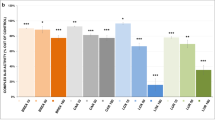
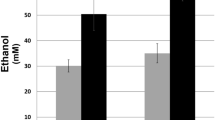
Abstract
In aripiprazole-treated PC12 cells, we previously showed that the mitochondrial membrane potential (Δψm) was rather increased in spite of lowered cytochrome c oxidase activity. To address these inconsistent results, we focused the NADPH generation by glucose-6-phosphate dehydrogenase (G6PD), a rate-limiting enzyme of the pentose phosphate pathway (PPP), to titrate reactive oxygen species (ROS) that results in the Δψm maintenance. G6PD may be also involved in another inconsistent result of lowered intracellular lactate level in aripiprazole-treated PC12 cells, because PPP competes glucose-6-phosphate with the glycolytic pathway, resulting in the downregulation of glycolysis. Therefore, we assayed intracellular amounts of NADPH, ROS, and the activities of the enzymes generating or consuming NADPH (G6PD, NADP+-dependent isocitrate dehydrogenase, NADP+-dependent malic enzyme, glutathione reductase, and NADPH oxidase [NOX]) and estimated glycolysis in 50 μM aripiprazole-, clozapine-, and haloperidol-treated PC12 cells. NADPH levels were enhanced only in aripiprazole-treated ones. Only haloperidol increased ROS. However, the enzyme activities did not show significant changes toward enhancing NADPH level except for the aripiprazole-induced decrease in NOX activity. Thus, the lowered NOX activity could have contributed to the aripiprazole-induced increase in the NADPH level by lowering ROS generation, resulting in maintained Δψm. Although the aforementioned assumption was invalid, the ratio of fructose-1,6-bisphosphate to fructose-6-phosphate was decreased by all antipsychotics examined. Pyruvate kinase activity was enhanced only by aripiprazole. In summary, these observations indicate that aripiprazole possibly possesses the pharmacological superiority to clozapine and haloperidol in the ROS generation and the adjustment of glycolytic pathway.






Similar content being viewed by others
Abbreviations
- AAPD:
-
Atypical antipsychotic drug
- ACC:
-
Acetyl-CoA carboxylase
- AMPK:
-
AMP-activated protein kinase
- COX:
-
Cytochrome c oxidase
- Δψm :
-
Mitochondrial membrane potential
- DCF-DA:
-
2′,7′-dichlorofluorescein diacetate
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- F1,6P:
-
Fructose-1,6-bisphosphate
- F2,6P:
-
Fructose-2,6-bisphosphate
- F6P:
-
Fructose-6-phosphate
- G6P:
-
Glucose-6-phosphate
- G6PD:
-
Glucose-6-phosphate dehydrogenase
- GR:
-
Glutathione reductase
- GSH:
-
Reduced form glutathione
- H6PD:
-
Hexose-6-phosphate dehydrogenase
- HPRT:
-
Hypoxanthine-guanine phosphoribosyltransferase
- LDH:
-
Lactate dehydrogenase
- NADP+-IDH:
-
NADP+-dependent isocitrate dehydrogenase
- NADP+-ME:
-
NADP+-dependent malic enzyme
- NOX:
-
NADPH oxidase
- O ·−2 :
-
Superoxide
- PFK1:
-
Phosphofructokinase-1
- PFKBP:
-
6-Phosphofructo-2-kinase/fructose-2,6-bisphosphatase
- PPi-PFK:
-
Pyrophosphate-dependent F6P kinase
- PPP:
-
Pentose phosphate pathway
- q-RT-PCR:
-
Quantitative real-time PCR
- R5P:
-
Ribose-5-phosphate
- ROS:
-
Reactive oxygen species
- Taldo1:
-
Transaldolase 1
- TIGAR:
-
TP53-induced glycolysis and apoptosis regulator
- Tkt:
-
Transketolase
References
Banno K, Fujioka T, Kikuchi T, Oshiro Y, Hiyama T, Nakagawa K (1988) Studies on 2(1H)-quinolinone derivatives as neuroleptic agents I. Synthesis and biological activities of (4-Phenyl-1-piperazinyl)propoxy-2(1H)-quinolinone derivatives. Chem Pharm Bull 36:4377–4388
Barja G, Herrero AJ (1998) Localization at complex I and mechanism of the higher free radical production of brain nonsynaptic mitochondria in the short-lived rat than in the longevous pigeon. J Bioenerg Biomembr 30:235–243
Bensaad K, Tsuruta A, Selak MA, Vidal MN, Nakano K, Bartrons R, Gottlieb E, Vousden KH (2006) TIGAR, a p53-inducible regulator of glycolysis and apoptosis. Cell 126:107–120
Berg JM, Tymoczko JL, Stryer L (2007) Biochemistry. WH Freeman and Company, New York
Betz A, Chance B (1965) Phase relationship of glycolytic intermediates in yeast cells with oscillatory metabolic control. Arch Biochem Biophys 109:585–594
Blair JB, Walker RG (1984) Rat liver pyruvate kinase: influence of ligands on activity and fructose 1,6-bisphosphate binding. Arch Biochem Biophys 232:202–213
Chandra FA, Buzi G, Doyle JC (2011) Glycolytic oscillations and limits on robust efficiency. Science 833:187–192
Chesney J, Mitchell R, Benigni F, Bacher M, Spiegel L, Al-Abed Y, Han JH, Metz C, Bucala R (1999) An inducible gene product for 6-phosphofructo-2-kinase with an AU-rich instability element: Role in tumor cell glycolysis and the Warburg effect. Proc Natl Acad Sci USA 96:3047–3052
Cui XL, Douglas JG (1997) Arachidonic acid activates c-jun N-terminal kinase through NADPH oxidase in rabbit proximal tubular epithelial cells. Proc Natl Acad Sci USA 94:3771–3776
Dombrauckas JD, Santarsiero BD, Mesecar AD (2005) Structural basis for tumor pyruvate kinase M2 allosteric regulation and catalysis. Biochemistry 44:9417–9429
El-Maghrabi MR, Claus TH, McGrane MM, Pilkis SJ (1982) Influence of phosphorylation on the interaction of effectors with rat liver pyruvate kinase. J Biol Chem 257:233–240
Haas A, Goebel W (1992) Microbial strategies to prevent oxygen-dependent killing by phagocytes. Free Radic Res Commun 16:137–157
Herrero-Mendez A, Almeida A, Fernández E, Maestre C, Moncada S, Bolaños JP (2009) The bioenergetic and antioxidant status of neurons is controlled by continuous degradation of a key glycolytic enzyme by APC/C-Cdh1. Nat Cell Biol 11:747–752
Jeon S-M, Chandel NS, Hay N (2012) AMPK regulates NADPH homeostasis to promote tumour cell survival during energy stress. Nature 485:661–665
Jurica MS, Mesecar A, Heath PJ, Shi W, Nowak T, Stoddard BL (1998) The allosteric regulation of pyruvate kinase by fructose-1,6-bisphosphate. Structure 6:195–210
Kato TA, Monji A, Yasukawa K, Mizoguchi Y, Horikawa H, Seki Y, Hashioka S, Han YH, Kasai M, Sonoda N, Hirata E, Maeda Y, Inoguchi T, Utsumi H, Kanba S (2011) Aripiprazole inhibits superoxide generation from phorbol-myristate-acetate (PMA)-stimulated microglia in vitro: implication for antioxidative psychotropic actions via microglia. Scizophr Res 129:172–182
Kawaguchi T, Veech RL, Uyeda K (2001) Regulation of energy metabolism in macrophages during hypoxia. Roles of fructose 2,6-bisphosphate and ribose 1,5-bisphosphate. J Biol Chem 276:28554–28561
Kemp RG, Foe LG (1983) Allosteric regulatory properties of muscle phosphofructokinase. Mol Cell Biochem 57:147–154
Kikuchi T, Tottori K, Uwahodo Y, Hirose T, Miwa T, Oshiro Y, Morita S (1995) 7-{4-[4-(2,3-Dichlorophenyl)-1-piperazinyl]butyloxy}-3,4-dihydro-2(1H)-quinolinone (OPC-14597), a new putative antipsychotic drug with both presynaptic dopamine autoreceptor agonistic activity and postsynaptic D2 receptor antagonistic activity. J Pharmacol Exp Ther 274:329–336
Kletzien RF, Harris PK, Foellmi LA (1994) Glucose-6-phosphate dehydrogenase: a “housekeeping” enzyme subject to tissue-specific regulation by hormones, nutrients, and oxidant stress. FASEB J 8:174–181
Kussmaul L, Hirst J (2006) The mechanism of superoxide production by NADH:ubiquinone oxidoreductase (complex I) from bovine heart mitochondria. Proc Natl Acad Sci USA 103:7607–7612
Manzano A, Rosa JL, Ventura F, Perez JX, Nadal M, Estivill X, Ambrosio S, Gil J, Bartrons R (1998) Molecular cloning, expression, and chromosomal localization of a ubiquitously expressed human 6-phosphofructo-2-kinase/fructose-2, 6-bisphosphatase gene (PFKFB3). Cytogenet Cell Genet 83:214–217
Marsin AS, Bertrand L, Rider MH, Deprez J, Beauloye C, Vinvent MF, Van den Berghe G, Carling D, Hue L (2000) Phosphorylation and activation of heart PFK-2 by AMPK has a role in the stimulation of glycolysis during ischaemia. Curr Biol 10:1247–1255
Marsin AS, Bouzin C, Bertrand L, Hue L (2002) The stimulation of glycolysis by hypoxia in activated monocytes is mediated by AMP-activated protein kinase and inducible 6-phosphofructo-2-kinase. J Biol Chem 277:30778–30783
Michal G (1986a) d-Glucose 6-phosphate and d-fructose 6-phosphate. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Academic Press, New York, pp 191–198
Michal G (1986b) d-Fructose 1,6-bisphosphate, dihydroxyacetone phosphate and d-glyceroaldehyde 3-phosphate. In: Bergmeyer HU (ed) Methods of Enzymatic Analysis. Academic Press, New York, pp 342–350
Noh KM, Koh JY (2000) Induction and activation by zinc of NADPH oxidase in cultured cortical neurons and astrocytes. J Neurosci 20:RC111
Oshiro Y, Sato S, Kurahashi N, Tanaka T, Kikuchi T, Tottori K, Uwahodo Y, Nishi T (1998) Novel antipsychotic agents with dopamine autoreceptor agonist properties: synthesis and pharmacology of 7-[4-(4-Phenyl-1-piperazinyl)butoxy]-3,4-dihydro-2(1H)-quinolinone derivatives. J Med Chem 41:658–667
Ota A, Nakashima A, Kaneko YS, Mori K, Nagasaki H, Takayanagi T, Itoh M, Kondo K, Nagatsu T, Ota M (2012) Effects of aripiprazole and clozapine on the treatment of glycolytic carbon in PC12 cells. J Neural Transm 119:1327–1342
Ozols J (1993) Isolation and the complete amino acid sequence of lumenal endoplasmic reticulum glucose-6-phosphate dehydrogenase. Proc Natl Acad Sci USA 90:5302–5306
Senesi S, Csala M, Marcolongo P, Fulceri R, Mandl J, Bánhegyi G, Benedetti A (2010) Hexose-6-phosphate dehydrogenase in the endoplasmic reticulum. Biol Chem 391:1–8
Seo BB, Marella M, Yagi T, Matsuno-Yagi A (2006) The single subunit NADH dehydrogenase reduces generation of reactive oxygen species from complex I. FEBS Lett 580:6105–6108
Sols A (1981) Multimodulation of enzyme activity. Curr Top Cell Regul 19:77–101
Suzukawa K, Miura K, Mitsushita J, Resau J, Hirose K, Crystal R, Kamata T (2000) Nerve growth factor-induced neuronal differentiation requires generation of Rac1-regulated reactive oxygen species. J Biol Chem 275:13175–13178
Takami G, Ota M, Nakashima A, Kaneko YS, Mori K, Nagatsu T, Ota A (2010) Effects of atypical antipsychotics and haloperidol on PC12 cells: only aripiprazole phosphorylates AMP-activated protein kinase. J Neural Transm 117:1139–1153
Tammariello SP, Quinn MT, Estus S (2000) NADPH oxidase contributes directly to oxidative stress and apoptosis in nerve growth factor-deprived sympathetic neurons. J Neurosci 20:RC53
Termonia Y, Ross J (1981) Oscillations and control features in glycolysis: numerical analysis of a comprehensive model. Proc Natl Acad Sci USA 78:2952–2956
Tornheim K, Lowenstein JM (1976) Control of phosphofructokinase from rat skeletal muscle. Effects of fructose diphosphate, AMP, ATP, and citrate. J Biol Chem 251:7322–7328
Underwood AH, Newsholme EA (1965) Properties of phosphofructokinase from rat liver and their relation to the control of glycolysis and gluconeogenesis. Biochem J 95:868–875
Uyeda K (1979) Phosphofructokinase. Adv Enzymol Relat Areas Mol Biol 48:193–124
Van Schaftingen E (1987) Fructose 2,6-bisphosphate. Adv Enzymol Relat Areas Mol Biol 59:315–395
Van Schaftingen E, Lederer B, Bartrons R, Hers HG (1982) A kinetic study of pyrophosphate: fructose-6-phosphate phosphotransferase from potato tubers. Application to a microassay of fructose 2,6-bisphosphate. Eur J Biochem 129:191–195
Vaz AR, Delgado-Esteban M, Brito MA, Bolaños JP, Brites D, Almeida A (2010) Bilirubin selectively inhibits cytochrome c oxidase activity and induces apoptosis in immature cortical neurons: assessment of the protective effects of glycoursodeoxycholic acid. J Neurochem 112:56–65
Vora S, Oskam R, Staal GE (1985) Isoenzymes of phosphofructokinase in the rat: demonstration of the three non-identical subunits by biochemical, immunochemical and kinetic studies. Biochem J 229:333–341
Wu R, Racker E (1959) Regulatory mechanism in carbohydrate metabolism: IV Pasteur effect and crabtree effect in ascites tumor cells. J Biol Chem 234:1036–1041
Yalcin A, Clem BF, Simmons A, Lane A, Nelson K, Clem AL, Brock E, Siow D, Wattenberg B, Telang S, Chesney J (2009) Nuclear targeting of 6-phosphofructo-2-kinase (PFKFB3) increases proliferation via cyclin-dependent kinases. J Biol Chem 284:24223–24232
Ying H, Kimmelman AC, Lyssiotis CA, Hua S, Chu GC, Fletcher-Sananikone E, Locasale JW, Son J, Zhang H, Coloff JL, Yan H, Wang W, Chen S, Viale A, Zheng H, Paik JH, Lim C, Guimaraes AR, Martin ES, Chang J, Hezel AF, Perry SR, Hu J, Gan B, Xiao Y, Asara JM, Weissleder R, Wang YA, Chin L, Cantley LC, DePinho RA (2012) Oncogenic Kras maintains pancreatic tumors through regulation of anabolic glucose metabolism. Cell 149:656–670
Acknowledgments
This work was supported by grants-in-aid from Fujita Health University to AO.
Conflict of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nagasaki, H., Nakashima, A., Kaneko, Y.S. et al. Aripiprazole increases NADPH level in PC12 cells: the role of NADPH oxidase. J Neural Transm 121, 91–103 (2014). https://doi.org/10.1007/s00702-013-1075-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-013-1075-0