Abstract
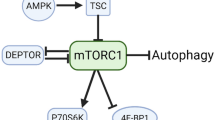
Amino acids are potent regulators of muscle protein synthesis and breakdown and have received considerable attention for the treatment of muscle wasting conditions. Arginine is critically involved in numerous physiological functions including providing substrate for the production of creatine, urea and nitric oxide (NO) and in the synthesis of new proteins. However, little is known about the direct effects of arginine on skeletal muscle protein synthesis during catabolic conditions. The aims of this study were to determine whether exogenous arginine could protect skeletal muscle cells from wasting directly and whether this effect was dependent on production of NO and/or activation of the rapamycin-sensitive mechanistic target of rapamycin complex 1 (mTORC1) signalling pathway. To explore these aims, we deprived mature C2C12 myotubes from nutrients and growth factors by incubating them in HEPES buffered saline with arginine or equimolar concentrations of alanine (control). Our results show that arginine: increased the ratio of phosphorylated to total mTOR (146 %), S6 (40 %) and 4EBP1 (69 %); increased protein synthesis (69 %) during the first hour of treatment; and increased myotube diameter by ~15 %. Experiments using the NO synthase inhibitor l-NG-Nitroarginine Methyl Ester showed a NO-independent protection from muscle wasting. On the other hand, the mTORC1 inhibitor rapamycin prevented increases in phosphorylated S6, protein synthesis and myotube diameter. The activation of mTORC1 and protein synthesis by arginine was not associated with changes in the phosphorylation status of Akt, but rather increased the expression of the amino acid-sensitive type III PI3-kinase Vps34 signalling protein. These data support a direct role for arginine in the regulation of mTORC1 in skeletal muscle.






Similar content being viewed by others
References
Anderson JE (2000) A role for nitric oxide in muscle repair: nitric oxide-mediated activation of muscle satellite cells. Mol Biol Cell 11(5):1859–1874
Atherton PJ, Smith K, Etheridge T, Rankin D, Rennie MJ (2010) Distinct anabolic signalling responses to amino acids in C2C12 skeletal muscle cells. Amino Acids 38(5):1533–1539. doi:10.1007/s00726-009-0377-x
Bauchart-Thevret C, Cui L, Wu G, Burrin DG (2010) Arginine-induced stimulation of protein synthesis and survival in IPEC-J2 cells is mediated by mTOR but not nitric oxide. Am J Physiol Endocrinol Metab 299(6):E899–E909. doi:10.1152/ajpendo.00068.2010
Choo AY, Yoon SO, Kim SG, Roux PP, Blenis J (2008) Rapamycin differentially inhibits S6Ks and 4E-BP1 to mediate cell-type-specific repression of mRNA translation. Proc Natl Acad Sci USA 105(45):17414–17419. doi:10.1073/pnas.0809136105
Collier SR, Casey DP, Kanaley JA (2005) Growth hormone responses to varying doses of oral arginine. Growth Horm IGF Res 15(2):136–139. doi:10.1016/j.ghir.2004.12.004
Corradetti MN, Guan KL (2006) Upstream of the mammalian target of rapamycin: do all roads pass through mTOR? Oncogene 25(48):6347–6360. doi:10.1038/sj.onc.1209885
Efeyan A, Zoncu R, Sabatini DM (2012) Amino acids and mTORC1: from lysosomes to disease. Trends Mol Med 18(9):524–533. doi:10.1016/j.molmed.2012.05.007
Evans WJ (1995) What is sarcopenia? J Gerontol A Biol Sci Med Sci 50:spec no 5–8
Evans RW, Fernstrom JD, Thompson J, Morris SM Jr, Kuller LH (2004) Biochemical responses of healthy subjects during dietary supplementation with l-arginine. J Nutr Biochem 15(9):534–539. doi:10.1016/j.jnutbio.2004.03.005
Goodman CA, Mabrey DM, Frey JW, Miu MH, Schmidt EK, Pierre P, Hornberger TA (2011) Novel insights into the regulation of skeletal muscle protein synthesis as revealed by a new nonradioactive in vivo technique. Faseb J 25(3):1028–1039. doi:10.1096/fj.10-168799
Goto C, Nishioka K, Umemura T, Jitsuiki D, Sakagutchi A, Kawamura M, Chayama K, Yoshizumi M, Higashi Y (2007) Acute moderate-intensity exercise induces vasodilation through an increase in nitric oxide bioavailability in humans. Am J Hypertens 20(8):825–830. doi:10.1016/j.amjhyper.2007.02.014
Hara K, Yonezawa K, Weng QP, Kozlowski MT, Belham C, Avruch J (1998) Amino acid sufficiency and mTOR regulate p70 S6 kinase and eIF-4E BP1 through a common effector mechanism. J Biol Chem 273(23):14484–14494
Herningtyas EH, Okimura Y, Handayaningsih AE, Yamamoto D, Maki T, Iida K, Takahashi Y, Kaji H, Chihara K (2008) Branched-chain amino acids and arginine suppress MaFbx/atrogin-1 mRNA expression via mTOR pathway in C2C12 cell line. Biochim Biophys Acta 1780(10):1115–1120. doi:10.1016/j.bbagen.2008.06.004
Jewell JL, Russell RC, Guan KL (2013) Amino acid signalling upstream of mTOR. Nat Rev Mol Cell Biol 14(3):133–139. doi:10.1038/nrm3522
Jun T, Wennmalm A (1994) NO-dependent and -independent elevation of plasma levels of insulin and glucose in rats by l-arginine. Br J Pharmacol 113(2):345–348
Kim E, Goraksha-Hicks P, Li L, Neufeld TP, Guan KL (2008) Regulation of TORC1 by Rag GTPases in nutrient response. Nat Cell Biol 10(8):935–945. doi:10.1038/ncb1753
Koh TJ, Tidball JG (1999) Nitric oxide synthase inhibitors reduce sarcomere addition in rat skeletal muscle. J Physiol 519(Pt 1):189–196
Koopman R, van Loon LJ (2009) Aging, exercise, and muscle protein metabolism. J Appl Physiol (1985) 106(6):2040–2048. doi:10.1152/japplphysiol.91551.2008
Koopman R, Wagenmakers AJ, Manders RJ, Zorenc AH, Senden JM, Gorselink M, Keizer HA, van Loon LJ (2005) Combined ingestion of protein and free leucine with carbohydrate increases postexercise muscle protein synthesis in vivo in male subjects. Am J Physiol Endocrinol Metab 288(4):E645–E653. doi:10.1152/ajpendo.00413.2004
Koopman R, Ly CH, Ryall JG (2014) A metabolic link to skeletal muscle wasting and regeneration. Front Physiol 5:32. doi:10.3389/fphys.2014.00032
Long JH, Lira VA, Soltow QA, Betters JL, Sellman JE, Criswell DS (2006) Arginine supplementation induces myoblast fusion via augmentation of nitric oxide production. J Muscle Res Cell Motil 27(8):577–584. doi:10.1007/s10974-006-9078-1
Lynch GS, Schertzer JD, Ryall JG (2007) Therapeutic approaches for muscle wasting disorders. Pharmacol Ther 113(3):461–487
Merry TL, Steinberg GR, Lynch GS, McConell GK (2010) Skeletal muscle glucose uptake during contraction is regulated by nitric oxide and ROS independently of AMPK. Am J Physiol Endocrinol Metab 298(3):E577–E585. doi:10.1152/ajpendo.00239.2009
Moinard C, Cynober L (2007) Citrulline: a new player in the control of nitrogen homeostasis. J Nutr 137(6 Suppl 2):1621S–1625S (pii 137/6/1621S)
Nobukuni T, Joaquin M, Roccio M, Dann SG, Kim SY, Gulati P, Byfield MP, Backer JM, Natt F, Bos JL, Zwartkruis FJ, Thomas G (2005) Amino acids mediate mTOR/raptor signaling through activation of class 3 phosphatidylinositol 3OH-kinase. Proc Natl Acad Sci USA 102(40):14238–14243. doi:10.1073/pnas.0506925102
Sarbassov DD, Ali SM, Sengupta S, Sheen JH, Hsu PP, Bagley AF, Markhard AL, Sabatini DM (2006) Prolonged rapamycin treatment inhibits mTORC2 assembly and Akt/PKB. Mol Cell 22(2):159–168. doi:10.1016/j.molcel.2006.03.029
Sellman JE, DeRuisseau KC, Betters JL, Lira VA, Soltow QA, Selsby JT, Criswell DS (2006) In vivo inhibition of nitric oxide synthase impairs upregulation of contractile protein mRNA in overloaded plantaris muscle. J Appl Physiol (1985) 100(1):258–265. doi:10.1152/japplphysiol.00936.2005
Smith LW, Smith JD, Criswell DS (2002) Involvement of nitric oxide synthase in skeletal muscle adaptation to chronic overload. J Appl Physiol (1985) 92(5):2005–2011. doi:10.1152/japplphysiol.00950.2001
Tangphao O, Grossmann M, Chalon S, Hoffman BB, Blaschke TF (1999) Pharmacokinetics of intravenous and oral l-arginine in normal volunteers. Br J Clin Pharmacol 47(3):261–266
Wu G, Morris SM Jr (1998) Arginine metabolism: nitric oxide and beyond. Biochem J 336(Pt 1):1–17
Acknowledgments
This work was funded by a grant from the Ajinomoto Amino Acid Research Program (3ARP: 09/07).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ham, D.J., Caldow, M.K., Lynch, G.S. et al. Arginine protects muscle cells from wasting in vitro in an mTORC1-dependent and NO-independent manner. Amino Acids 46, 2643–2652 (2014). https://doi.org/10.1007/s00726-014-1815-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-014-1815-y