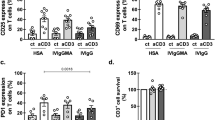
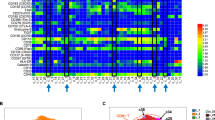
Abstract
Purpose
Tyrosine kinase inhibitors (TKIs) have well-characterized immunomodulatory effects on T and NK cells, but the effects on the humoral immunity are less well known. In this project, we studied TKI-induced changes in B cell-mediated immunity.
Methods
We collected peripheral blood (PB) and bone marrow (BM) samples from chronic myeloid leukemia (CML) patients before and during first-line imatinib (n = 20), dasatinib (n = 16), nilotinib (n = 8), and bosutinib (n = 12) treatment. Plasma immunoglobulin levels were measured, and different B cell populations in PB and BM were analyzed with flow cytometry.
Results
Imatinib treatment decreased plasma IgA and IgG levels, while dasatinib reduced IgM levels. At diagnosis, the proportion of patients with IgA, IgG, and IgM levels below the lower limit of normal (LLN) was 0, 11, and 6% of all CML patients, respectively, whereas at 12 months timepoint the proportions were 6% (p = 0.13), 31% (p = 0.042) and 28% (p = 0.0078). Lower initial Ig levels predisposed to the development of hypogammaglobulinemia during TKI therapy. Decreased Ig levels in imatinib-treated patients were associated with higher percentages of immature BM B cells. The patients, who had low Ig levels during the TKI therapy, had significantly more frequent minor infections during the follow-up compared with the patients with normal Ig values (33% vs. 3%, p = 0.0016). No severe infections were reported, except recurrent upper respiratory tract infections in one imatinib-treated patient, who developed severe hypogammaglobulinemia.
Conclusions
TKI treatment decreases plasma Ig levels, which should be measured in patients with recurrent infections.






Similar content being viewed by others
References
Baccarani M, Deininger MW, Rosti G et al (2013) European LeukemiaNet recommendations for the management of chronic myeloid leukemia: 2013. Blood 122:872–884. doi:10.1182/blood-2013-05-501569
Bantscheff M, Eberhard D, Abraham Y et al (2007) Quantitative chemical proteomics reveals mechanisms of action of clinical ABL kinase inhibitors. Nat Biotechnol 25:1035–1044. doi:10.1038/nbt1328
Caldwell CW, Patterson WP (1987) Relationship between T200 antigen expression and stages of B cell differentiation in resurgent hyperplasia of bone marrow. Blood 70:1165–1172
Carulli G, Cannizzo E, Ottaviano V et al (2010) Abnormal phenotype of bone marrow plasma cells in patients with chronic myeloid leukemia undergoing therapy with Imatinib. Leuk Res 34:1336–1339. doi:10.1016/j.leukres.2010.01.012
Druker BJ, Guilhot F, O’Brien SG et al (2006) Five-year follow-up of patients receiving Imatinib for chronic myeloid leukemia. N Engl J Med 355:2408–2417. doi:10.1056/NEJMoa062867
Duyvestyn JM, Taylor SJ, Dagger SA, et al (2014) Dasatinib targets B-lineage cells but does not provide an effective therapy for myeloproliferative disease in c-Cbl RING finger mutant mice. PLoS ONE 9:e94717. doi:10.1371/journal.pone.0094717
Hantschel O, Rix U, Superti-Furga G (2008) Target spectrum of the BCR-ABL inhibitors imatinib, nilotinib and dasatinib. Leuk Lymphoma 49:615–619. doi:10.1080/10428190801896103
Hochhaus A, O’Brien SG, Guilhot F et al (2009) Six-year follow-up of patients receiving imatinib for the first-line treatment of chronic myeloid leukemia. Leukemia 23:1054–1061. doi:10.1038/leu.2009.38
Ilander M, Hekim C, Mustjoki S (2014) Immunology and immunotherapy of chronic myeloid leukemia. Curr Hematol Malig Rep 9:17–23. doi:10.1007/s11899-013-0190-1
Kantarjian H, Shah NP, Hochhaus A et al (2010) Dasatinib versus Imatinib in newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med 362:2260–2270. doi:10.1056/NEJMoa1002315
Kantarjian HM, Giles FJ, Bhalla KN et al (2011) Nilotinib is effective in patients with chronic myeloid leukemia in chronic phase after imatinib resistance or intolerance: 24-month follow-up results. Blood 117:1141–1145. doi:10.1182/blood-2010-03-277152
Khoury HJ, Cortes JE, Kantarjian HM et al (2012) Bosutinib is active in chronic phase chronic myeloid leukemia after imatinib and dasatinib and/or nilotinib therapy failure. Blood 119:3403–3412. doi:10.1182/blood-2011-11-390120
Kim A, Seong KM, Kang HJ et al (2015) Inhibition of Lyn is a promising treatment for mantle cell lymphoma with bortezomib resistance. Oncotarget 6:38225–38238. doi:10.18632/oncotarget.5425
Lavallade H de, Khoder A, Hart M et al (2013) Tyrosine kinase inhibitors impair B-cell immune responses in CML through off-target inhibition of kinases important for cell signaling. Blood 122:227–238. doi:10.1182/blood-2012-11-465039
McCaig AM, Cosimo E, Leach MT, Michie AM (2011) Dasatinib inhibits B cell receptor signalling in chronic lymphocytic leukaemia but novel combination approaches are required to overcome additional pro-survival microenvironmental signals. Br J Haematol 153:199–211. doi:10.1111/j.1365-2141.2010.08507.x
Nitta M, Kato Y, Strife A et al (1985) Incidence of involvement of the B and T lymphocyte lineages in chronic myelogenous leukemia. Blood 66:1053–1061
Oksvold MP, Duyvestyn JM, Dagger SA et al (2015) The targeting of human and mouse B lymphocytes by dasatinib. Exp Hematol 43(352–363):e4. doi:10.1016/j.exphem.2015.01.008
Quintás-Cardama A, De Souza Santos FP, Kantarjian H et al (2009) Dynamics and management of cytopenias associated with dasatinib therapy in patients with chronic myeloid leukemia in chronic phase after Imatinib failure. Cancer 115:3935–3943. doi:10.1002/cncr.24432
Reinwald M, Boch T, Hofmann W-K, Buchheidt D (2016) Risk of infectious complications in hemato-oncological patients treated with kinase inhibitors. Biomark Insights 10:55–68. doi:10.4137/BMI.S22430
Rix U, Hantschel O, Dürnberger G et al (2007) Chemical proteomic profiles of the BCR-ABL inhibitors imatinib, nilotinib, and dasatinib reveal novel kinase and nonkinase targets. Blood 110:4055–4063. doi:10.1182/blood-2007-07-102061
Santachiara R, Maffei R, Martinelli S et al (2008) Development of hypogammaglobulinemia in patients treated with imatinib for chronic myeloid leukemia or gastrointestinal stromal tumor. Haematologica 93:1252–1255. doi:10.3324/haematol.12642
Shah VO, Civin CI, Loken MR (1988) Flow cytometric analysis of human bone marrow. IV. Differential quantitative expression of T-200 common leukocyte antigen during normal hemopoiesis. J Immunol 140:1861–1867
Steegmann JL, Moreno G, Alaez C et al (2003) Chronic myeloid leukemia patients resistant to or intolerant of interferon alpha and subsequently treated with imatinib show reduced immunoglobulin levels and hypogammaglobulinemia. Haematologica 88:762–768
Takahashi N, Miura I, Saitoh K, Miura AB (1998) Lineage involvement of stem cells bearing the philadelphia chromosome in chronic myeloid leukemia in the chronic phase as shown by a combination of fluorescence-activated cell sorting and fluorescence in situ hybridization. Blood 92:4758–4763
Talpaz M, Shah NP, Kantarjian H et al (2006) Dasatinib in Imatinib-resistant philadelphia chromosome–positive leukemias. N Engl J Med 354:2531–2541. doi:10.1056/NEJMoa055229
Tybulewicz VLJ, Crawford CE, Jackson PK et al (1991) Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell 65:1153–1163. doi:10.1016/0092-8674(91)90011-M
Zipfel PA, Grove M, Blackburn K et al (2000) The c-Abl Tyrosine Kinase Is Regulated Downstream of the B Cell Antigen Receptor and Interacts with CD19. J Immunol 165:6872–6879. doi:10.4049/jimmunol.165.12.6872
Acknowledgements
The authors thank the personnel at the Hematology Research Unit Helsinki, Helsinki University Hospital HUSLAB flow cytometry laboratory, and Aija Helin from HUSLAB for their expert technical assistance. This work was supported by the Academy of Finland, the Finnish Cancer Societies, the Sigrid Juselius Foundation, the Finnish Cancer Institute, the Signe and Ane Gyllenberg Foundation, the Otto A. Malm Foundation, the EUTOS project for CML 2016, and investigator initiated research grants from Pfizer, and Novartis. NordCML Study Group has received research funding from Bristol-Myers Squibb for the NordCML006 and NordCML007 clinical studies.
Funding
This work was supported by the Academy of Finland, the Finnish Cancer Societies, the Sigrid Juselius Foundation, the Finnish Cancer Institute, the Signe and Ane Gyllenberg Foundation, the Otto A. Malm Foundation, the EUTOS project for CML 2016, and investigator initiated research grants from Pfizer and Novartis. NordCML Study Group has received research funding from Bristol-Myers Squibb for the NordCML006 and NordCML007 clinical studies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
SM, JR, and HHjH have obtained honoraria and research funding from Bristol-Myers Squibb, Novartis, Ariad, and Pfizer.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rajala, H.L.M., Missiry, M.E., Ruusila, A. et al. Tyrosine kinase inhibitor therapy-induced changes in humoral immunity in patients with chronic myeloid leukemia. J Cancer Res Clin Oncol 143, 1543–1554 (2017). https://doi.org/10.1007/s00432-017-2378-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-017-2378-6