peroxidation. J Biol Chem 22:10438–10440
Buttgereit F, Hiepe F, Burmester GR (1997) The therapeutic potential of lazaroids (21-aminosteroids). A recent survey. Dtsch Med Wochenschr 122:1363–1367
Chaudière J (1994) Some chemical and biochemical constraints of oxidative stress in living cells. In: Rice-Evans CA, Burdon RH (eds) Free radical damage and its control. Elsevier Science, Amsterdam
Crutzen TJ (1992) Ultraviolet on the increase. Nature 356:104–105
Dissemond J, Schneider LA, Brenneisen P, Briviba K, Wenk J, Wlaschek M, Scharffetter-Kochanek K (2003) Generation and modulation of lipid peroxidation in normal and stably antioxidant enzymes overexpressing fibroblasts in vitro following expose to UVA irradiation. Br J Dermatol 149:341–349
Dorsch NW, Kassell NF, Sinkula MS (2001) Metaanalysis of trials of tirilazad mesylate in aneurysmal SAH. Acta Neurochir Suppl 77:233–235
Esterbauer H (1996) Estimation of peroxidative damage. Pathol Biol 44:25–28
Finkel T, Holbrook NJ (2000) Oxidants, oxidative stress and the biology of ageing. Nature 408:239–247
Fisher GJ, Datta SC, Talwar HS, Wang ZQ, Varani J, Kang S, Voorhees JJ (1996) Molecular basis of sun-induced premature skin ageing and retinoid antagonism. Nature 379:335–339
Girotti AW (2001) Photosensitized oxidation of membrane lipids: reaction pathways, cytotoxic effects, and cytoprotective mechanisms. J Photochem Photobiol B 63:103–113
Hall ED (1995) Inhibition of lipid peroxidation in central nervous system trauma and ischemia. J Neurol Sci 134:79–83
Hall ED, Yonkers PA (1988) Attenuation of postischemic cerebral hypoperfusion by the 21-aminosteroid U74006F. Stroke 19:340–344
Hall ED, Yonkers PA, McCall JM, Braughler JM (1988) Effects of the 21-aminosteroid U74006F on experimental head injury in mice. J Neurosurg 68:456–461
Halliwell B (2000) A super way to kill cancer cells? Nat Med 6:1105–1106
Halliwell B (2001) Role of free radicals in the neurodegenerative diseases: therapeutic implications for antioxidant treatment. Drugs Aging 18:685–716
Knight JA (1998) Free radicals: their history and current status in aging and disease. Ann Clin Lab Sci 28:331–346
Koc RK, Kurtsoy A, Pasaoglu H, Karakucuk EI, Oktem IS, Meral M (1999) Lipid peroxidation and oedema in experimental brain injury: comparison of treatment with methylprednisolone, tirilazad mesylate and vitamin E. Res Exp Med 199:21–28
Kraemer KM (1997) Sunlight and skin cancer. Another link revealed. Proc Natl Acad Sci U S A 94:11–14
Liochev SI, Fridovich I (1994) The role of O2 - in the production of HO.: in vitro and in vivo. Free Radic Biol Med 16:29–33
Marnett LJ (1999) Lipid peroxidation—DNA damage by malondialdehyde. Mutat Res 424:83–95
Meyer M, Schreck R, Baeuerle PA (1993) H2O2 and antioxidants have opposite effects on activation of NF-kappa B and AP-1 in intact cells: AP-1 as secondary antioxidant-responsive factor. EMBO J 12:2005–2015
Minotti G, Aust SD (1987) The role of iron in the initiation of lipid peroxidation. Chem Phys Lipids 44:191–208
Mitani H, Koshiishi I, Sumita T, Imanari T (2001) Prevention of the photodamage in the hairless mouse dorsal skin by kojic acid as an iron chelator. Eur J Pharmacol 411:169–174
Morliere P, Moysan A, Tirache I (1995) Action spectrum for UV-induced lipid peroxidation in cultured human fibroblasts. Free Radic Biol Med 19:365–371
Morliere P, Salmon S, Aubailly M, Risler A, Santus R (1997) Sensitisation of skin fibroblasts to UVA by excess iron. Biochim Biophys Acta 1334:283–290
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Packer L (1994) Ultraviolet radiation (UVA, UVB) and skin antioxidants. In: Rice-Evans CA, Burdon RH (eds) Free radical damage and its control. Elsevier Science, Amsterdam
Podda M, Traber MG, Weber C, Yan LJ, Packer L (1998) UV-irradiation depletes antioxidants and causes oxidative damage in a model of human skin. Free Radic Biol Med 24:55–65
Pourzand C, Watkin RD, Brown JE, Tyrrell RM (1999) Ultraviolet A radiation induces immediate release of iron in human primary skin fibroblasts: the role of ferritin. Proc Natl Acad Sci U S A 96:6751–6756
Scharffetter-Kochanek K, Wlaschek M, Brenneisen P, Schauen M, Blaudschun R, Wenk J (1997) UV-induced reactive oxygen species in photocarcinogenesis and photoaging. Biol Chem 378:1247–1257
Scharffetter-Kochanek K, Brenneisen P, Wenk J, Hermann G, Ma W, Kuhr L, Meewes C, Wlaschek M (2000) Photoaging of the skin from phenotype to mechanisms. Exp Gerontol 35:307–316
Schmid D, Burmester GR, Tripmacher R, Fici G, von Voigtlander P, Buttgereit F (2001) Short-term effects of the 21-aminosteroid lazaroid tirilazad mesylate (PNU-74006F) and the pyrrolopyrimidine lazaroid PNU-101033E on energy metabolism of human peripheral blood mononuclear cells. Biosci Rep 21:101–110
Sies H (1986) Biochemistry of oxidative stress. Angew Chem 25:1058–1071
Tadolini B, Hakim G (1996) The mechanism of iron (III) stimulation of lipid peroxidation. Free Radic Res 25:221–227
Trommer H, Bottcher R, Poppl A, Hoentsch J, Wartewig S, Neubert RH (2002) Role of ascorbic acid in stratum corneum lipid models expose to UV irradiation. Pharm Res 19:982–990
Vollmer DG, Kassel NF, Hongo K, Ogawa H, Tsukahara T (1989) Effect of the non-glucocorticoid 21-aminosteroid U74006F on experimental cerebral vasospasm. Surg Neurol 31:190–194
Wang S, Lantz RC, Rider E, Chen GJ, Breceda V, Hays AM, Robledo R, Tollinger BJ, Dinesh SVR, Witten ML (1997) A free radical scavenger (lazaroid U75412E) attenuates tumor necrosis factor-alpha generation in a rabbit model of smoke-induced lung injury. Respiration 64:358–363
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dissemond, J., Schneider, L.A., Wlaschek, M. et al. The lazaroid tirilazad is a new inhibitor of direct and indirect UVA-induced lipid peroxidation in human dermal fibroblasts. Arch Dermatol Res 295, 287–292 (2003). https://doi.org/10.1007/s00403-003-0432-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-003-0432-5