Abstract
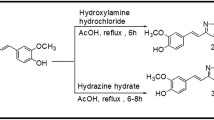
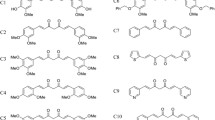
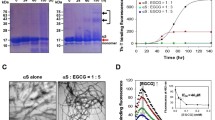
Curcumin, a dietary polyphenol, has shown a potential to act on the symptoms of neurodegenerative disorders, including Alzheimer’s and Parkinson’s diseases, as a consequence of its antioxidant, anti-inflammatory and anti-protein aggregation properties. Unfortunately, curcumin undergoes rapid degradation at physiological pH into ferulic acid, vanillin and dehydrozingerone, making it an unlikely drug candidate. Here, we evaluated the ability of some curcumin by-products: dehydrozingerone (1), its O-methyl derivative (2), zingerone (3), and their biphenyl analogues (4–6) to interact with α-synuclein (AS), using CD and fluorescence spectroscopy. In addition, the antioxidant properties and the cytoprotective effects in rat pheochromocytoma (PC12) cells prior to intoxication with H2O2, MPP+ and MnCl2 were examined while the Congo red assay was used to evaluate the ability of these compounds to prevent aggregation of AS. We found that the biphenyl zingerone analogue (6) interacts with high affinity with AS and also displays the best antioxidant properties while the biphenyl analogues of dehydrozingerone (4) and of O-methyl-dehydrozingerone (5) are able to partially inhibit the aggregation process of AS, suggesting the potential role of a hydroxylated biphenyl scaffold in the design of AS aggregation inhibitors.











Similar content being viewed by others
References
Bartels T, Choi JG, Selkoe DJ (2011) alpha-Synuclein occurs physiologically as a helically folded tetramer that resists aggregation. Nature 477:107–111
Baures PW, Peterson SA, Kelly JW (1998) Discovering transthyretin amyloid fibril inhibitors by limited screening. Bioorg Med Chem 6:1389–1401
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci Technol. 28:25–30
Delogu G, Fabbri D, Dettori MA, Forni A, Casalone G (2000) Chiral nonracemic C2-symmetry biphenyls by desymmetrization of 6,6′,2,2′-tetramethoxy-1,1′-biphenyl. Tetrahedron Asymmetry 11:4417–4427
Delomenede M, Bedos-Belval F, Duran H, Vindis C, Baltas M, Negre-Salvayre A (2008) Development of novel antiatherogenic biaryls: design, synthesis, and reactivity. J Med Chem 51:3171–3181
Elias G, Rao MNA (1988) Synthesis and anti-inflammatory activity of substituted (e)-4-phenyl-3-buten-2-ones. Eur J Med Chem 23:379–380
Hauser DN, Hastings TG (2013) Mitochondrial dysfunction and oxidative stress in Parkinson’s disease and monogenic parkinsonism. Neurobiol Dis 51:35–42
Hong DP, Fink AL, Uversky VN (2008) Structural characteristics of alpha-synuclein oligomers stabilized by the flavonoid baicalein. J Mol Biol 383:214–223
Howie AJ, Brewer DB (2009) Optical properties of amyloid stained by Congo red: history and mechanisms. Micron 40:285–301
Huang CJ, Ren GP, Zhou H, Wang CC (2005) A new method for purification of recombinant human alpha-synuclein in Escherichia coli. Protein Expr Purif 42:173–177
Jiang M, Xie MX, Zheng D, Liu Y, Li XY, Chen X (2004) Spectroscopic studies on the interaction of cinnamic acid and its hydroxyl derivatives with human serum albumin. J Mol Struct 692:71–80
Liu YZ, Dolence J, Ren J, Rao MNA, Sreejayan N (2008) Inhibitory effect of dehydrozingerone on vascular smooth muscle cell function. J Cardiovasc Pharmacol 52:422–429
Marques FA, Simonelli F, Oliveira ARM, Gohr GL, Leal PC (1998) Oxidative coupling of 4-substituted 2-methoxy phenols using methyltributylammonium permanganate in dichloromethane. Tetrahedron Lett 39:943–946
Montalti M, Credi A, Prodi L, Gandolfi MT (2006) Handbook of photochemistry. CRC Press, Taylor & Francis Group, Boca Raton, pp 561–600
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Naiki H, Nagai Y (2009) Molecular pathogenesis of protein misfolding diseases: pathological molecular environments versus quality control systems against misfolded proteins. J Biochem 146:751–756
Narender T, Reddy KP (2007) A simple and highly efficient method for the synthesis of chalcones by using borontrifluoride-etherate. Tetrahedron Lett 48:3177–3180
Ono K, Yamada M (2006) Antioxidant compounds have potent anti-fibrillogenic and fibril-destabilizing effects for alpha-synuclein fibrils in vitro. J Neurochem 97:105–115
Oueslati A, Fournier M, Lashuel HA (2010) Role of post-translational modifications in modulating the structure, function and toxicity of alpha-synuclein: implications for Parkinson’s disease pathogenesis and therapies. In: Bjorklund A, Cenci M (eds) Recent Advances in Parkinsons Disease: Basic Research. Elsevier Science BV, Amsterdam, pp 115–145
Parihar VK, Dhawan J, Kumar S, Manjula SN, Subramanian G, Unnikrishnan MK, Rao CM (2007) Free radical scavenging and radioprotective activity of dehydrozingerone against whole body gamma irradiation in Swiss albino mice. Chem Biol Interact 170:49–58
Pisano M, Pagnan G, Dettori MA, Cossu S, Caffa I, Sassu I, Emionite L, Fabbri D, Cilli M, Pastorino F, Palmieri G, Delogu G, Ponzoni M, Rozzo C (2010) Enhanced anti-tumor activity of a new curcumin-related compound against melanoma and neuroblastoma cells. Mol Cancer 9:137–148
Priyadarsini KI, Guha SN, Rao MNA (1998) Physico-chemical properties and antioxidant activities of methoxy phenols. Free Radic Biol Med 24:933–941
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26:1231–1237
Roth J (2009) Are there common biochemical and molecular mechanisms controlling manganism and parkisonism. NeuroMol Med 11:281–296
Schildknecht S, Gerding HR, Karreman C, Drescher M, Lashuel HA, Outeiro TF, Di Monte DA, Leist M (2013) Oxidative and nitrative alpha-synuclein modifications and proteostatic stress: implications for disease mechanisms and interventions in synucleinopathies. J Neurochem. doi:10.1111/jnc.12226
Siddiqui MA, Kashyap MP, Kumar V, Tripathi VK, Khanna VK, Yadav S, Pant AB (2011) Differential protection of pre-, co- and post-treatment of curcumin against hydrogen peroxide in PC12 cells. Hum Exp Toxicol 30:192–198
Wang YJ, Pan MH, Cheng AL, Lin LI, Ho YS, Hsieh CY, Lin JK (1997) Stability of curcumin in buffer solutions and characterization of its degradation products. J Pharm Biomed Anal 15:1867–1876
Wang MS, Boddapati S, Emadi S, Sierks MR (2010) Curcumin reduces alpha-synuclein induced cytotoxicity in Parkinson’s disease cell model. BMC Neurosci 11:57
Witting PK, Westerlund C, Stocker R (1996) A rapid and simple screening test for potential inhibitors of tocopherol-mediated peroxidation of LDL lipids. J Lipid Res 37:853–867
Xiang W, Schlachetzki JC, Helling S, Bussmann JC, Berlinghof M, Schäffer TE, Marcus K, Winkler J, Klucken J, Becker CM (2013) Oxidative stress-induced posttranslational modifications of alpha-synuclein: specific modification of alpha-synuclein by 4-hydroxy-2-nonenal increases dopaminergic toxicity. Mol Cell Neurosci 54:71–83
Xu J, Wei CZ, Xu CQ, Bennett MC, Zhang GH, Li FC, Tao EX (2007) Rifampicin protects PC12 cells against MPP+-induced apoptosis and inhibits the expression of an alpha-Synuclein multimer. Brain Res 1139:220–225
Zhou HY, Beevers CS, Huang SL (2011) The Targets of Curcumin. Curr Drug Targets 12:332–347
Acknowledgments
This work was supported by Sardinia Autonomous Region, L.R. 7th August 2007, no. 7; and by FIRB-MERIT Program “Molecular bases in ageing-related degenerative syndromes” (RBNE08HWLZ).
Conflict of interest
The authors declare no competing financial interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marchiani, A., Mammi, S., Siligardi, G. et al. Small molecules interacting with α-synuclein: antiaggregating and cytoprotective properties. Amino Acids 45, 327–338 (2013). https://doi.org/10.1007/s00726-013-1503-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-013-1503-3