Abstract
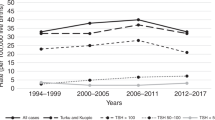
Little is known about the global prevalence of congenital hypothyroidism (CH), though it is known to vary across countries and time periods. This meta-analysis aims to estimate the global and regional prevalence of CH among births between 1969 and 2020. PubMed, Web of Sciences, and Embase databases were searched for relevant studies between January 1, 1975, and March 2, 2020. Pooled prevalence was calculated using a generalized linear mixed model, and expressed as a rate per 10,000 neonates. The meta-analysis involved 116 studies, which analyzed 330,210,785 neonates, among whom 174,543 were diagnosed with CH. The pooled global prevalence of CH from 1969 to 2020 was 4.25 (95% confidence interval (CI) 3.96–4.57). The geographic region with highest prevalence was the Eastern Mediterranean (7.91, 95% CI 6.09–10.26), where the prevalence was 2.48-fold (95% CI 2.04–3.01) that in Europe. The national income level with the highest prevalence was upper-middle (6.76, 95% CI 5.66–8.06), which was 1.91-fold (95% CI 1.65–2.22) that in high-income countries. Global prevalence of CH was 52% (95% CI 4–122%) higher in 2011–2020 than in 1969–1980, after adjusting for geographic region, national income level, and screening strategy.
Conclusion: The global prevalence of CH increased from 1969 to 2020, which may reflect the implementation of national neonatal screening, neonatal testing for thyroid-stimulating hormone, and a lowering of the diagnostic level of this hormone. Additional factors are likely to be driving the increase, which should be identified in future research.
What is Known: • Cumulated evidence had suggested that the occurrences of congenital hypothyroidism (CH) among newborns were varied in different countries.. • Up-trends of the birth prevalence of CH were observed in many European and American countries. | |
What is New: • This is the first meta-analysis to estimate global and regional prevalence of CH among newborns. • The global prevalence of CH has increased by 127% since 1969. The Eastern Mediterranean has the highest prevalence and stands out with the most pronounced escalation in the prevalence of CH. |


Similar content being viewed by others
Availability of data and code
Available upon request from the co-corresponding authors.
Data availability
The data are available from the corresponding author on reasonable request.
Abbreviations
- CH:
-
Congenital hypothyroidism
- DHG:
-
Dyshormonogenesis
- HTT:
-
Hyperthyrotropinemia
- NBS:
-
Newborn screening
- PCH:
-
Permanent congenital hypothyroidism
- TCH :
-
Transient congenital hypothyroidism
- TSH :
-
Thyroid-stimulating hormone
- T4 :
-
Thyroxine
References
Agrawal P, Philip R, Saran S, Gutch M, Razi MS, Agroiya P, Gupta K (2015) Congenital hypothyroidism. Indian J. Endocrinol Metab 19:221–227
Dimitropoulos A, Molinari L, Etter K, Torresani T, Lang-Muritano M, Jenni OG, Largo RH, Latal B (2009) Children with congenital hypothyroidism: long-term intellectual outcome after early high-dose treatment. Pediatr Res 65:242–248
Grosse SD, Van Vliet G (2011) Prevention of intellectual disability through screening for congenital hypothyroidism: how much and at what level? Arch Dis Child 96:374-U133
Salerno M, Militerni R, Bravaccio C, Micillo M, Capalbo D, Di Maio S, Tenore A (2002) Effect of different starting doses of levothyroxine on growth and intellectual outcome at four years of age in congenital hypothyroidism. Thyroid 12:45–52
Salerno M, Militerni R, Di Maio S, Bravaccio C, Gasparini N, Tenore A (1999) Intellectual outcome at 12 years of age in congenital hypothyroidism. Eur J Endocrinol 141:105–110
Rastogi MV, LaFranchi SH (2010) Congenital hypothyroidism. Orphanet J Rare Dis 5:17
Padilla CD, Therrell BL (2007) Newborn screening in the Asia Pacific region. J Inherit Metab Dis 30:490–506
Rose SR, Blunden CE, Jarrett OO, Kaplan K, Caravantes R, Akinbi HT (2022) Utility of repeat testing for congenital hypothyroidism in infants with very low birth weight. J Pediatr 242:152-158.e151
Centers for Disease Control and Prevention (CDC) (2008) Update on overall prevalence of major birth defects--Atlanta, Georgia, 1978-2005. MMWR Morb Mortal Wkly Rep 57:1-5
McGrath N, Hawkes CP, McDonnell CM, Cody D, O'Connell SM, Mayne PD, Murphy NP (2018) Incidence of congenital hypothyroidism over 37 years in Ireland. Pediatrics 142
Barry Y, Bonaldi C, Goulet V, Coutant R, Léger J, Paty AC, Delmas D, Cheillan D, Roussey M (2016) Increased incidence of congenital hypothyroidism in France from 1982 to 2012: a nationwide multicenter analysis. Ann Epidemiol 26:100-105.e104
Pearce MS, Korada M, Day J, Turner S, Allison D, Kibirige M, Cheetham TD (2010) Increasing incidence, but lack of seasonality, of elevated TSH levels, on newborn screening, in the North of England. J Thyroid Res 2010:101948–101948
Yarahmadi S, Azhang N, Nikkhoo B, Rahmani K (2020) A success story: review of the implementation and achievements of the National Newborn Screening Program for congenital hypothyroidism in Iran. Int J Endocrinol Metabol 18
Olivieri A, Corbetta C, Weber G, Vigone MC, Fazzini C, Medda E (2013) Congenital hypothyroidism due to defects of thyroid development and mild increase of TSH at screening: data from the Italian National Registry of infants with congenital hypothyroidism. J Clin Endocrinol Metab 98:1403–1408
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ (Clinical research ed) 339
Hoy D, Brooks P, Woolf A, Blyth F, March L, Bain C, Baker P, Smith E, Buchbinder R (2012) Assessing risk of bias in prevalence studies: modification of an existing tool and evidence of interrater agreement. J Clin Epidemiol 65:934–939
Noubiap JJ, Nansseu JR, Nyaga UF, Sime PS, Francis I, Bigna JJ (2019) Global prevalence of resistant hypertension: a meta-analysis of data from 3.2 million patients. Heart 105:98–105
Al Amer HS (2020) Low back pain prevalence and risk factors among health workers in Saudi Arabia: a systematic review and meta-analysis. J Occupat Health 62
Barican JL, Yung D, Schwartz C, Zheng Y, Georgiades K, Waddell C (2022) Prevalence of childhood mental disorders in high-income countries: a systematic review and meta-analysis to inform policymaking. Evid Based Ment Health 25:36–44
Barker MM, Beresford B, Bland M, Fraser LK (2019) Prevalence and incidence of anxiety and depression among children, adolescents, and young adults with life-limiting conditions: a systematic review and meta-analysis. JAMA Pediatr 173:835–844
Balduzzi S, Ruecker G, Schwarzer G (2019) How to perform a meta-analysis with R: a practical tutorial. Evid Based Ment Health 22:153–160
Viechtbauer W (2010) Conducting meta-analyses in R with the metafor Package. J Stat Softw 36:1–48
Wassner AJ (2018) Congenital hypothyroidism. Clin Perinatol 45:1–18
Albert BB, Cutfield WS, Webster D, Carll J, Derraik JG, Jefferies C, Gunn AJ, Hofman PL (2012) Etiology of increasing incidence of congenital hypothyroidism in New Zealand from 1993–2010. J Clin Endocrinol Metab 97:3155–3160
Delgado JA, Bauça JM, Pérez Esteban G, Caimari Jaume M, Robles Bauza J (2021) Challenges in screening for congenital hypothyroidism: optimization of thyrotropin cut-off values. Clin Chim Acta 512:20–25
Kumorowicz-Czoch M, Tylek-Lemanska D, Starzyk J (2011) Thyroid dysfunctions in children detected in mass screening for congenital hypothyroidism. J Pediatr Endocrinol Metab 24:141–145
Tucker D, Woods G, Langham S, Biassoni L, Krywawych S, Hindmarsh P, Peters C (2016) The incidence and clinical features of dual thyroid ectopia in congenital hypothyroidism. J Clin Endocrinol Metab 101:2063–2068
Fisher DA, Dussault JH, Foley TP, Klein AH, LaFranchi S, Larsen PR, Mitchell ML, Murphey WH, Walfish PG (1979) Screening for congenital hypothyroidism: results of screening one million North American infants. J Pediatr 94:700–705
Schwarzer G, Chemaitelly H, Abu-Raddad LJ, Rücker G (2019) Seriously misleading results using inverse of Freeman-Tukey double arcsine transformation in meta-analysis of single proportions. Res Synth Methods 10:476–483
Lin L, Chu H (2020) Meta-analysis of proportions using generalized linear mixed models. Epidemiology 31:713–717
Liu Y, Chen S, Zühlke L, Black GC, Choy MK, Li N, Keavney BD (2019) Global birth prevalence of congenital heart defects 1970–2017: updated systematic review and meta-analysis of 260 studies. Int J Epidemiol 48:455–463
Wikipedia (2022) List of WHO regions
Bank TW (2022) World Bank Country and Lending Groups
Ekundayo TC, Igere BE, Iwu CD, Oluwafemi YD, Tiamiyu AM, Adesina IA, Anuoluwa IA, Ekundayo EA, Bello OO, Olaniyi OO, Ijabadeniyi OA (2022) Prevalence of Laribacter hongkongensis in food and environmental matrices: a systematic review and meta-analysis. Food Microbiol 107
Hunter JP, Saratzis A, Sutton AJ, Boucher RH, Sayers RD, Bown MJ (2014) In meta-analyses of proportion studies, funnel plots were found to be an inaccurate method of assessing publication bias. J Clin Epidemiol 67:897–903
Alm J, Larsson A, Zetterstrom R (1978) Congenital hypothyroidism in Sweden. Incidence and age at diagnosis. Acta Paediatr Scand 67:1–3
Fisher DA (1983) Second International Conference on Neonatal Thyroid Screening: progress report. J Pediatr 102:653–654
Toublanc JE (1992) Comparison of epidemiological data on congenital hypothyroidism in Europe with those of other parts in the world. Horm Res 38:230–235
Ford G, LaFranchi SH (2014) Screening for congenital hypothyroidism: a worldwide view of strategies. Best Pract Res Clin Endocrinol Metab 28:175–187
LaFranchi SH (2010) Newborn screening strategies for congenital hypothyroidism: an update. J Inherit Metab Dis 33:S225-233
Working Group on Neonatal Screening of the European Society for Paediatric Endocrinology (1999) Revised guidelines for neonatal screening programmes for primary congenital hypothyroidism. Horm Res 52:49–52
Mengreli C, Kanaka-Gantenbein C, Girginoudis P, Magiakou MA, Christakopoulou I, Giannoulia-Karantana A, Chrousos GP, Dacou-Voutetakis C (2010) Screening for congenital hypothyroidism: the significance of threshold limit in false-negative results. J Clin Endocrinol Metab 95:4283–4290
Corbetta C, Weber G, Cortinovis F, Calebiro D, Passoni A, Vigone MC, Beck-Peccoz P, Chiumello G, Persani L (2009) A 7-year experience with low blood TSH cutoff levels for neonatal screening reveals an unsuspected frequency of congenital hypothyroidism (CH). Clin Endocrinol 71:739–745
Deladoëy J, Ruel J, Giguère Y, Van Vliet G (2011) Is the incidence of congenital hypothyroidism really increasing? A 20-year retrospective population-based study in Québec. J Clin Endocrinol Metab 96:2422–2429
Hashemipour M, Kelishadi R, Amin MM, Poursafa P, Rashidi M, Mehrnejat N, Hovsepian S (2020) The association between familial and environmental factors and prevalence of congenital hypothyroidism in center of Iran. Environmental science and pollution research international 15
Jiang X, Huang YL, Feng Y, Tang F, Jia XF, Chen QY, Tang CF, Liu SC, Li B, Zheng RD, Liu JL (2020) Same-sex twins have a high incidence of congenital hypothyroidism and a high probability to be missed at newborn screening. Clin Chim Acta 502:111–115
Medda E, Vigone MC, Cassio A, Calaciura F, Costa P, Weber G, de Filippis T, Gelmini G, Di Frenna M, Caiulo S, Ortolano R, Rotondi D, Bartolucci M, Gelsomino R, De Angelis S, Gabbianelli M, Persani L, Olivieri A (2019) Neonatal screening for congenital hypothyroidism: what can we learn from discordant twins? J Clin Endocrinol Metab 104:5765–5779
LaFranchi SH (2021) Thyroid function in preterm/low birth weight infants: impact on diagnosis and management of thyroid dysfunction. Front Endocrinol (Lausanne) 12
Alavi ER, Rafiei N, Rafiei R, Farokhi E (2021) Prevalence of transient congenital hypothyroidism among neonates. Ann Med Surg (Lond) 72
Kiran Z, Sheikh A, Humayun KN, Islam N (2021) Neonatal outcomes and congenital anomalies in pregnancies affected by hypothyroidism. Ann Med 53:1560–1568
Rose SR, Brown RS, Foley T, Kaplowitz PB, Kaye CI, Sundararajan S, Varma SK et al (2006) Update of newborn screening and therapy for congenital hypothyroidism. Pediatrics 117:2290–2303
Rezaeian S, Poorolajal J, Moghimbegi A, Esmailnasab N (2013) Risk factors of congenital hypothyroidism using propensity score: a matched case-control study. J Res Health Sci 13:151–156
Kollati Y, Ambati RR, Reddy PN, Kumar NSS, Patel RK, Dirisala VR (2017) Congenital hypothyroidism: facts, facets & therapy. Curr Pharm Des 23:2308–2313
Alfadhel M, Al Othaim A, Al Saif S, Al Mutairi F, Alsayed M, Rahbeeni Z, Alzaidan H et al (2017) Expanded Newborn Screening Program in Saudi Arabia: incidence of screened disorders. J Paediatr Child Health 53:585–591
Ahmad A, Wasim A, Hussain S, Saeed M, Ahmad BM, Rehman KU (2017) Congenital hypothyroidism in neonates of a tertiary care hospital. Pak J Med Sci 33:1269–1272
Golbahar J, Al-Khayyat H, Hassan B, Agab W, Hassan E, Darwish A (2010) Neonatal screening for congenital hypothyroidism: a retrospective hospital based study from Bahrain. J Pediatr Endocrinol Metab 23:39–44
Haque KN (1993) Pediatric inpatients at the King Khalid University Hospital, Riyadh, Saudi Arabia, 1985G–1989G. Ann Saudi Med 13:570
Hamamy H, Alwan A (1994) Hereditary disorders in the Eastern Mediterranean Region. Bull World Health Organ 72:145–154
Hinton CF, Harris KB, Borgfeld L, Drummond-Borg M, Eaton R, Lorey F, Therrell BL, Wallace J, Pass KA (2010) Trends in incidence rates of congenital hypothyroidism related to select demographic factors: data from the United States, California, Massachusetts, New York, and Texas. Pediatrics 125(Suppl 2):S37-47
Al-Hosani H, Osman H, Abdel Wareth L, Saade D, Salah M (2003) Prevalence of iodine deficiency disorders in the United Arab Emirates measured by raised TSH levels. East Mediterr Health J 9:123–130
World Health Organization, International Council for Control of Iodine Deficiency Disorders, United Nations Children's Fund (UNICEF) (1994) Indicators for assessing iodine deficiency disorders and their control through salt iodization. https://apps.who.int/iris/handle/10665/70715
Mohammadi M, Azizi F, Hedayati M (2018) Iodine deficiency status in the WHO Eastern Mediterranean Region: a systematic review. Environ Geochem Health 40:87–97
Patriota ESO, Lima ICC, Nilson EAF, Franceschini SCC, Gonçalves VSS, Pizato N (2022) Prevalence of insufficient iodine intake in pregnancy worldwide: a systematic review and meta-analysis. Eur J Clin Nutr 76:703–715
Navarro-Zambrana AN, Sheets LR (2022) Ethnic and national differences in congenital adrenal hyperplasia incidence: a systematic review and meta-analysis. Horm Res Paediatr
Shoraka HR, Haghdoost AA, Baneshi MR, Bagherinezhad Z, Zolala F (2020) Global prevalence of classic phenylketonuria based on Neonatal Screening Program Data: systematic review and meta-analysis. Clin Exp Pediatr 63:34–43
Therrell BL Jr, Lloyd-Puryear MA, Ohene-Frempong K, Ware RE, Padilla CD, Ambrose EE, Barkat A, Ghazal H, Kiyaga C, Mvalo T, Nnodu O, Ouldim K, Rahimy MC, Santos B, Tshilolo L, Yusuf C, Zarbalian G, Watson MS, faculty, speakers at the First Pan African Workshop on Newborn Screening RMJ (2020) Empowering newborn screening programs in African countries through establishment of an international collaborative effort. J Community Genet 11:253–268
Author information
Authors and Affiliations
Contributions
Xiaohong Li, Yongna Yao, Lei Liu, Wenchong He, Jun Zhu, Kui Deng, Huiwen Tan, Liangcheng Xiang, Xuelian Yuan, and Yingkun Guo designed the project. Jun Zhu, Kui Deng, Xuelian Yuan, Liangcheng Xiang, Huiwen Tan, Yingkun Guo, and Menglan Huang contributed to the systematic search. Lei Liu, Menglan Huang, Liangcheng Xiang, Qi Li, and Liangcheng Xiang contributed to screening. Lei Liu, Yongna Yao, and Xiaohong Li contributed to data collection. Wenchong He, Kui Deng, Liangcheng Xiang, Qi Li, and Yingkun Guo contributed to data extraction. Lei Liu, Yongna Yao, Qi Li, and Xuelian Yuan contributed to data analysis. Xiaohong Li and Wenchong He contributed to data interpretation. Yongna Yao, Lei Liu, Wenchong He, and Xiaohong Li prepared the manuscript. All the authors revised and approved the manuscript.
Corresponding authors
Ethics declarations
Ethical approval
Not required in meta-analysis.
Consent to participate
Not required in meta-analysis.
Consent for publication
Not required in meta-analysis.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Gregorio Milani
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, L., He, W., Zhu, J. et al. Global prevalence of congenital hypothyroidism among neonates from 1969 to 2020: a systematic review and meta-analysis. Eur J Pediatr 182, 2957–2965 (2023). https://doi.org/10.1007/s00431-023-04932-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-023-04932-2