Abstract
Background
Dimethyl fumarate (DMF) tolerability and safety in multiple sclerosis (MS) has been analyzed in randomized clinical trials. Real-life studies are needed to assess possible harms of this therapy in a wider MS population.
Objective
To evaluate DMF tolerability, safety and persistence in MS in a real-world setting.
Methods
We conducted a multicenter prospective study of patients who started DMF, attended in 16 public hospitals of Spain. A specific database was elaborated to collect data on most frequent adverse events (AE). Regression models were used to analyze the effect of demographic and clinical characteristics on risk of AEs and DMF discontinuation.
Results
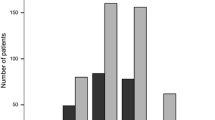
We collected data of 886 patients (2681 patients/years-exposition) with median 39.5 (IQR 23, 51.5) months on DMF exposure; 25.3% were treatment naïve and 74.7% switched to DMF from other disease-modifying therapies. DMF was discontinued in 29.9% of patients, in 13.2% due to AEs and in 13.5% to inefficacy. AEs were experienced by 71.2%, being flushing the most frequent (44.1%), 5.4% developed grade III lymphopenia, without cases of grade IV. Females showed a higher risk of flushing and gastroenteric symptoms (OR 1.49, p = 0.011; OR 1.69, p = 0.001, respectively); lymphopenia was associated with older age (OR 1.04, p < 0.001), and a higher EDSS with lymphopenia (OR 1.10, p = 0.035) and DMF withdrawal (HR 1.43, p = 0.012). No safety problems were reported.
Conclusions
Our findings confirm good tolerability and safety of DMF in real-world setting and suggest that women have an increased risk of AEs and higher baseline disability involves greater risk of drug discontinuation.


Similar content being viewed by others
References
Frischer JM, Weigand SD, Guo Y, Kale N, Parisi JE, Pirko I et al (2015) Clinical and pathological insights into the dynamic nature of the white matter multiple sclerosis plaque. Ann Neurol 78:710–721
Linker RA, Lee DH, Ryan S, Van Dam AM, Conrad R, Bista P et al (2011) Fumaric acid esters exert neuroprotective effects in neuroinflammation via activation of the Nrf2 antioxidant pathway. Brain 134:678–692
Fox RJ, Miller DH, Phillips JT, Hutchinson M, Havrdova E, Kita M et al (2012) Placebo-controlled phase 3 study of oral BG-12 or glatiramer in multiple sclerosis. N Engl J Med 367:1087–1097
Gold R, Kappos L, Arnold DL, Bar-Or A, Giovannoni G, Selmaj K et al (2012) Placebo-controlled phase 3 study of oral BG-12 for relapsing multiple sclerosis. N Engl J Med 367:1098–1107
Havrdova E, Giovannoni G, Gold R, Fox RJ, Kappos L, Phillips JT et al (2017) Effect of delayed-release dimethyl fumarate on no evidence of disease activity in relapsing–remitting multiple sclerosis: integrated analysis of the phase III DEFINE and CONFIRM studies. Eur J Neurol 24:726–733
Gold R, Arnold DL, Bar-Or A, Hutchinson M, Kappos L, Havrdova E et al (2017) Long-term effects of delayed-release dimethyl fumarate in multiple sclerosis: Interim analysis of ENDORSE, a randomized extension study. Mult Scler 23:253–265
Cohen TA, Goto S, Schreiber K, Torp-Pedersen C (2015) Why do we need observational studies of everyday patients in the real-life setting? Eur Heart J Supp. https://doi.org/10.1093/eurheartj/suv035
Kalincik T, Kuhle J, Pucci E, Rojas JI, Tsolaki M, Sirbu CA et al (2017) Data quality evaluation for observational multiple sclerosis registries. Mult Scler J 23:647–655
Mallucci G, Annovazzi P, Miante S, Torri-Clerici V, Matta M, La Gioia S et al (2018) Two-year real-life efficacy, tolerability and safety of dimethyl fumarate in an Italian multicentre study. J Neurol 265:1850–1859
Mirabella M, Prosperini L, Lucchini M, Boffa L, Borriello G, Buscarinu MC et al (2018) Safety and efficacy of dimethyl fumarate in multiple sclerosis: an Italian, multicenter, real-world Study. CNS Drugs 32:963–970
Miclea A, Leussink VI, Hartung HP, Gold R, Hoepner R (2016) Safety and efficacy of dimethyl fumarate in multiple sclerosis: a multi-center observational study. J Neurol 263:1626–1632
Sejbaek T, Nybo M, Petersen T, Illes Z (2018) Real-life persistence and tolerability with dimethyl fumarate. Mult Scler Relat Disord 24:42–46
Alroughani R, Ahmed SF, Behbehani R, Al-Hashel J (2017) Effectiveness and safety of dimethyl fumarate treatment in relapsing multiple sclerosis patients: real-world evidence. Neurol Ther 6:189–196
Purchiaroni F, Salvetti M, Buscarinu MC, Annibale B (2016) Eosinophilic gastroenteritis in a woman with multiple esclerosis on dimethyl fumarate. Neurlogy 87:952–953
Min J, Cohan S, Alvarez E, Sloane J, Phillips JT, Van der Walt A et al (2019) Real-world characterization of dimethyl fumarate-related gastrointestinal events in multiple sclerosis: management strategies to improve persistence on treatment and patient outcomes. Neurol Ther 8:109–119. https://doi.org/10.1007/s40120-019-0127-2
Marrie RA, Reider N, Cohen J, Stuve O, Trojano M, Sorensen PS et al (2015) A systematic review of the incidence and prevalence of cancer in multiple sclerosis. Mult Scler J 21:294–304. https://doi.org/10.1177/1352458514564489
Granqvist M, Burman J, Gunnarsson M, Lycke J, Nilsson P, Olsson T et al (2019) Comparative effectiveness of dimethyl fumarate as the initial and secondary treatment for MS. Mult Scler J. https://doi.org/10.1177/1352458519866600
Acknowledgements
Dimethyl fumarate Study Group: M. Rosario Blasco, University Hospital Puerta de Hierro Majadahonda, Spain; J. Manuel García,Dominguez, University Hospital “Gregorio Marañón”, Madrid, Spain; Laura Rubio, University Hospital Villalba. Madrid, Spain; Patricia Perea Hospital Complex Torrecárdenas, Almería, Spain; Paula Salgado, University Hospital 12 de Octubre, Madrid, Spain; Aida Orviz, University Hospital Fundación Jimenez-Díaz, Madrid, Spain; Ricardo Ginestal, University Hospital Fundación Jimenez-Díaz, Madrid, Spain; Cristina Andreu, Universidad Europea de Madrid, Faculty of Biomedical and Health Sciences.
Funding
This study has been conducted without any funding.
Author information
Authors and Affiliations
Consortia
Contributions
Design/conceptualization of the study: YA, JS, SU, BP, VG, SS, MG. Analysis/interpretation of the data: YA, JS, IT. Drafting and revising the manuscript: YA, JS, BP. Statistical analysis: JS, YA, IT, IM, CA. Acquisition of data and revising the manuscript for intellectual content: all authors. All authors give final approval of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest for this work.
Additional information
Details of the DMF Study Group are given in Acknowledgement section.
Rights and permissions
About this article
Cite this article
Sabin, J., Urtiaga, S., Pilo, B. et al. Tolerability and safety of dimethyl fumarate in relapsing multiple sclerosis: a prospective observational multicenter study in a real-life Spanish population. J Neurol 267, 2362–2371 (2020). https://doi.org/10.1007/s00415-020-09848-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-09848-7