Abstract
Purpose
Renal cell carcinoma (RCC) is the most common renal tumor and accounts for nearly 3 % of adult cancers. In the recent years, seven new targeted agents have been approved changing the treatment in metastatic RCC dramatically. So far, however, it remains unclear which sequence is best for those patients. This study analyzed retrospectively the outcome of patients treated with everolimus after failure of a vascular endothelial growth factor receptor-directed therapy and which therapies were used after everolimus.
Patients and methods
In a retrospective analysis, patients receiving everolimus after failure of first-line VEGFR-directed therapy have been analyzed in regard to response, duration of treatment and subsequent therapies. In total, the data of 81 patients have been analyzed.
Results
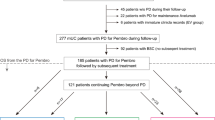
The most observed first-line therapy was sunitinib followed by sorafenib. Thirty-two patients received everolimus as second-line therapy, and 49 as third-line therapy. The median duration of treatment with everolimus was 4.5 months. Seventy-seven of eighty-one patients (95 %) received a further therapy after discontinuation of everolimus. The agents administered beyond were sunitinib (28.6 %), sorafenib (28.6 %) and 42.8 % received other therapies. Twenty-seven patients received an additional sequence of therapy (fourth to fifth line). Fifty-eight percentage of patients have still been alive at time of analysis.
Conclusion
The duration of everolimus therapy beyond failure of anti-VEGF-directed therapy and the reported time to progression was in the range of the RECORD-1 trial in daily practice as well. After failure of everolimus, reexposure to tyrosine kinase inhibitors is a common clinical practice and demonstrates a clinical benefit of therapies beyond everolimus.

Similar content being viewed by others
References
Bergmann L, Goebell PJ, Kube U et al (2013) Everolimus in metastatic renal cell carcinoma after failure of initial vascular endothelial growth factor receptor-tyrosine kinase inhibitor (VEGFr-TKI) therapy: results of an interim analysis of a non-interventional study. Onkologie 36(3):95–100
Bergmann L, Beck J, Bothe K et al (2014) Treatment algorithm for metastatic renal cell carcinoma. Recommendations based on evidence and clinical practice. Oncol Res Treat (in press)
Bergmann L, Luise Maute L, Guschmann L (2014b) Drug profile of temsirolimus for advanced renal cell carcinoma. Expert Rev Anticancer Ther 14(1):9–21
Beuvink I, Boulay A, Fumagalli S et al (2005) The mTOR inhibitor RAD001 sensitizes tumor cells to DNA-damaged induced apoptosis through inhibition of p21 translation. Cell 120(6):747–759
Bukowski RM (2012) Temsirolimus: a safety and efficacy review. Expert Opin Drug Saf 11(5):861–879
Cho I, Chung J (2012) Current status of targeted therapy for advanced renal cell carcinoma. Korean J Urol 53:217–228
Chow WH, Devesa SS, Warren JL, Fraumeni JF Jr (1999) Rising incidence of renal cell cancer in the United States. JAMA 281(17):1628–1631
Del Bufalo D, Ciuffreda L, Trisciuoglio D et al (2006) Antiangiogenic potential of the Mammalian target of rapamycin inhibitor temsirolimus. Cancer Res 66(11):5549–5554
Dutcher JP, de Souza P, McDermott D et al (2009) Effect of temsirolimus versus interferon-alpha on outcome of patients with advanced renal cell carcinoma of different tumor histologies. Med Oncol 26(2):202–209
Escudier B, Albiges L, Sonpavde G (2013) Optimal management of metastatic renal cell carcinoma: current status. Drugs 73(5):427–438
Faivre S, Kroemer G, Raymond E (2006) Current development of mTOR inhibitors as anticancer agents. Nat Rev Drug Discov 5(8):671–688
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674
Hollingsworth JM, Miller DC, Daignault S, Hollenbeck BK (2006) Rising incidence of small renal masses: a need to reassess treatment effect. J Natl Cancer Inst 98(18):1331–1334
Hutson TE, Escudier B, Esteban E et al (2013) Randomized phase III trial of temsirolimus versus sorafenib as second-line therapy after sunitinib in patients with metastatic renal cell carcinoma. J Clin Oncol [Epub ahead of print]
Janzen NK, Kim HL, Figlin RA, Belldegrun AS (2003) Surveillance after radical or partial nephrectomy for localized renal cell carcinoma and management of recurrent disease. Urol Clin North Am 30(4):843–852
Levine B, Kroemer G (2008) Autophagy in the pathogenesis of disease. Cell 132(1):27–42
Mathew A, Devesa SS, Fraumeni JF Jr, Chow WH (2002) Global increases in kidney cancer incidence, 1973–1992. Eur J Cancer Prev 11(2):171–178
Motzer RJ, Escudier B, Oudard S et al (2008) Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet 372(9637):449–456
Motzer RJ, Escudier B, Oudard S et al (2010) Phase 3 trial of everolimus for metastatic renal cell carcinoma : final results and analysis of prognostic factors. Cancer 116(18):4256–4265
Motzer RJ, Barrios CH, Kim TM et al (2013) Record-3: phase II randomized trial comparing sequential first-line everolimus (EVE) and second-line sunitinib (SUN) versus first-line SUN and second-line EVE in patients with metastatic renal cell carcinoma (mRCC). J Clin Oncol 31(Suppl; abstr 4504)
Pantuck AJ, Seligson DB, Klatte T et al (2007) Prognostic relevance of the mTOR pathway in renal cell carcinoma: implications for molecular patient selection for targeted therapy. Cancer 109(11):2257–2267
Populo H, Lopes JM, Soares P (2012) The mTOR signalling pathway in human cancer. Int J Mol Sci 13(2):1886–1918
Rini BI, Escudier B, Tomczak P et al (2011) Comparative effectiveness of axitinib versus sorafenib in advanced renal cell carcinoma (AXIS): a randomised phase 3 trial. Lancet 378(9807):1931–1939
Shinohara ET, Cao C, Niermann K et al (2005) Enhanced radiation damage of tumor vasculature by mTOR inhibitors. Oncogene 24(35):5414–5422
Wu C, Wangpaichitr M, Feun L et al (2005) Overcoming cisplatin resistance by mTOR inhibitor in lung cancer. Mol Cancer 4(1):25
Yuan R, Kay A, Berg WJ, Lebwohl D (2009) Targeting tumorigenesis: development and use of mTOR inhibitors in cancer therapy. J Hematol Oncol 2:45
Acknowledgments
The authors would like to thank the following participating investigators: S. Weigert, K. Miller (Berlin); W. Hölzer (Berlin); U. Kube (Chemnitz); S. Zastrow (Dresden); T. Gauler (Essen); A. Becker (Hamburg), M. von Staden (Hamburg); V. Grünewald (Hannover); C. Kahl (Magdeburg); M. Staehler (München); S. Bierer (Münster); A. Lück (Rostock); A. Stenzl (Tübingen); and A. Schrader (Ulm). The retrospective analysis was partially supported by Novartis Pharma, Nürnberg, Germany.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maute, L., Grünwald, V., Weikert, S. et al. Therapy of mRCC beyond mTOR-inhibition in clinical practice: results of a retrospective analysis. J Cancer Res Clin Oncol 140, 823–827 (2014). https://doi.org/10.1007/s00432-014-1610-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-014-1610-x