Abstract
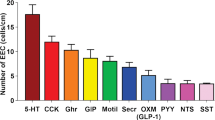
Enteroendocrine cells were historically classified by a letter code, each linked to a single hormone, deduced to be the only hormone produced by the cell. One type, the L cell, was recognised to store and secrete two products, peptide YY (PYY) and glucagon-related peptides. Many other exceptions to the one-cell one-hormone classifications have been reported over the last 40 years or so, and yet the one-hormone dogma has persisted. In the last 6 years, a plethora of data has appeared that makes the concept unviable. Here, we describe the evidence that multiple hormone transcripts and their products reside in single cells and evidence that the hormones are often, but not always, processed into separate storage vesicles. It has become clear that most enteroendocrine cells contain multiple hormones. For example, most secretin cells contain 5-hydroxytryptamine (5-HT), and in mouse many of these also contain cholecystokinin (CCK). Furthermore, CCK cells also commonly store ghrelin, glucose-dependent insulinotropic peptide (GIP), glucagon-like peptide-1 (GLP-1), neurotensin, and PYY. Several hormones, for example, secretin and 5-HT, are in separate storage vesicles at a subcellular level. Hormone patterns can differ considerably between species. Another complication is that relative levels of expression vary substantially. This means that data are significantly influenced by the sensitivities of detection techniques. For example, a hormone that can be detected in storage vesicles by super-resolution microscopy may not be above threshold for detection by conventional fluorescence microscopy. New nomenclature for cell clusters with common attributes will need to be devised and old classifications abandoned.



Similar content being viewed by others
References
Aiken KD, Kisslinger JA, Roth KA (1994) Immunohistochemical studies indicate multiple enteroendocrine cell differentiation pathways in the mouse proximal small intestine. Dev Dyn 201:63–70
Alcaino C, Knutson KR, Treichel AJ, Yildiz G, Strege PR, Linden DR, Li JH, Leiter AB, Szurszewski JH, Farrugia G, Beyder A (2018) A population of gut epithelial enterochromaffin cells is mechanosensitive and requires Piezo2 to convert force into serotonin release. Proc Natl Acad Sci USA. doi.https://doi.org/10.1073/pnas.1804938115
Ali Rachedi A, Varndell IM, Adrian TE, Gapp DA, Van Noorden S, Bloom SR, Polak JM (1984) Peptide YY immunoreactivity is co-stored with glucagon related immunoreactants in endocrine cells of the gut and pancreas. Histochemistry 80:487–491
Andrews PLR, Davis CJ, Bingham S, Davidson HI, Hawthorn J, Maskell L (1990) The abdominal visceral innervation and the emetic reflex: pathways, pharmacology, and plasticity. Can J Physiol Pharmacol 68:325–345
Asmar M, Asmar A, Simonsen L, Gasbjerg LS, Sparre-Ulrich AH, Rosenkilde M, Hartmann B, Dela F, Holst JJ, Bülow J (2017) The gluco- and liporegulatory and the vasodilatory effects of glucose-dependent insulinotropic polypeptide (GIP) are abolished by an antagonist of the human GIP receptor. Diabetes 66:db170480
Bagger JI, Holst JJ, Hartmann B, Andersen B, Knop FK, Vilsbøll T (2015) Effect of oxyntomodulin, glucagon, GLP-1, and combined glucagon + GLP-1 infusion on food intake, appetite, and resting energy expenditure. J Clin Endocrinol Metab 100:4541–4552
Baggio LL, Drucker DJ (2007) Biology of incretins: GLP-1 and GIP. Gastroenterology 132:2131–2157
Batterham RL, Cowley MA, Small CJ et al (2002) Gut hormone PYY3 – 36 physiologically inhibits food intake. Nature 418:650
Bayliss WM, Starling EH (1902) The mechanism of pancreatic secretion. J Physiol (Lond) 28:325–353
Billing LJ, Smith CA, Larraufie P et al (2018) Co-storage and release of insulin-like peptide-5, glucagon-like peptide-1 and peptideYY from murine and human colonic enteroendocrine cells. Molec Metab. 16:65–75. doi.https://doi.org/10.1016/j.molmet.2018.07.011
Böttcher G, Alumets J, Håkanson R, Sundler F (1986) Co-existence of glicentin and peptide YY in colorectal L-cells in cat and man. An electron microscopic study. Regul Pept 13:283–291
Burrin DG, Stoll B, Guan X, Cui L, Chang X, Hadsell D (2007) GLP-2 rapidly activates divergent intracellular signaling pathways involved in intestinal cell survival and proliferation in neonatal piglets. Am J Physiol Endocrinol Metab 292:E281–E291
Carmichael J, Cantwell BMJ, Edwards CM, Zussman BD, Thompson S, Rapeport WG, Harris AL (1989) A pharmacokinetic study of granisetron (BRL 43694A), a selective 5-HT3 receptor antagonist: correlation with anti-emetic response. Cancer Chemother Pharmacol 24:45–49
Castan I, Valet P, Larrouy D, Voisin T, Remaury A, Daviaud D, Laburthe M, Lafontan M (1993) Distribution of PYY receptors in human fat cells: an antilipolytic system alongside the α2-adrenegic system. Am J Physiol Endocrinol Metab 265:E74–E80
Cetin Y (1990) Secretin-cells of the mammalian intestine contain serotonin. Histochemistry 93:601–606
Cheung GWC, Kokorovic A, Lam CKL, Chari M, Lam TKT (2009) Intestinal cholecystokinin controls glucose production through a neuronal network. Cell Metab 10:99–109
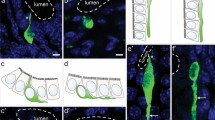
Cho H-J, Robinson ES, Rivera LR, McMillan PJ, Testro A, Nikfarjam M, Bravo DM, Furness JB (2014) Glucagon-like peptide 1 and peptide YY are in separate storage organelles in enteroendocrine cells. Cell Tissue Res 357:63–69
Cho H-J, Kosari S, Hunne B, Callaghan B, Rivera LR, Bravo DM, Furness JB (2015) Differences in hormone localisation patterns of K and L type enteroendocrine cells in the mouse and pig small intestine and colon. Cell Tissue Res 359:693–698
Christensen MB, Lund A, Calanna S, Jørgensen NR, Holst JJ, Vilsbøll T, Knop FK (2018) Glucose-dependent insulinotropic polypeptide (GIP) inhibits bone resorption independently of insulin and glycemia. J Clin Endocrinol Metab 103:288–294
Coate KC, Kliewer SA, Mangelsdorf DJ (2014) SnapShot: Hormones of the gastrointestinal tract. Cell 159:1478–1479
Collden G, Tschop MH, Muller TD (2017) Therapeutic potential of targeting the ghrelin pathway. Int J Mol Sci 18:798–837
Cummings DE, Purnell JQ, Frayo RS, Schmidova K, Wisse BE, Weigle DS (2001) A preprandial rise in plasma ghrelin levels suggests a role in meal initiation in humans. Diabetes 50:1714–1719
Drucker DJ (2015) Evolving concepts and translational relevance of enteroendocrine cell biology. J Clin Endocrinol Metab 101:778–786
Edkins JS (1906) The chemical mechanism of gastric secretion. J Physiol (Lond) 34:133–144
Egerod KL, Engelstoft MS, Grunddal KV et al (2012) A major lineage of enteroendocrine cells coexpress CCK, secretin, GIP, GLP-1, PYY, and neurotensin but not somatostatin. Endocrinology 153:5782–5795
Egerod KL, Engelstoft MS, Lund ML, Grunddal KV, Zhao M, Barir-Jensen D, Nygaard EB, Petersen N, Holst JJ, Schwartz TW (2015) Transcriptional and functional characterization of the G protein-coupled receptor repertoire of gastric somatostatin cells. Endocrinology 156:3909–3923
Eissele R, Göke R, Willemer S, Harthus H-P, Vermeer H, Arnold R, Göke B (1992) Glucagon-like peptide-I cells in the gastrointestinal tract and pancreas of rat, pig and man. Eur J Clin Invest 22:283–291
Engelstoft MJ, Lund ML, Grunddal KV, Egerod KL, Osborne-Lawrence S, Poulsen SS, Zigman JM, Schwartz TW (2015) Research resource: a chromogranin A reporter for serotonin and histamine secreting enteroendocrine cells. Mol Endocrinol 29:1658–1671
Fakhry J, Wang J, Martins P, Fothergill LJ, Hunne B, Prieur P, Shulkes A, Rehfeld JF, Callaghan B, Furness JB (2017) Distribution and characterisation of CCK containing enteroendocrine cells of the mouse small and large intestine. Cell Tissue Res 369:245–253
Fothergill LJ, Callaghan B, Hunne B, Bravo DM, Furness JB (2017) Costorage of enteroendocrine hormones evaluated at the cell and subcellular levels in male mice. Endocrinology 158:2113–2123
Fothergill LJ, Ringuet MT, Sioras E, Hunne B, Fazio Coles TE, Martins P, Furness JB (2018) Cellular and sub-cellular localisation of oxyntomodulin-like immunoreactivity in enteroendocrine cells of human, mouse, pig, and rat. Cell Tissue Res (In press)
Galsgaard KD, Winther-Sørensen M, Ørskov C et al (2018) Disruption of glucagon receptor signaling causes hyperaminoacidemia exposing a possible liver-alpha-cell axis. Am J Physiol Endocrinol Metab 314:E93–E103
Glass LL, Calero-Nieto FJ, Jawaid W, Larraufie P, Kay RG, Göttgens B, Reimann F, Gribble FM (2017) Single-cell RNA-sequencing reveals a distinct population of proglucagon-expressing cells specific to the mouse upper small intestine. Mol Metab 6:1296–1303
Gribble FM, Reimann F (2016) Enteroendocrine cells: chemosensors in the intestinal epithelium. Annu Rev Physiol 78:277–299
Gribble FM, Reimann F, Roberts GP (2018) Gastrointestinal hormones. In: Physiology of the gastrointestinal tract, 6. (eds.) HM Said. Elsevier, New York, pp. 31–70
Grunddal KV, Ratner CF, Svendsen B et al (2015) Neurotensin is co-expressed, co-released and acts together with GLP-1 and PYY in enteroendocrine control of metabolism. Endocrinology 157:176–194
Gutzwiller J et al (1999) Glucagon-like peptide-1: a potent regulator of food intake in humans. Gut 44:81–86
Haber AL, Biton M, Rogel N et al (2017) A single-cell survey of the small intestinal epithelium. Nature 551:333
Habib AM, Richards P, Cairns LS, Rogers GJ, Bannon CAM, Parker HE, Morley TCE, Yeo GSH, Reimann F, Gribble FM (2012) Overlap of endocrine hormone expression in the mouse intestine revealed by transcriptional profiling and flow cytometry. Endocrinology 153:3054–3065
Håkanson R, Ding X-Q, Norlén P, Chen D (1995) Circulating pancreastatin is a marker for the enterochromaffin-like cells of the rat stomach. Gastroenterology 108:1445–1452
Heidenhain R (1870) Untersuchungen über den Bau der Labdrüsen. Arch Mikroskop Anat 6:368–406
Helander HF, Fändriks L (2012) The enteroendocrine “letter cells”—time for a new nomenclature? Scand J Gastroenterol 47:3–12
Hörsch D, Fink T, Göke B, Arnold R, Büchler M, Weihe E (1994) Distribution and chemical phenotypes of neuroendocrine cells in the human anal canal. Regul Pept 54:527–542
Krieger J-P et al (2018) Glucagon-like peptide-1 regulates brown adipose tissue thermogenesis via the gut-brain axis in rats. Am J Physiol 315:R708–R720
Lieverse RJ, Jansen JBMJ, Masclee AAM, Rovati LC, Lamers CBHW (1994) Effect of a low dose of intraduodenal fat on satiety in humans: studies using the type A cholecystokinin receptor antagonist loxiglumide. Gut 35:501–505
Lopez MJ, Upchurch BH, Rindi G, Leiter AB (1995) Studies in transgenic mice reveal potential relationships between secretin-producing cells and other endocrine cell types. J Biol Chem 270:885–891
Lukinius AIC, Ericsson JLE, Lundqvist MK, Wilander EMO (1986) Ultrastructural localization of serotonin and polypeptide YY (PYY) in endocrine cells of the human rectum. J Histochem Cytochem 34:719–726
Lund ML, Egerod KL, Engelstoft MS, Dmytriyeva O, Theodorsson E, Patel BA, Schwartz TW (2018) Enterochromaffin 5-HT cells - a major target for GLP-1 and gut microbial metabolites. Mol Metab 11:70–83
Mani BK, Zigman JM (2017) Ghrelin as a survival hormone. Trends Endocrinol Metab 28:843–854
Martin AM, Lumsden AL, Young RL, Jessup CF, Spencer NJ, Keating DJ (2017) The nutrient-sensing repertoires of mouse enterochromaffin cells differ between duodenum and colon. Neurogastroenterol Motil 29:e13046
Martins P, Fakhry J, Chaves de Oliveira E, Hunne B, Fothergill LJ, Ringuet M, d’Ávila Reis D, Rehfeld JF, Callaghan B, Furness JB (2017) Analysis of enteroendocrine cell populations in the human colon. Cell Tissue Res 367:361–368
Maton PN, Selden AC, Chadwick VS (1984) Differential distribution of molecular forms of cholecystokinin in human and porcine small intestinal mucosa. Regul Pept 8:9–19
Mawe GM, Hoffman JM (2013) Serotonin signalling in the gut—functions, dysfunctions and therapeutic targets. Nat Rev Gastroenterol Hepatol 10:473–486
Mazzawi T, El-Salhy M (2016) Changes in small intestinal chromogranin A-immunoreactive cell densities in patients with irritable bowel syndrome after receiving dietary guidance. Int J Mol Med 37:1247–1253
Moriya R, Shirakura T, Ito J, Mashiko S, Seo T (2009) Activation of sodium-glucose cotransporter 1 ameliorates hyperglycemia by mediating incretin secretion in mice. Am J Physiol Endocrinol Metab 297:E1358–E1365
Mortensen K, Christensen LL, Holst JJ, Orskov C (2003) GLP-1 and GIP are colocalized in a subset of endocrine cells in the small intestine. Regul Pept 114:189–196
Nilsson O, Bilchik AJ, Goldenring JR, Ballantyne GH, Adrian TE, Modlin IM (1991) Distribution and immunocytochemical colocalization of peptide YY and enteroglucagon in endocrine cells of the rabbit colon. Endocrinology 129:139–148
Pearse AGE, Polak JM, Bloom SR, Adams C, Dryburgh JR, Brown JC (1974) Enterochromaffin cells of the mammalian small intestine as the source of motilin. Virchows Arch B 16:111–120
Portela–Gomes GM, Stridsberg M (2002) Chromogranin A in the human gastrointestinal tract: an immunocytochemical study with region-specific antibodies. J Histochem Cytochem 50:1487–1492
Rehfeld JF (1978) Immunochemical studies on cholecystokinin. II. Distribution and molecular heterogeneity in the central nervous system and small intestine of man and hog. J Biol Chem 253:4022–4030
Rehfeld JF (2012) Beginnings: A reflection on the history of gastrointestinal endocrinology. Regul Pept 177:S1–S5
Reynaud Y, Fakhry J, Fothergill L, Callaghan B, Ringuet MT, Hunne B, Bravo DM, Furness JB (2016) The chemical coding of 5-hydroxytryptamine containing enteroendocrine cells in the mouse gastrointestinal tract. Cell Tissue Res 364:489–497
Roth KA, Gordon JI (1990) Spatial differentiation of the intestinal epithelium: Analysis of enteroendocrine cells containing immunoreactive serotonin, secretin, and substance P in normal and transgenic mice. Proc Natl Acad Sci USA 87:6408–6412
Roth KA, Kim S, Gordon JI (1992) Immunocytochemical studies suggest two pathways for enteroendocrine cell differentiation in the colon. Am J Physiol 263:G174–G180
Sanger GJ, Furness JB (2016) Ghrelin and motilin receptors as drug targets for gastrointestinal disorders. Nat Rev Gastroenterol Hepatol 19:38–48
Sanger GJ, Holbrook JD, Andrews PLR (2011) The translational value of rodent gastrointestinal functions: a cautionary tale. Trends Pharmacol Sci 32:402–409
Schjoldager B et al (1988) Oxyntomodulin: a potential hormone from the distal gut. Pharmacokinetics and effects on gastric acid and insulin secretion in man. Eur J Clin Invest 18:499–503
Smith PH, Davis BJ, Seino Y, Yanaihara N (1981) Localization of motilin-containing cells in the intestinal tract of mammals: a further comparison using region-specific motilin antisera. Gen Comp Endocrinol 44:288–291
Stengel A, Goebel M, Yakubov I, Wang L, Witcher D, Coskun T, Tache Y, Scachs G, Lambrecht NWG (2009) Identification and characterization of nesfatin-1 immunoreactivity in endocrine cell types of the rat gastric oxyntic mucosa. Endocrinology 150:232–238
Stengel A, Goebel M, Wang L, Taché Y (2010a) Ghrelin, des-acyl ghrelin and nesfatin-1 in gastric X/A-like cells: Role as regulators of food intake and body weight. Peptides 31:357–369
Stengel A, Goebel M, Wang L, Taché Y, Sachs G, Lambrecht NWG (2010b) Differential distribution of ghrelin-O-acyltransferase (GOAT) immunoreactive cells in the mouse and rat gastric oxyntic mucosa. Biochem Biophys Res Commun 392:67–71
Sumara G, Sumara O, Kim JK, Karsenty G (2012) Gut-derived serotonin is a multifunctional determinant to fasting adaptation. Cell Metab 16:588–600
Svendsen B, Pedersen J, Albrechtsen NJW, Hartmann B, Toräng S, Rehfeld JF, Poulsen SS, Holst JJ (2015) An analysis of cosecretion and coexpression of gut hormones from male rat proximal and distal small intestine. Endocrinology 156:847–857
Svendsen B, Pais R, Engelstoft MS et al (2016) GLP1- and GIP-producing cells rarely overlap and differ by bombesin receptor-2 expression and responsiveness. J Endocrinol 228:39–48
Sykaras AG, Demenis C, Cheng L, Pisitkun T, Mclaughlin JT, Fenton RA, Smith CP (2014) Duodenal CCK cells from male mice express multiple hormones including ghrelin. Endocrinology 155:3339–3351
Theodorakis MJ, Carlson O, Michopoulos S, Doyle ME, Juhaszova M, Petraki K, Egan JM (2006) Human duodenal enteroendocrine cells: source of both incretin peptides, GLP-1 and GIP. Am J Physiol Endocrinol Metab 290:E550–E559
Usellini L, Finzi G, Riva C, Capella C, Mochizuki T, Yanaihara C, Yanaihara N, Solcia E (1990) Ultrastructural identification of human secretin cells by the immunogold technique.Their costorage of chromogranin A and serotonin. Histochemistry 94:113–120
Vialli M, Erspamer V (1933) Celluli enterocromaffini e cellule basigranulose acidofile nei vertebrati. Z Zellforsch Mikroskop Anat 19:743–773
Wewer Albrechtsen NJ, Kuhre RE, Toräng S, Holst JJ (2016) The intestinal distribution pattern of appetite and glucose regulatory peptides in mice, rats and pigs. BMC Res Notes 9:60
Wierup N, Björkqvist M, WestrÓ§m B, Pierzynowski S, Sundler F, Sjölund K (2007) Ghrelin and motilin are cosecreted from a prominent endocrine cell population in the small intestine. J Clin Endocrinol Metab 92:3573–3581
Wilder-Smith OHG, Borgeat A, Chappuis P, Fathi M, Forni M (1993) Urinary serotonin metabolite excretion during cisplatin chemotherapy. Cancer 72:2239–2241
Wong IPL, Driessler F, Khor EC, Shi Y-C, Hörmer B, Nguyen AD, Enriquez RF, Eisman JA, Sainsbury A, Herzog H, Baldock PA (2012) Peptide YY regulates bone remodeling in mice: a link between gut and skeletal biology. PLoS One 7:e40038
Worthington JJ, Reimann F, Gribble FM (2018) Enteroendocrine cells-sensory sentinels of the intestinal environment and orchestrators of mucosal immunity. Mucosal Immunol 11:3
Yadav VK, Ryu J-H, Suda N et al (2008) Lrp5 controls bone formation by inhibiting serotonin synthesis in the duodenum. Cell 135:825–837
Acknowledgements
This work was supported by NIH (SPARC) grant ID # OT2OD023847 (TW Powley) to JBF and an Australian Government Research Training Program Scholarship to LJF.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fothergill, L.J., Furness, J.B. Diversity of enteroendocrine cells investigated at cellular and subcellular levels: the need for a new classification scheme. Histochem Cell Biol 150, 693–702 (2018). https://doi.org/10.1007/s00418-018-1746-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-018-1746-x