Abstract
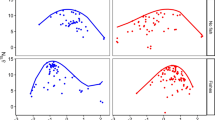
The mismatch between the turnover rates of predators and prey is one of the oldest explanations for the existence of inverted trophic pyramids. To date, the hypotheses regarding trophic pyramids have all been based on consumptive trophic links between predators and prey, and the relative contribution of non-consumptive effects is still unknown. In this study, we investigated if the inversion of pyramids in bromeliad ecosystems is driven by (i) a rapid colonization of organisms having short cohort interval production (CPI), and (ii) the prevalence of consumptive or non-consumptive effects of top predators. We used a manipulative experiment to investigate the patterns of prey colonization and to partition the net effects of the dominant predator (damselfly larvae) on biomass pyramids into consumptive (uncaged damselfly larvae) and non-consumptive effects (caged damselfly larvae). Consumptive effects of damselflies strengthened the inversion of trophic pyramids. Non-consumptive effects, however, did not affect the shape of biomass pyramids. Instead, the rapid colonization of organisms with predominantly short CPI sustained the large biomass of top predators found in natural bromeliad ecosystems. Prey colonized bromeliads rapidly, but this high production was never visible as standing stock because damselflies reduce prey densities by more than a magnitude through direct consumption. Our study adds to the growing evidence that there are a variety of possible ways that biomass can be trophically structured. Moreover, we suggest that the strength of biomass pyramids inversion may change with the time of ecological succession as prey communities become more equitable.




Similar content being viewed by others
References
Amundrud SL, Srivastava DS (2015) Drought sensitivity predicts habitat size sensitivity in an aquatic ecosystem. Ecology 96:1957–1965. https://doi.org/10.1890/14-1828.1
Armbruster P, Hutchinson RA, Cotgreave P (2002) Factors influencing community structure in a South American tank bromeliad fauna. Oikos 96:225–234. https://doi.org/10.1034/j.1600-0706.2002.960204.x
Barry M (1994) The costs of crest induction for Daphnia carinata. Oecologia 97:278–288. https://doi.org/10.1007/bf00323161
Benke AC, Huryn AD (2007) Secondary production of macroinvertebrates. In: Hauer FR, Lamberti G (eds) Methods in stream ecology. Academic Press, Elsevier, pp 691–710
Benke AC, Van Arsdall TC, Gillespie DM, Parrish FK (1984) Invertebrate productivity in a subtropical blackwater river: the importance of habitat and life history. Ecol Monogr 54:25–63. https://doi.org/10.2307/1942455
Bernabé TN, de Omena PM, Santos VPD, de Siqueira VM, de Oliveira VM, Romero GQ (2018) Warming weakens facilitative interactions between decomposers and detritivores, and modifies freshwater ecosystem functioning. Glob Chang Biol 24:3170–3186. https://doi.org/10.1111/gcb.14109
Breviglieri CPB, Romero GQ (2017) Terrestrial vertebrate predators drive the structure and functioning of aquatic food webs. Ecology 98:2069–2080. https://doi.org/10.1002/ecy.1881
Brown JH, Gillooly JF, Allen AP, Savage VM, West GB (2004) Toward a metabolic theory of ecology. Ecology 7:1771–1789. https://doi.org/10.1890/03-9000
Buck KR, Chavez FP, Campbell L (1996) Basin-wide distributions of living carbon components and the inverted trophic pyramid of the central gyre of the North Atlantic Ocean, summer 1993. Aquat Microb Ecol 10:283–298. https://doi.org/10.3354/ame010283
Buskirk JV (2000) The costs of an inducible defense in anuran larvae. Ecology 10:2813–2821. https://doi.org/10.1890/0012-9658(2000)081%5b2813:tcoaid%5d2.0.co;2
de Omena PM, Srivastava DS, Romero GQ (2017) Does the strength of cross-ecosystem trophic cascade vary with ecosystem size? A test using a natural microcosm. Freshw Biol 62:724–736. https://doi.org/10.1111/fwb.12897
Del Giorgio PA, Cole JJ, Caraco NF, Peters RH (1999) Linking planktonic biomass and metabolism to net gas fluxes in northern temperate lakes. Ecology 80:1422–1424. https://doi.org/10.1890/0012-9658(1999)080%5b1422:lpbamt%5d2.0.co;2
Dézerald O, Céréghino R, Corbara B, Dejean A, Leroy C (2015) Temperature: diet interactions affect survival through foraging behavior in a bromeliad-dwelling predator. Biotropica 47:569–578. https://doi.org/10.1111/btp.12249
Dézerald O, Leroy C, Corbara DB, Talaga S, Céréghino R (2017) Environmental drivers of invertebrate population dynamics in Neotropical tank bromeliads. Freshw Biol 62:229–242. https://doi.org/10.1111/fwb.12862
Dézerald O, Leroy C, Corbara B et al (2018) Tank bromeliads sustain high secondary production in neotropical forests. Aquat Sci 80:14. https://doi.org/10.1007/s00027-018-0566-3
Downes S (2001) Trading heat and food for safety: costs of predator avoidance in a lizard. Ecology 82:2870–2881. https://doi.org/10.1890/0012-9658(2001)082%5b2870:thaffs%5d2.0.co;2
Drake EF, Bishop BP (1971) Life cycle and laboratory diet for Atrichopogon jacobsoni (de Meijere) (Diptera: Ceratopogonidae). Proc Hawaii Entomol Soc 21:63–66
Elser JJ, Fagan WF, Denno RF, Dobberfuhl DR, Folarin A, Huberty A, Interlandi S, Kilham SS, McCauley E, Schulz KL, Siemann EH, Sterner RW (2000) Nutritional constraints in terrestrial and freshwater food webs. Nature 408:578–580. https://doi.org/10.1038/35046058
Elton C (1927) Animal ecology. Macmillan, New York
Farjalla VF, González AL, Céréghino R, Dézeral O, Marino NAC, Piccoli GCO, Richardson BA, Richardsion MJ, Romero GQ, Srivastava DS (2016) Terrestrial support of aquatic food webs depends on light inputs: a geographically-replicated test using tank bromeliads. Ecology 97:2147–2156. https://doi.org/10.1002/ecy.1432
Gasol JM, Del Giorgio PA, Duarte CM (1997) Biomass distribution in marine planktonic communities. Limnol Oceanogr 42:1353–1363. https://doi.org/10.4319/lo.1997.42.6.1353
Gilbert B, Tunney TD, McCann KS, DeLong JP, Vasseur DA, Savage V, Shurin JB, Dell AI, Barton BT, Harley CDG, Kharouba HM, Kratina P, Blanchard JL, Clements C, Winder M, Greig HS, O’Connor MI (2014) A bioenergetics framework for the temperature dependence of trophic interactions. Ecol Lett 17:902–914. https://doi.org/10.1111/ele.12307
Hammill E, Atwood TB, Corvalan P, Srivastava DS (2015a) Behavioural responses to predation may explain shifts in community structure. Freshw Biol 60:125–135. https://doi.org/10.1111/fwb.12475
Hammill E, Atwood TB, Srivastava DS (2015b) Predation threat alters composition and functioning of bromeliad ecosystems. Ecosystems 18:857–866. https://doi.org/10.1007/s10021-015-9866-9
Hatton IA, McCann KS, Fryxell JM, Davies TJ, Smerlak M, Sinclair ARE, Loreau M (2015) The predator-prey power law: biomass scaling across terrestrial and aquatic biomes. Science 344:aac6284. https://doi.org/10.1126/science.aac6284
Hedström I, Sahlén G (2001) A key to the adult Costa Rican “helicopter” damselflies (Odonata: Pseudostigmatidae) with notes on their phenology and life zone preferences. Rev Biol Trop 49:1037–1056
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363. https://doi.org/10.1002/bimj.200810425
Huryn AD, Benke AC (2007) Relationship between biomass turnover and body size for stream communities. Body size: the structure and function of aquatic ecosystems. Cambridge University Press, Cambridge, pp 55–76
Kitching RL (2004) Food webs and container habitats: the natural history and ecology of phytotelmata. Cambridge University Press, Cambridge
Marino NAC, Guariento RD, Dib V, Azevedo FD, Farjalla VF (2011) Habitat size determine algae biomass in tank-bromeliads. Hydrobiologia 678:191–199. https://doi.org/10.1007/s10750-011-0848-4
Marino NAC, Srivastava DS, Farjalla VF (2015) Predator kairomone change food web structure and function, regardless of cues from consumed prey. Oikos 125:1017–1026. https://doi.org/10.5061/dryad.3m2c3
McCauley DJ, Gellner G, Martinez ND, Williams RJ, Sandin SA, Micheli F, Mumby PJ, McCann KS (2018) On the prevalence and dynamics of inverted trophic pyramids and otherwise top-heavy communities. Ecol Lett. https://doi.org/10.1111/ele.12900
Merritt RW, Cummings KW (1996) An introduction to the aquatic insects of North America, 3rd edn. Kendall/Hunt Publishing Company, Dubuque
Morrison L (1999) Indirect effects of phorid fly parasitoids on the mechanisms of interspecific competition among ants. Oecologia 121:113–122. https://doi.org/10.1007/s004420050912
Mourier J, Maynard J, Parravicini V, Ballesta L, Clua E, Domeier ML, Planes S (2016) Extreme inverted trophic pyramid of reef sharks supported by spawning groupers. Curr Biol 26(201):1–2016. https://doi.org/10.1016/j.cub.2016.05.058
Moustaka-Gouni M, Vardaka E, Michaloudi E, Tryfon E, Mihalatou H, Gkelis S, Lanaras T (2006) Plankton food web structure in a eutrophic polymictic lake with a history of toxic cyanobacterial blooms. Limnol Oceanogr 51:715–727. https://doi.org/10.4319/lo.2006.51.1_part_2.0715
Ngai JT, Srivastava DS (2006) Predators accelerate nutrient cycling in a bromeliad ecosystem. Science 314:963. https://doi.org/10.1126/science.1132598
O’Neill RV (1976) Ecosystem persistence and heterotrophic regulation. Ecology 57:1244–1253. https://doi.org/10.2307/1935048
Odum EP (1971) Fundamentals of Ecology, 3rd edn. WB Saunders Co, Philadelphia
Petermann JS, Farjalla VF, Jocque M, Kratina P, MacDonald AAM, Marino NAC, de Omena PM, Piccoli GC, Richardson BA, Richardson MJ, Romero GQ, Videla M, Srivastava DS (2015) Dominant predators mediate the impact of habitat size on trophic structure in bromeliad invertebrate communities. Ecology 96:428–439. https://doi.org/10.1890/14-0304.1
Preisser EL, Blonick DI, Bernard MF (2005) Scared to death? The effects of intimidation and consumption in predator-prey interactions. Ecology 86:501–509. https://doi.org/10.1890/04-0719
Relyea RA (2001) Morphological and behavioral plasticity of larval anurans in response to different predators. Ecology 82:523–540. https://doi.org/10.2307/2679877
Romero GQ, Srivastava DS (2010) Food-web composition affects cross-ecosystem interactions and subsidies. J Anim Ecol 79:1122–1131. https://doi.org/10.1111/j.1365-2656.2010.01716.x
Romero GQ, Piccoli GCO, de Omena PM, Gonçalves-Souza T (2016) Food web structure shaped by habitat size and climate across a latitudinal gradient. Ecology 97:2705–2715. https://doi.org/10.1890/14-0304.1
Sandin SA, Smith JE, DeMartini EE, Dinsdale EA, Donner SD, Friedlander AM, Konotchick T, Malay M, Maragos JE, Obura D, Pantos O, Paulay G, Richie M, Rohwer F, Schroeder RE, Walsh S, Jackson JBC, Knowlton N, Sala E (2008) Baselines and degradation of coral reefs in the Northern Line Islands. PLoS One 3:e1548. https://doi.org/10.1371/journal.pone.0001548
Schmitz OJ (1998) Direct and indirect effects of predation and predation risk in old-field interaction webs. Am Nat 151:327–342. https://doi.org/10.1086/286122
Schmitz OJ (2008) Effects of predator hunting mode on grassland ecosystem function. Science 319:952–954. https://doi.org/10.1126/science.1152355
Schmitz OJ, Krivan V, Ovadia O (2004) Trophic cascades: the primacy of trait-mediated indirect interactions. Ecol Lett 7:153–163. https://doi.org/10.1111/j.1461-0248.2003.00560.x
Shurin JB, Gruner DS, Hillebrand H (2006) All wet or dried up? Real differences between aquatic and terrestrial food webs. Proc R Soc B 273:1–9. https://doi.org/10.1098/rspb.2005.3377
Shurin JB, Clasen JL, Greig HS, Kratina P, Thompson PL (2012) Warming shifts top–down and bottom–up control of pond food web structure and function. Phil Trans R Soc B 367:3008–3017. https://doi.org/10.1098/rstb.2012.0243
Srivastava DS (2006) Habitat structure, trophic structure and ecosystem function: interactive effects in a bromeliad–insect community. Oecologia 149:493–504. https://doi.org/10.1007/s00442-006-0467-3
Srivastava DS, Trzcinski MK, Richardson BA, Gilbert B (2008) Why are predators more sensitive to habitat size than their prey? Insights from bromeliad insect food webs. Am Nat 172:761–771. https://doi.org/10.1086/592868
Trebilco R, Baum JK, Salomon AK, Dulvy NK (2013) Ecosystem ecology: size-based constraints on the pyramids of life. Trends Ecol Evol 28:423–431. https://doi.org/10.1016/j.tree.2013.03.008
Tunney TD, McCann KS, Lester NP, Shuter BJ (2012) Food web expansion and contraction in response to changing environmental conditions. Nat Comm 3:1105. https://doi.org/10.1038/ncomms2098
Vonesh JR, Kraus JM, Rosenberg JS, Chase JM (2009) Predator effects on aquatic community assembly: disentangling the roles of habitat selection and post-colonization processes. Oikos 118:1219–1229. https://doi.org/10.1111/j.1600-0706.2009.17369.x
Wang H, Morrison W, Singh A, Weiss HH (2009) Modeling inverted biomass pyramids and refuges in ecosystems. Ecol Model 220:1376–1382. https://doi.org/10.1016/j.ecolmodel.2009.03.005
Werner EE, Peacor SD (2003) A review of trait-mediated indirect interactions in ecological communities. Ecology 84:1083–1100. https://doi.org/10.1890/0012-9658(2003)084%5b1083:AROTII%5d2.0.CO;2
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
The authors thank T. N. Bernabé, M. R. Braga, A. Degressi, C. Vieira, G. C. O. Piccoli, G. H. Migliorini, N. A. C. Marino for field assistance; the staff of the “Parque Estadual da Ilha do Cardoso” for the logistic support. P. M. de Omena received fellowship from São Paulo Research Foundation (FAPESP: 2009/51702-0), “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior” and Department of Foreign Affairs, Trade and Development Canada (CAPES-DFAIT: BEX 8377/12-0), and “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior” (PNPD-CAPES; 2014/04603-4). G. Q. Romero was supported by the “Conselho Nacional de Desenvolvimento Científico e Tecnológico” (CNPq) research grant, and D.S. Srivastava by Natural Science and Engineering Research Council (Canada) Discovery and Accelerator Grants. This study was also supported by CESAB-FRB as part of the activities of the FunctionalWebs Working Group.
Funding
All data analysed during this study are available as electronic supplementary material.
Author information
Authors and Affiliations
Contributions
PMO, DSS, GQR conceived the study. PMO performed the field experiment, insect identification and biomass estimations. PMO, DSS and GQR interpreted the data. PMO wrote the paper with the assistance of DSS and GQR.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable institutional and/or national guidelines for care and use of animals were followed.
Additional information
Communicated by Jonathan Shurin.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Omena, P.M., Srivastava, D.S. & Romero, G.Q. Consumptive effects and mismatch in predator–prey turnover rates cause inversion of biomass pyramids. Oecologia 190, 159–168 (2019). https://doi.org/10.1007/s00442-019-04394-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-019-04394-0