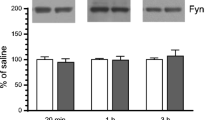
Abstract
The medial septum/diagonal band (MS/DB) is a relay region connecting the hypothalamus and brainstem with the hippocampus, and both the MS/DB and dorsal/ventral hippocampus receive strong topographic GABA/peptidergic projections from the nucleus incertus of the pontine tegmentum. The neuropeptide relaxin-3, released by these neurons, is the cognate ligand for a Gi/o-protein-coupled receptor, RXFP3, which is highly expressed within the MS/DB, and both cholinergic and GABAergic neurons in this region of rat brain receive relaxin-3 positive terminals/boutons. Comprehensive in vitro studies have demonstrated that the cell signaling pathways altered by RXFP3 stimulation, include inhibition of forskolin-activated cAMP levels and activation of ERK phosphorylation. In this study we investigated whether intracerebroventricular (icv) injection of RXFP3-A2, a selective relaxin-3 receptor agonist, altered ERK phosphorylation levels in the MS/DB of adult male rats. We subsequently assessed the neurochemical phenotype of phosphorylated (p) ERK-positive neurons in MS/DB after icv RXFP3-A2 administration by dual-label immunostaining for pERK and neuronal markers for cholinergic and GABAergic neurons. Central RXFP3-A2 injection significantly increased levels of pERK immunoreactivity (IR) in MS/DB at 20 and 90 min post-injection, compared to vehicle and naive levels. In addition, RXFP3-A2 increased the number of cells expressing pERK-IR in the MS/DB at 90 (but not 20) min post-injection in cholinergic (but not GABAergic) neurons, which also expressed putative RXFP3-IR. Moreover, icv injection of RXFP3-A2 impaired alternation in a delayed spontaneous T-maze test of spatial working memory. The presence of RXFP3-like IR and the RXFP3-related activation of the MAPK/ERK pathway in MS/DB cholinergic neurons identifies them as a key target of ascending relaxin-3 projections with implications for the acute and chronic modulation of cholinergic neuron activity and function by relaxin-3/RXFP3 signaling.







Similar content being viewed by others
References
Ahn HJ, Hernandez CM, Levenson JM et al (2008) c-Rel, an NF-kappaB family transcription factor, is required for hippocampal long-term synaptic plasticity and memory formation. Learn Mem 15:539–549. doi:10.1101/lm.866408
Bannerman DM, Rawlins JN, McHugh SB et al (2004) Regional dissociations within the hippocampus-memory and anxiety. Neurosci Biobehav Rev 28:273–283. doi:10.1016/j.neubiorev.2004.03.004
Bathgate RAD, Samuel CS, Burazin TCD et al (2002) Human relaxin gene 3 (H3) and the equivalent mouse relaxin (M3) gene. Novel members of the relaxin peptide family. J Biol Chem 277:1148–1157. doi:10.1074/jbc.M107882200
Borhegyi Z, Freund TF (1998) Dual projection from the medial septum to the supramammillary nucleus in the rat. Brain Res Bull 46:453–459
Brito GNO, Thomas GJ (1981) T-maze alternation, response patterning, and septo-hippocampal circuitry in rats. Behav Brain Res 3:319–340. doi:10.1016/0166-4328(81)90003-6
Burazin TCD, Bathgate RAD, Macris M et al (2002) Restricted, but abundant, expression of the novel rat gene-3 (R3) relaxin in the dorsal tegmental region of brain. J Neurochem 82:1553–1557
Buzsáki G, Moser EI (2013) Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nat Neurosci 16:130–138. doi:10.1038/nn.3304
Calvez J, de Ávila C, Matte L-O et al (2016) Role of relaxin-3/RXFP3 system in stress-induced binge-like eating in female rats. Neuropharmacology 102:207–215. doi:10.1016/j.neuropharm.2015.11.014
Cammarota M, Bevilaqua LRM, Ardenghi P et al (2000) Learning-associated activation of nuclear MAPK, CREB and Elk-1, along with Fos production, in the rat hippocampus after a one-trial avoidance learning: abolition by NMDA receptor blockade. Mol Brain Res 76:36–46. doi:10.1016/S0169-328X(99)00329-0
Davis S, Vanhoutte P, Pages C et al (2000) The MAPK/ERK cascade targets both Elk-1 and cAMP response element-binding protein to control long-term potentiation-dependent gene expression in the dentate gyrus in vivo. J Neurosci 20:4563–4572
Fadda F, Melis F, Stancampiano R (1996) Increased hippocampal acetylcholine release during a working memory task. Eur J Pharmacol 307:R1–R2
Fitz NF, Gibbs RB, Johnson DA (2008) Selective lesion of septal cholinergic neurons in rats impairs acquisition of a delayed matching to position T-maze task by delaying the shift from a response to a place strategy. Brain Res Bull 77:356–360. doi:10.1016/j.brainresbull.2008.08.016
Ford B, Holmes CJ, Mainville L, Jones BE (1995) GABAergic neurons in the rat pontomesencephalic tegmentum: codistribution with cholinergic and other tegmental neurons projecting to the posterior lateral hypothalamus. J Comp Neurol 363:177–196. doi:10.1002/cne.903630203
Freund TF, Antal M (1988) GABA-containing neurons in the septum control inhibitory interneurons in the hippocampus. Nature 336:170–173. doi:10.1038/336170a0
Freund TF, Gulyas AI (1997) Inhibitory control of GABAergic interneurons in the hippocampus. Can J Physiol Pharmacol 75:479–487
Fuhrmann F, Justus D, Sosulina L et al (2015) Locomotion, theta oscillations, and the speed-correlated firing of hippocampal neurons are controlled by a medial septal glutamatergic circuit. Neuron 86:1253–1264. doi:10.1016/j.neuron.2015.05.001
Gibbs RB, Johnson DA (2007) Cholinergic lesions produce task-selective effects on delayed matching to position and configural association learning related to response pattern and strategy. Neurobiol Learn Mem 88:19–32. doi:10.1016/j.nlm.2007.03.007
Goto M, Swanson LW, Canteras NS (2001) Connections of the nucleus incertus. J Comp Neurol 438:86–122
Gritti I, Manns ID, Mainville L, Jones BE (2003) Parvalbumin, calbindin, or calretinin in cortically projecting and GABAergic, cholinergic, or glutamatergic basal forebrain neurons of the rat. J Comp Neurol 458:11–31. doi:10.1002/cne.10505
Hangya B, Borhegyi Z, Szilagyi N et al (2009) GABAergic neurons of the medial septum lead the hippocampal network during theta activity. J Neurosci 29:8094–8102. doi:10.1523/JNEUROSCI.5665-08.2009
Hasselmo ME (2005) What is the function of hippocampal theta rhythm? Linking behavioral data to phasic properties of field potential and unit recording data. Hippocampus 15:936–949. doi:10.1002/hipo.20116
Hepler D, Olton D, Wenk G, Coyle J (1985) Lesions in nucleus basalis magnocellularis and medial septal area of rats produce qualitatively similar memory impairments. J Neurosci 5:866–873
Hosken IT, Sutton SW, Smith CM, Gundlach AL (2015) Relaxin-3 receptor (Rxfp3) gene knockout mice display reduced running wheel activity: implications for role of relaxin-3/RXFP3 signalling in sustained arousal. Behav Brain Res 278:167–175. doi:10.1016/j.bbr.2014.09.028
Huh CYL, Goutagny R, Williams S (2010) Glutamatergic neurons of the mouse medial septum and diagonal band of Broca synaptically drive hippocampal pyramidal cells: relevance for hippocampal theta rhythm. J Neurosci 30:15951–15961. doi:10.1523/JNEUROSCI.3663-10.2010
Impey S, Obrietan K, Storm DR (1999) Making new connections: role of ERK/MAP kinase signaling in neuronal plasticity. Neuron 23:11–14. doi:10.1016/S0896-6273(00)80747-3
Johnson DA, Zambon NJ, Gibbs RB (2002) Selective lesion of cholinergic neurons in the medial septum by 192 IgG-saporin impairs learning in a delayed matching to position T-maze paradigm. Brain Res 943:132–141
Kania A, Gundlach AL, Lewandowski MH, Blasiak A (2014) Activation of relaxin-3 receptors (RXFP3) on hypothalamic paraventricular nucleus (PVN) neurons in the rat in vitro. FENS 9th Forum Neurosci Abstr P2706
Kelsey JE, Vargas H (1993) Medial septal lesions disrupt spatial, but not nonspatial, working memory in rats. Behav Neurosci 107:565–574
Kemp IR, Kaada BR (1975) The relation of hippocampal theta activity to arousal, attentive behaviour and somato-motor movements in unrestrained cats. Brain Res 95:323–342. doi:10.1016/0006-8993(75)90110-9
Kirk IJ (1998) Frequency modulation of hippocampal theta by the supramammillary nucleus, and other hypothalamo-hippocampal interactions: mechanisms and functional implications. Neurosci Biobehav Rev 22:291–302
Kitabatake Y, Hikida T, Watanabe D et al (2003) Impairment of reward-related learning by cholinergic cell ablation in the striatum. Proc Natl Acad Sci USA 100:7965–7970. doi:10.1073/pnas.1032899100
Kocan M, Sarwar M, Hossain MA et al (2014) Signalling profiles of H3 relaxin, H2 relaxin and R3(BΔ23-27)R/I5 acting at the relaxin family peptide receptor 3 (RXFP3). Br J Pharmacol 171:2827–2841. doi:10.1111/bph.12623
Lee SH, Dan Y (2012) Neuromodulation of brain states. Neuron 76:109–222. doi:10.1016/j.neuron.2012.09.012
Leranth C, Kiss J (1996) A population of supramammillary area calretinin neurons terminating on medial septal area cholinergic and lateral septal area calbindin-containing cells are aspartate/glutamatergic. J Neurosci 16:7699–7710
Leung LS, Yim CY (1986) Intracellular records of theta rhythm in hippocampal CA1 cells of the rat. Brain Res 367:323–327. doi:10.1016/0006-8993(86)91611-2
Liu C, Eriste E, Sutton S et al (2003) Identification of relaxin-3/INSL7 as an endogenous ligand for the orphan G-protein-coupled receptor GPCR135. J Biol Chem 278:50754–50764. doi:10.1074/jbc.M308995200
Liu C, Chen J, Kuei C et al (2005) Relaxin-3/insulin-like peptide 5 chimeric peptide, a selective ligand for G protein-coupled receptor (GPCR)135 and GPCR142 over leucine-rich repeat-containing G protein-coupled receptor 7. Mol Pharmacol 67:231–240. doi:10.1124/mol.104.006700
Ma S, Bonaventure P, Ferraro T et al (2007) Relaxin-3 in GABA projection neurons of nucleus incertus suggests widespread influence on forebrain circuits via G-protein-coupled receptor-135 in the rat. Neuroscience 144:165–190. doi:10.1016/j.neuroscience.2006.08.072
Ma S, Olucha-Bordonau FE, Hossain MA et al (2009) Modulation of hippocampal theta oscillations and spatial memory by relaxin-3 neurons of the nucleus incertus. Learn Mem 16:730–742. doi:10.1101/lm.1438109
Mamad O, McNamara HM, Reilly RB, Tsanov M (2015) Medial septum regulates the hippocampal spatial representation. Front Behav Neurosci 9:166. doi:10.3389/fnbeh.2015.00166
Mattson MP, Camandola S (2001) NF-kappaB in neuronal plasticity and neurodegenerative disorders. J Clin Invest 107:247–254. doi:10.1172/JCI11916
McGowan BM, Stanley SA, Smith KL et al (2005) Central relaxin-3 administration causes hyperphagia in male Wistar rats. Endocrinology 146:3295–3300. doi:10.1210/en.2004-1532
McGowan BM, Stanley SA, White NE et al (2007) Hypothalamic mapping of orexigenic action and Fos-like immunoreactivity following relaxin-3 administration in male Wistar rats. Am J Physiol Metab 292:E913–E919. doi:10.1152/ajpendo.00346.2006
Morales FR, Roig JA, Monti JM et al (1971) Septal unit activity and hippocampal EEG during the sleep-wakefulness cycle of the rat. Physiol Behav 6:563–567. doi:10.1016/0031-9384(71)90206-X
Mysin IE, Kitchigina VF, Kazanovich Y (2015) Modeling synchronous theta activity in the medial septum: key role of local communications between different cell populations. J Comput Neurosci 39:1–16. doi:10.1007/s10827-015-0564-6
Nerad L, McNaughton N (2006) The septal EEG suggests a distributed organization of the pacemaker of hippocampal theta in the rat. Eur J Neurosci 24:155–166. doi:10.1111/j.1460-9568.2006.04902.x
Nunez A, Cervera-Ferri A, Olucha-Bordonau F et al (2006) Nucleus incertus contribution to hippocampal theta rhythm generation. Eur J Neurosci 23:2731–2738. doi:10.1111/j.1460-9568.2006.04797.x
O’Keefe J (1976) Place units in the hippocampus of the freely moving rat. Exp Neurol 51:78–109. doi:10.1016/0014-4886(76)90055-8
Olucha-Bordonau FE, Teruel V, Barcia-Gonzalez J et al (2003) Cytoarchitecture and efferent projections of the nucleus incertus of the rat. J Comp Neurol 464:62–97. doi:10.1002/cne.10774
Olucha-Bordonau FE, Otero-Garcia M, Sanchez-Perez AM et al (2012) Distribution and targets of the relaxin-3 innervation of the septal area in the rat. J Comp Neurol 520:1903–1939. doi:10.1002/cne.23018
Otsubo H, Onaka T, Suzuki H et al (2010) Centrally administered relaxin-3 induces Fos expression in the osmosensitive areas in rat brain and facilitates water intake. Peptides 31:1124–1130. doi:10.1016/j.peptides.2010.02.020
Pan WX, McNaughton N (1997) The medial supramammillary nucleus, spatial learning and the frequency of hippocampal theta activity. Brain Res 764:101–108
Paxinos G, Watson C (2014) The rat brain in stereotaxic coordinates. Academic Press, San Diego, CA, USA
Raghavachari S, Kahana MJ, Rizzuto DS et al (2001) Gating of human theta oscillations by a working memory task. J Neurosci 21:3175–3183
Ragozzino ME, Unick KE, Gold PE (1996) Hippocampal acetylcholine release during memory testing in rats: augmentation by glucose. Proc Natl Acad Sci USA 93:4693–4698
Rajkumar R, Wu Y, Farooq U et al (2016) Stress activates the nucleus incertus and modulates plasticity in the hippocampo-medial prefrontal cortical pathway. Brain Res Bull 120:83–89. doi:10.1016/j.brainresbull.2015.10.010
Ramanan N, Shen Y, Sarsfield S et al (2005) SRF mediates activity-induced gene expression and synaptic plasticity but not neuronal viability. Nat Neurosci 8:759–767. doi:10.1038/nn1462
Robinson J, Manseau F, Ducharme G, Amilhon B, Vigneault E, El Mestikawy S, Williams S (2016) Optogenetic activation of septal glutamatergic neurons drive hippocampal theta rhythms. J Neurosci 36:3016–3023
Roland JJ, Stewart AL, Janke KL et al (2014) Medial septum-diagonal band of Broca (MSDB) GABAergic regulation of hippocampal acetylcholine efflux is dependent on cognitive demands. J Neurosci 34:506–514. doi:10.1523/JNEUROSCI.2352-13.2014
Ryan PJ, Ma S, Olucha-Bordonau FE, Gundlach AL (2011) Nucleus incertus—an emerging modulatory role in arousal, stress and memory. Neurosci Biobehav Rev 35:1326–1341. doi:10.1016/j.neubiorev.2011.02.004
Ryan PJ, Buchler E, Shabanpoor F et al (2013) Central relaxin-3 receptor (RXFP3) activation decreases anxiety- and depressive-like behaviours in the rat. Behav Brain Res 244:142–151. doi:10.1016/j.bbr.2013.01.034
Sánchez-Pérez AM, Arnal-Vicente I, Santos FN et al (2015) Septal projections to the nucleus incertus in the rat: bidirectional pathways for modulation of hippocampal function. J Comp Neurol 523:565–588. doi:10.1002/cne.23687
Shabanpoor F, Akhter Hossain M, Ryan PJ et al (2012) Minimization of human relaxin-3 leading to high-affinity analogues with increased selectivity for relaxin-family peptide 3 receptor (RXFP3) over RXFP1. J Med Chem 55:1671–1681. doi:10.1021/jm201505p
Smith CM, Shen PJ, Banerjee A et al (2010) Distribution of relaxin-3 and RXFP3 within arousal, stress, affective, and cognitive circuits of mouse brain. J Comp Neurol 518:4016–4045. doi:10.1002/cne.22442
Smith CM, Hosken IT, Sutton SW et al (2012) Relaxin-3 null mutation mice display a circadian hypoactivity phenotype. Genes Brain Behav 11:94–104. doi:10.1111/j.1601-183X.2011.00730.x
Sotty F, Danik M, Manseau F et al (2003) Distinct electrophysiological properties of glutamatergic, cholinergic and GABAergic rat septohippocampal neurons: novel implications for hippocampal rhythmicity. J Physiol 551:927–943. doi:10.1113/jphysiol.2003.046847
Stumpf C, Pettsche H, Gogolak G (1962) The significance of the rabbit’s septum as a relay station between the midbrain and the hippocampus. II. The differential influence of drugs upon both the septal cell firing pattern and the hippocampus theta activity. Electroencephalogr Clin Neurophysiol 14:212–219
Sutton SW, Bonaventure P, Kuei C et al (2004) Distribution of G-protein-coupled receptor (GPCR)135 binding sites and receptor mRNA in the rat brain suggests a role for relaxin-3 in neuroendocrine and sensory processing. Neuroendocrinology 80:298–307. doi:10.1159/000083656
Sweatt JD (2001) The neuronal MAP kinase cascade: a biochemical signal integration system subserving synaptic plasticity and memory. J Neurochem 76:1–10
Tanaka M, Iijima N, Miyamoto Y et al (2005) Neurons expressing relaxin 3/INSL 7 in the nucleus incertus respond to stress. Eur J Neurosci 21:1659–1670. doi:10.1111/j.1460-9568.2005.03980.x
Thomas GJ, Gash DM (1986) Differential effects of posterior septal lesions on dispositional and representational memory. Behav Neurosci 100:712–719. doi:10.1037/h0090392
Toth K, Freund TF, Miles R (1997) Disinhibition of rat hippocampal pyramidal cells by GABAergic afferents from the septum. J Physiol 500(Pt 2):463–474
Tsanov M (2015) Septo-hippocampal signal processing: breaking the code. Prog Brain Res 219:103–120. doi:10.1016/bs.pbr.2015.04.002
Ujfalussy B, Kiss T (2006) How do glutamatergic and GABAergic cells contribute to synchronization in the medial septum? J Comput Neurosci 21:343–357. doi:10.1007/s10827-006-9082-x
Van der Westhuizen ET, Sexton PM, Bathgate RA, Summers RJ (2005) Responses of GPCR135 to human gene 3 (H3) relaxin in CHO-K1 cells determined by microphysiometry. Ann N Y Acad Sci 1041:332–337. doi:10.1196/annals.1282.053
van der Westhuizen ET, Werry TD, Sexton PM, Summers RJ (2007) The relaxin family peptide receptor 3 activates extracellular signal-regulated kinase 1/2 through a protein kinase C-dependent mechanism. Mol Pharmacol 71:1618–1629. doi:10.1124/mol.106.032763
van der Westhuizen ET, Christopoulos A, Sexton PM et al (2010) H2 relaxin is a biased ligand relative to H3 relaxin at the relaxin family peptide receptor 3 (RXFP3). Mol Pharmacol 77:759–772. doi:10.1124/mol.109.061432
Vandecasteele M, Varga V, Berenyi A et al (2014) Optogenetic activation of septal cholinergic neurons suppresses sharp wave ripples and enhances theta oscillations in the hippocampus. Proc Natl Acad Sci 111:13535–13540. doi:10.1073/pnas.1411233111
Vertes RP, Kocsis B (1997) Brainstem-diencephalo-septohippocampal system controlling the theta rhythm of the hippocampus. Neuroscience 81:893–926
Wei B, Huang Z, He S et al (2012) The onion skin-like organization of the septum arises from multiple embryonic origins to form multiple adult neuronal fates. Neuroscience 222:110–123. doi:10.1016/j.neuroscience.2012.07.016
Wu M, Shanabrough M, Leranth C, Alreja M (2000) Cholinergic excitation of septohippocampal GABA but not cholinergic neurons: implications for learning and memory. J Neurosci 20:3900–3908
Yoder RM, Pang KC (2005) Involvement of GABAergic and cholinergic medial septal neurons in hippocampal theta rhythm. Hippocampus 15:381–392. doi:10.1002/hipo.20062
Zhang H, Lin SC, Nicolelis MA (2011) A distinctive subpopulation of medial septal slow-firing neurons promote hippocampal activation and theta oscillations. J Neurophysiol 106:2749–2763. doi:10.1152/jn.00267.2011
Acknowledgments
This research was supported by a predoctoral fellowship (FPI-UJI: PREDOC/2014/35) to HAG; a traineeship fellowship (UJI P1·1A2014-06) to AGA; the FP7-PEOPLE-IRSES PIRSES-GA-2012-318997 NEUREN project to ALG and FEO-B; NHMRC (Australia) project grants (1027522, 1026939) and a Brain and Behavior Research Foundation (USA) NARSAD Independent Investigator Award to ALG; Generalitat Valenciana (AICO/2015/042) project grant and Universitat Jaume I (P1·1A2014-06) project grant to AMS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Francisco E. Olucha-Bordonau and Ana M. Sánchez-Pérez contributed equally to this work.
Rights and permissions
About this article
Cite this article
Albert-Gascó, H., García-Avilés, Á., Moustafa, S. et al. Central relaxin-3 receptor (RXFP3) activation increases ERK phosphorylation in septal cholinergic neurons and impairs spatial working memory. Brain Struct Funct 222, 449–463 (2017). https://doi.org/10.1007/s00429-016-1227-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-016-1227-8