Abstract
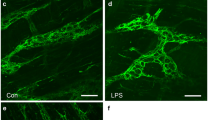
Changes in intestinal function, notably impaired transit, following ischemia/reperfusion (I/R) injury are likely to derive, at least in part, from damage to the enteric nervous system. Currently, there is a lack of quantitative data and methods on which to base quantitation of changes that occur in enteric neurons. In the present work, we have investigated quantifiable changes in response to ischemia of the mouse small intestine followed by reperfusion from 1 h to 7 days. I/R caused distortion of nitric oxide synthase (NOS)-containing neurons, the appearance of a TUNEL reaction in neurons, protein nitrosylation and translocation of Hu protein. Protein nitrosylation was detected after 1 h and was detectable in 10% of neurons by 6 h in the ischemic region, indicating that reactive peroxynitrites are rapidly produced and can interact with proteins soon after reperfusion. Apoptosis, revealed by TUNEL staining, was apparent at 6 h. The profile sizes of NOS neurons were increased by 60% at 2 days and neurons were still swollen at 7 days, both in the ischemic region and proximal to the ischemia. The distribution of the enteric neuron marker and oligonucleotide binding protein, Hu, was significantly changed in both regions. Hu protein translocation to the nucleus was apparent by 3 h and persisted for up to 7 days. Particulate Hu immunoreactivity was observed in the ganglia 3 h after I/R but was never observed in control. Our observations indicate that effects of I/R injury can be detected after 1 h and that neuronal changes persist to at least 7 days. Involvement of NO and reactive oxygen species in the changes is indicated by the accumulation of nitrosylated protein aggregates and the swelling and distortion of nitrergic neurons. It is concluded that damage to the enteric nervous system, which is likely to contribute to functional deficits following ischemia and re-oxygenation in the intestine, can be quantified by Hu protein translocation, protein nitrosylation, swelling of nitrergic neurons and apoptosis.









Similar content being viewed by others
References
An S, Hishikawa Y, Koji T (2005) Induction of cell death in rat small intestine by ischemia reperfusion: differential roles of Fas/Fas ligand and Bcl-2/Bax systems depending upon cell types. Histochem Cell Biol 123:249–261
Aube AC, Cherbut C, Barbier M, Xing JH, Roze C, Galmiche JP (1999) Altered myoelectrical activity in noninflamed ileum of rats with colitis induced by trinitrobenzene sulphonic acid. Neurogastroenterol Motil 11:55–62
Barreau C, Paillard L, Osborne HB (2005) AU-rich elements and associated factors: are there unifying principles? Nucleic Acids Res 33:7138–7150
Blandizzi C, Fornai M, Colucci R, Baschiera F, Barbara G, De Giorgio R, De Ponti F, Breschi MC, Del Tacca M (2003) Altered prejunctional modulation of intestinal cholinergic and noradrenergic pathways by α2-adrenoceptors in the presence of experimental colitis. Br J Pharmacol 139:309–320
Brehmer A, Schrödl F, Neuhuber W (2006) Morphology of VIP/nNOS-immunoreactive myenteric neurons in the human gut. Histochem Cell Biol 125:557–565
Calcina F, Barocelli E, Bertoni S, Furukawa O, Kaunitz J, Impicciatore M, Sternini C (2005) Effect of N-methyl-D-aspartate receptor blockade on neuronal plasticity and gastrointestinal transit delay induced by ischemia/reperfusion in rats. Neuroscience 134:39–49
Cuzzocrea S, Dugo L, Patel NSA, Di Paola R, Cockerill GW, Genovese T, Thiemermann C (2004) High-density lipoproteins reduce the intestinal damage associated with ischemia/reperfusion and colitis. Shock 21:342–351
De Giorgio R, Barbara G, Furness JB, Tonini M (2007) Novel therapeutic targets for enteric nervous system disorders. Trends Pharmacol Sci 28:473–481
De Giorgio R, Camilleri M (2004) Human enteric neuropathies: morphology and molecular pathology. Neurogastroenterol Motil 16:515–531
De Giorgio R, Guerrini S, Barbara G, Stanghellini V, De Ponti F, Corinaldesi R, Moses PL, Sharkey KA, Mawe GM (2004) Inflammatory neuropathies of the enteric nervous system. Gastroenterology 126:1872–1883
Deng Y, Thompson BM, Gao X, Hall ED (2007) Temporal relationship of peroxynitrite-induced oxidative damage, calpain-mediated cytoskeletal degradation and neurodegeneration after traumatic brain injury. Exp Neurol 205:154–165
Di Paola R, Menegazzi M, Mazzon E, Genovese T, Crisafulli C, Dal Bosco M, Zou Z, Suzuki H, Cuzzocrea S (2009) Protective effects of glycyrrhizin in a gut hypoxia (ischemia)-reoxygenation (reperfusion) model. Intensive Care Med 35:687–697
Dong Z, Saikumar P, Weinberg JM, Venkatachalam MA (2006) Calcium in cell injury and death. Annu Rev Pathol Mech Dis 1:405–434
Fairman CL, Clagett Dame M, Lennon VA, Epstein ML (1995) Appearance of neurons in the developing chick gut. Dev Dyn 204:192–201
Ferens DM, Chang EC, Bogeski G, Shafton AD, Kitchener PD, Furness JB (2005) Motor patterns and propulsion in the rat intestine in vivo recorded by spatio-temporal maps. Neurogastroenterol Motil 17:714–720
Ferrer-Sueta G, Radi R (2009) Chemical biology of peroxynitrite: kinetics, diffusion, and radicals. ACS Chem Biol 4:161–177
Fryer JP (2005) Intestinal transplantation: an update. Curr Opin Gastroenterol 21:162–168
Furness JB (2006) The enteric nervous system. Blackwell, Oxford
Granger N, Korthuis RJ (1995) Physiologic mechanisms of postischemic tissue injury. Annu Rev Physiol 57:311–332
Grossie VB, Weisbrodt NW, Moore FA, Moody F (2001) Ischemia/reperfusion-induced disruption of rat small intestine transit is reversed by total enteral nutrition. Nutrition 17:939–943
Hakgüder G, Akgür FM, Ateş O, Olguner M, Aktuğ T, Özer E (2002) Short-term intestinal ischemia-reperfusion alters intestinal motility that can be preserved by xanthine oxidase inhibition. Dig Dis Sci 47:1279–1283
Hinman MN, Lou H (2008) Diverse molecular functions of Hu proteins. Cell Mol Life Sci 65:3168–3181
Hossmann K-A (2006) Pathophysiology and therapy of experimental stroke. Cell Mol Neurobiol 26:1057–1083
Jacobson K, McHugh K, Collins SM (1995) Experimental colitis alters myenteric nerve function at inflamed and noninflamed sites in the rat. Gastroenterology 109:718–722
Kim J, Seok YM, Jung K-J, Park KM (2009) Reactive oxygen species/oxidative stress contributes to progression of kidney fibrosis following transient ischemic injury in mice. Am J Physiol 297:F461–F470
Lindeström L-M, Ekblad E (2004) Structural and neuronal changes in rat ileum after ischemia with reperfusion. Dig Dis Sci 49:1212–1222
Mallick IH, Yang W, Winslet MC, Seifalian AM (2004) Ischemia–reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci 49:1359–1377
Matthijsen RA, Derikx JPM, Kuipers D, van Dam RM, Dejong CHC, Buurman WA (2009) Enterocyte shedding and epithelial lining repair following ischemia of the human small intestine attenuate inflammation. PLoS ONE 4:e7045
Mei F, Guo S, He Y-T, Zhu J, Zhou D-S, Niu J-Q, Wang H-Z, Tian Y-P (2009) Apoptosis of interstitial cells of Cajal, smooth muscle cells, and enteric neurons induced by intestinal ischemia and reperfusion injury in adult guinea pigs. Virchows Arch 454:401–409
O'Hara JR, Lomax AE, Mawe GM, Sharkey KA (2007) Ileitis alters neuronal and enteroendocrine signalling in guinea pig distal colon. Gut 56:186–194
Piao DX, Jiang HC, Kosaka M, Shibata T, Ohtsuka A, Murakami T (1999) Cytoplasmic delayed neuronal death in the myenteric plexus of the rat small intestine after ischemia. Arch Histol Cytol 62:383–392
Pompolo S, Furness JB (1990) Ultrastructure and synaptology of neurons immunoreactive for gamma-aminobutyric acid in the myenteric plexus of the guinea pig small intestine. J Neurocytol 19:539–549
Pontell L, Sharma, P, Rivera, LR, Thacker, M, Tan, YH, Brock, JA, Furness, JB (2010) Damaging effects of ischemia/ reperfusion on intestinal muscle. Cell Tissue Res (in press)
Qu Z-D, Thacker M, Castelucci P, Bagyánszki M, Epstein ML, Furness JB (2008) Immunohistochemical analysis of neuron types in the mouse small intestine. Cell Tissue Res 334:147–161
Rao SSC, Read NW, Brown C, Bruce C, Holdsworth CD (1987) Studies on the mechanism of bowel disturbance in ulcerative colitis. Gastroenterology 93:934–940
Rivera LR, Thacker M, Castelucci P, Bron R, Furness JB (2009) The reactions of specific neuron types to intestinal ischemia in the guinea pig enteric nervous system. Acta Neuropathol 118:261–270
Silva MACP, de Meirelles LR, Bustorff-Silva JM (2007) Changes in intestinal motility and in the myenteric plexus in a rat model of intestinal ischemia-reperfusion. J Pediatr Surg 42:1062–1065
Soller M, White K (2003) ELAV inhibits 3’-end processing to promote neural splicing of ewg pre-mRNA. Genes Dev 17:2526–2538
Swenson O (2002) Hirschsprung's Disease: a review. Pediatrics 109:914–918
Türler A, Kalff JC, Heeckt P, Abu-Elmagd KM, Schraut WH, Bond GJ, Moore BA, Brünagel G, Bauer AJ (2002) Molecular and functional observations on the donor intestinal muscularis during human small bowel transplantation. Gastroenterology 122:1886–1897
Udassin R, Eimeri D, Schiffman J, Haskel Y (1995) Postischemic intestinal motility in rat is inversely correlated to length of ischemia, an in vivo animal model. Dig Dis Sci 40:1035–1038
Van Nassauw L, Bogers J, Van Marck E, Timmermans JP (2001) Role of reactive nitrogen species in neuronal cell damage during intestinal schistosomiasis. Cell Tissue Res 303:329–336
Wedel T, Krammer HJ, Kühnel W, Sigge W (1998) Alterations of the enteric nervous system in neonatal necrotizing enterocolitis revealed by whole-mount immunohistochemistry. Pediatr Pathol Lab Med 18:57–70
Williamson S, Pompolo S, Furness JB (1996) GABA and nitric oxide synthase immunoreactivities are colocalized in a subset of inhibitory motor neurons of the guinea-pig small intestine. Cell Tissue Res 284:29–37
Acknowledgements
We thank Dr Daniel Poole for helpful comments on the manuscript and Dr Trung Nguyen for advising on quantitation of neuronal changes
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rivera, L.R., Thacker, M., Pontell, L. et al. Deleterious effects of intestinal ischemia/reperfusion injury in the mouse enteric nervous system are associated with protein nitrosylation. Cell Tissue Res 344, 111–123 (2011). https://doi.org/10.1007/s00441-010-1126-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-010-1126-x