Abstract
Background
Advanced hepatocellular carcinoma (HCC) responds poorly to conventional systemic therapies. Therefore, new effective therapy strategies are urgently needed. Molecular targeted therapies have entered the field of anti-neoplastic treatment and are being used on their own and in combination with other drugs. Sorafenib inhibits proliferation and angiogenesis of HCC by suppressing the Raf serine/threonine kinases and the receptor tyrosine kinases. The proteasome inhibitor bortezomib has shown activity in a variety of solid tumors, including HCC. However, the precise anti-proliferative mechanisms of these agents remain unclear.
Methods
We treated human hepatoma cell lines (Huh7 and Hep3B) and immortalized human hepatocyte (OUMS29) with sorafenib and/or proteasome inhibitors, including epoxomicin and acetyl-leucyl-leucyl-norleucinal. Cytotoxic effects were examined by morphometric analyses of apoptosis and necrosis. Apoptosis was also evaluated by Western blotting of keratin18, PARP and caspase3. The activity of Akt and stress-activated protein kinases was examined by Western blotting.
Results
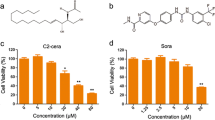
Both sorafenib and proteasome inhibitors induced apoptosis in Huh7 and OUMS29. However, sorafenib attenuated proteasome inhibitor-induced apoptosis. Sorafenib induced necrosis, especially in combination with proteasome inhibitors. Sorafenib induced down-regulation of Akt synergistically in combination with proteasome inhibitors in Huh7. Sorafenib inhibited both the JNK and p38 pathways in a time- and dose-dependent manner. In addition, sorafenib also inhibited proteasome inhibitor-mediated JNK and p38 activation in both Huh7 and OUMS29.
Conclusions
Sorafenib enhances the anti-proliferative effect of proteasome inhibitors in part by inactivating the Akt signaling pathway and modulating stress-activated protein kinases. The combination of these agents could be an ideal molecular targeted therapy for HCC.





Similar content being viewed by others
Abbreviations
- Ab:
-
Antibody
- ALLN:
-
Acetyl-leucyl-leucyl-norleucinal
- CREB:
-
cAMP response element-binding protein
- DAPI:
-
4′6-diamidino-2-phenylindole
- ER:
-
Endoplasmic reticulum
- ERK:
-
Extracellular signal-regulated protein kinase
- HCC:
-
Hepatocellular carcinoma
- IGF-1:
-
Insulin-like growth factor-1
- JNK:
-
c-Jun N-terminal kinase
- K:
-
Keratin
- MAPK:
-
Mitogen-activated protein kinase
- MEK:
-
MAPK/ERK kinase
- mTOR:
-
Mammalian target of rapamycin
- mTORC:
-
Mammalian target of rapamycin complex
- NFκB:
-
Nuclear factor-kappa B
- PARP:
-
Poly-ADP-ribose-polymerase
- PDGFR:
-
Platelet-derived growth factor receptor
- PI:
-
Proteasome inhibitor
- PI3K:
-
Phosphoinositide 3-kinase
- ROS:
-
Reactive oxygen species
- TNF-α:
-
Tumor necrosis factor-α
- TRAF2:
-
Tumor necrosis factor receptor-associated factor-2
- VEGFR:
-
Vascular endothelial growth factor receptor
References
Forner A, Llovet JM, Bruix J. Hepatocellular carcinoma. Lancet. 2012;379:1245–55.
Finn RS. Development of molecularly targeted therapies in hepatocellular carcinoma: where do we go now? Clin Cancer Res. 2010;16:390–7.
Schwartz M, Roayaie S, Konstadoulakis M. Strategies for the management of hepatocellular carcinoma. Nat Clin Pract Oncol. 2007;4:424–32.
Zhu AX. Development of sorafenib and other molecularly targeted agents in hepatocellular carcinoma. Cancer. 2008;112:250–9.
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, SHARP Investigation Study Group, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–90.
Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomized, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25–34.
Wilhelm SM, Carter C, Tang L, Wilkie D, McNabola A, Rong H, et al. BAY 43–9006 exhibits broad spectrum oral autitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res. 2004;64:7099–109.
Adams J. The proteasome: a suitable antineoplastic target. Nat Rev Cancer. 2004;4:349–60.
Kim GP, Mahoney MR, Szydlo D, Mok TS, Marshke R, Holen K, et al. An international, multicenter phase II trial of bortezomib in patients with hepatocellular carcinoma. Invest New Drugs. 2012;30:387–94.
Voorhees PM, Orlowski RZ. The proteasome and proteasome inhibitors in cancer therapy. Annu Rev Pharmacol Toxicol. 2006;46:189–213.
Chen KF, Yeh PY, Yeh KH, Lu YS, Huang SY, Cheng AL. Down-regulation of phosphor-Akt is a major molecular determinant of bortezomib-induced apoptosis in hepatocellular carcinoma cells. Cancer Res. 2008;68:6698–707.
Ganten TM, Koschny R, Haas TL, Sykora J, Li-Weber M, Herzer K, et al. Proteasome inhibition sensitizes hepatocellular carcinoma cells, but not human hepatocytes, to TRAIL. Hepatology. 2005;42:588–97.
Yu C, Friday BB, Lai JP, Yang L, Sarkaria J, Kay NE, et al. Cytotoxic synergy between the multikinase inhibitor sorafenib and the proteasome inhibitor bortezomib in vitro: induction of apoptosis through Akt and c-Jun NH2-terminal kinase pathways. Mol Cancer Ther. 2006;5:2378–87.
Chen KF, Yu HC, Liu TH, Lee SS, Chen PJ, Cheng AL. Synergistic interactions between sorafenib and bortezomib in hepatocellular carcinoma involve PP2A-dependent Akt inactivation. J Hepatol. 2010;52:88–95.
Wright JJ. Combination therapy of bortezomib with novel targeted agents: an emerging treatment strategy. Clin Cancer Res. 2010;16:4094–104.
Kobayashi N, Miyazaki M, Fukaya K, Inoue Y, Sakaguchi M, Uemura T, et al. Transplantation of highly differentiated immortalized human hepatocytes to treat acute liver failure. Transplantation. 2000;69:202–7.
Harada M, Strnad P, Toivola DM, Omary MB. Autophagy modulates keratin-containing inclusion formation and apoptosis in cell culture in a context-dependent fashion. Exp Cell Res. 2008;314:1753–64.
Groll M, Huber R. Inhibitors of the eukaryotic 20S proteasome core particle: a structural approach. Biochim Biophys Acta. 2004;1695:33–44.
Alexia C, Fourmatgeat P, Delautier D, Groyer A. Insulin-like growth factor-I stimulates H4II rat hepatoma cell proliferation: dominant role of PI3′K/Akt signaling. Exp Cell Res. 2006;312:1142–52.
Kawaguchi T, Yoshida T, Harada M, Hisamoto T, Nagao Y, Ide T, et al. Hepatitis C virus down-regulates insulin receptor substrates 1 and 2 through up-regulation of suppressor of cytokine signaling 3. Am J Pathol. 2004;165:1499–508.
Ku NO, Liao J, Omary MB. Apoptosis generates stable fragments of human type I keratins. J Biol Chem. 1997;272:33197–203.
Caulin C, Salvesen GS, Oshima RG. Caspase cleavage of keratin 18 and reorganization of intermediate filaments during epitherial cell apoptosis. J Cell Biol. 1997;138:1379–94.
Liu L, Cao Y, Chen C, Zhang X, McNabola A, Wilkie D, et al. Sorafenib blocks the RAF/MEK/ERK pathway, inhibits tumor angiogenesis, and induces tumor cell apoptosis in hepatocellular carcinoma model PLC/PRF/5. Cancer Res. 2006;66:11851–8.
Bressac B, Galvin KM, Liang TJ, Isselbacher KJ, Wands JR, Ozturk M. Abnormal structure and expression of p53 gene in human hepatocellular carcinoma. Proc Natl Acad Sci USA. 1990;87:1973–7.
Shimizu S, Takehara T, Hikita H, Kodama T, Tsunematsu H, Miyagi T, et al. Inhibition of autophagy potentiates the antitumor effect of the multikinase inhibitor sorafenib in hepatocellular carcinoma. Int J Cancer. 2012;131:548–57.
Foster KG, Fingar DC. Mammalian target of rapamycin (mTOR): conducting the cellular signaling symphony. J Biol Chem. 2010;285:14071–7.
Whittaker S, Marais R, Zhu AX. The role of signaling pathways in the development and treatment of hepatocellular carcinoma. Oncogene. 2010;29:4989–5005.
Krens SF, Spaink HP, Snaar-Jagalska BE. Functions of the MAPK family in vertebrate-development. FEBS Lett. 2006;580:4984–90.
Hagiwara S, Kudo M, Nagai T, Inoue T, Ueshima K, Nishida N, et al. Activation of JNK and high expression level of CD133 predict a poor response to sorafenib in hepatocellular carcinoma. Br J Cancer. 2012;106:1997–2003.
Takeda K, Naguro I, Nishitoh H, Matsuzawa A, Ichijo H. Apoptosis signaling kinases: from stress response to health outcomes. Antioxid Redox Signal. 2011;15:719–61.
Huynh H. Molecularly targeted therapy in hepatocellular carcinoma. Biochem Pharmacol. 2010;80:550–60.
Acknowledgments
Our work is supported by in part by a grant-in-aid (2059798, 23591000) from the Ministry of Education, Culture, Sports, Science and Technology, Japan and UOEH Grant for Advanced Research (H22-1) to M.H. We thank Ms. Y. Katsuki and Ms. H. Mihara for their expert technical assistance.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Honma, Y., Shimizu, S., Takehara, T. et al. Sorafenib enhances proteasome inhibitor-induced cell death via inactivation of Akt and stress-activated protein kinases. J Gastroenterol 49, 517–526 (2014). https://doi.org/10.1007/s00535-013-0796-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-013-0796-z