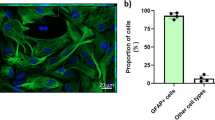
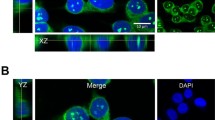
Abstract
l-Carnosine (β-alanyl-l-histidine) is a well-known antioxidant and neuroprotector in various models on animals and cell cultures. However, while there is a plethora of data demonstrating its efficiency as a neuroprotector, there is a distinct lack of data regarding the mechanism of its take up by neurons. According to literature, cultures of rat astrocytes, SKPT cells and rat choroid plexus epithelial cells take up carnosine via the H+-coupled PEPT2 membrane transporter. We’ve assessed the effectiveness and mechanism of carnosine transport, and its stability in primary rat cortical culture neurons. We demonstrated that neurons take up carnosine via active transport with Km = 119 μM and a maximum velocity of 0.289 nmol/mg (prot)/min. Passive transport speed constituted 0.21∙10–4 nmol/mg (prot)/min (with 119 μM concentration in the medium)—significantly less than active transport speed. However, carnosine concentrations over 12.5 mM led to passive transport speed becoming greater than active transport speed. Using PEPT2 inhibitor zofenopril, we demonstrated that PEPT2-dependent transport is one of the main modes of carnosine take up by neurons. Our experiments demonstrated that incubation with carnosine does not affect PEPT2 amount present in culture. At the same time, after removing carnosine from the medium, its elimination speed by culture cells reached 0.035 nmol/mg (prot)/min, which led to a decrease in carnosine quantity to control levels in culture within 1 h. Thus, carnosine is taken up by neurons with an effectiveness comparable to that of other PEPT2 substrates, but its elimination rate suggests that for effective use as a neuroprotector it’s necessary to either maintain a high concentration in brain tissue, or increase the effectiveness of glial cell synthesis of endogenous carnosine and its shuttling into neurons, or use more stable chemical modifications of carnosine.





Similar content being viewed by others
References
Agu R, Cowley E, Shao D, Macdonald C, Kirkpatrick D, Renton K, Massoud E (2011) Proton-coupled oligopeptide transporter (POT) family expression in human nasal epithelium and their drug transport potential. Mol Pharm 8:664–672
Alghamdi OA, King N, Jones GL, Moens PDJ (2017) Kinetic measurements of di- and tripeptide and peptidomimetic drug transport in different kidney regions using the fluorescent membrane potential-sensitive dye, DiS-C3-(3). J Membr Biol 250:641–649
Bellia F, Vecchio G, Rizzarelli E (2014) Carnosinases, their substrates and diseases. Molecules 19:2299–2329
Berezhnoy DS, Stvolinsky SL, Lopachev AV, Devyatov AA, Lopacheva OM, Kulikova OI, Abaimov DA, Fedorova TN (2019) Carnosine as an effective neuroprotector in brain pathology and potential neuromodulator in normal conditions. Amino Acids 51:139–150
Bermudez ML, Seroogy KB, Genter MB (2019) Evaluation of carnosine intervention in the Thy1-aSyn mouse model of Parkinson’s disease. Neuroscience 411:270–278
Boldyrev A, Abe H (1999) Metabolic transformation of neuropeptide carnosine modifies its biological activity. Cell Mol Neurobiol 19:163–175
Boldyrev A, Fedorova T, Stepanova M, Dobrotvorskaya I, Kozlova E, Boldanova N, Bagyeva G, Ivanova-Smolenskaya I, Illarioshkin S (2008) Carnosine [corrected] increases efficiency of DOPA therapy of Parkinson’s disease: a pilot study. Rejuvenation Res 11:821–827
Boldyrev AA, Aldini G, Derave W (2013) Physiology and pathophysiology of carnosine. Physiol Rev 93:1803–1845
Chistyakov DV, Azbukina NV, Lopachev AV, Kulichenkova KN, Astakhova AA, Sergeeva MG (2018) Rosiglitazone as a modulator of TLR4 and TLR3 signaling pathways in rat primary neurons and astrocytes. Int J Mol Sci. https://doi.org/10.3390/ijms19010113
Chu XP, Grasing KA, Wang JQ (2014) Acid-sensing ion channels contribute to neurotoxicity. Transl Stroke Res 5:69–78
De Marchis S, Melcangi RC, Modena C, Cavaretta I, Peretto P, Agresti C, Fasolo A (1997) Identification of the glial cell types containing carnosine-related peptides in the rat brain. Neurosci Lett 237:37–40
Dieck ST, Heuer H, Ehrchen J, Otto C, Bauer K (1999) The peptide transporter PepT2 is expressed in rat brain and mediates the accumulation of the fluorescent dipeptide derivative beta-Ala-Lys-Nepsilon-AMCA in astrocytes. Glia 25:10–20
Ferrer I (2018) Oligodendrogliopathy in neurodegenerative diseases with abnormal protein aggregates: the forgotten partner. Prog Neurobiol 169:24–54
Fujita T, Kishida T, Okada N, Ganapathy V, Leibach FH, Yamamoto A (1999) Interaction of kyotorphin and brain peptide transporter in synaptosomes prepared from rat cerebellum: implication of high affinity type H+/peptide transporter PEPT2 mediated transport system. Neurosci Lett 271:117–120
Fujita T, Kishida T, Wada M, Okada N, Yamamoto A, Leibach FH, Ganapathy V (2004) Functional characterization of brain peptide transporter in rat cerebral cortex: identification of the high-affinity type H+/peptide transporter PEPT2. Brain Res 997:52–61
Hu Y, Ocheltree SM, Xiang J, Keep RF, Smith DE (2005) Glycyl-L-glutamine disposition in rat choroid plexus epithelial cells in primary culture: role of PEPT2. Pharm Res 22:1281–1286
Hu Y, Xie Y, Keep RF, Smith DE (2014) Divergent developmental expression and function of the proton-coupled oligopeptide transporters PepT2 and PhT1 in regional brain slices of mouse and rat. J Neurochem 129:955–965
Jappar D, Hu Y, Keep RF, Smith DE (2009) Transport mechanisms of carnosine in SKPT cells: contribution of apical and basolateral membrane transporters. Pharm Res 26:172–181
Knutter I, Wollesky C, Kottra G, Hahn MG, Fischer W, Zebisch K, Neubert RH, Daniel H, Brandsch M (2008) Transport of angiotensin-converting enzyme inhibitors by H+/peptide transporters revisited. J Pharmacol Exp Ther 327:432–441
Kumar VV, Pichon C, Refregiers M, Guerin B, Midoux P, Chaudhuri A (2003) Single histidine residue in head-group region is sufficient to impart remarkable gene transfection properties to cationic lipids: evidence for histidine-mediated membrane fusion at acidic pH. Gene Ther 10:1206–1215
Lopachev AV, Lopacheva OM, Abaimov DA, Koroleva OV, Vladychenskaya EA, Erukhimovich AA, Fedorova TN (2016) Neuroprotective effect of carnosine on primary culture of rat cerebellar cells under oxidative stress. Biochemistry Mosc 81:511–520
Lu H, Klaassen C (2006) Tissue distribution and thyroid hormone regulation of Pept1 and Pept2 mRNA in rodents. Peptides 27:850–857
McKenzie AT, Wang M, Hauberg ME, Fullard JF, Kozlenkov A, Keenan A, Hurd YL, Dracheva S, Casaccia P, Roussos P, Zhang B (2018) Brain cell type specific gene expression and co-expression network architectures. Sci Rep 8:8868
Park HS, Han KH, Shin JA, Park JH, Song KY, Kim DH (2014) The neuroprotective effects of carnosine in early stage of focal ischemia rodent model. J Korean Neurosurg Soc 55:125–130
Rybakova Y, Akkuratov E, Kulebyakin K, Brodskaya O, Dizhevskaya A, Boldyrev A (2012) Receptor-mediated oxidative stress in murine cerebellar neurons is accompanied by phosphorylation of MAP (ERK 1/2) kinase. Curr Aging Sci 5:225–230
Sariev AK, Abaimov DA, Tankevich MV, Pantyukhova EY, Prokhorov DI, Fedorova TN, Lopachev AV, Stvolinskii SL, Konovalova EV, Seifulla RD (2015) Experimental study of the basic pharmacokinetic characteristics of dipeptide carnosine and its efficiency of penetration into brain tissues. Eksp Klin Farmakol 78:30–35
Song F, Hu Y, Jiang H, Smith DE (2017) species differences in human and rodent PEPT2-mediated transport of glycylsarcosine and cefadroxil in pichia pastoris transformants. Drug Metab Dispos 45:130–136
Teuscher NS, Keep RF, Smith DE (2001) PEPT2-mediated take up of neuropeptides in rat choroid plexus. Pharm Res 18:807–813
Xiang J, Hu Y, Smith DE, Keep RF (2006) PEPT2-mediated transport of 5-aminolevulinic acid and carnosine in astrocytes. Brain Res 1122:18–23
Xiang J, Jiang H, Hu Y, Smith DE, Keep RF (2010) Kyotorphin transport and metabolism in rat and mouse neonatal astrocytes. Brain Res 1347:11–18
Yamashita T, Shimada S, Guo W, Sato K, Kohmura E, Hayakawa T, Takagi T, Tohyama M (1997) Cloning and functional expression of a brain peptide/histidine transporter. J Biol Chem 272:10205–10211
Yang NJ, Hinner MJ (2015) Getting across the cell membrane: an overview for small molecules, peptides, and proteins. Methods Mol Biol 1266:29–53
Yee SW, Buitrago D, Stecula A, Ngo HX, Chien HC, Zou L, Koleske ML, Giacomini KM (2020) Deorphaning a solute carrier 22 family member, SLC22A15, through functional genomic studies. FASEB J 34:15734–15752
Zhao J, Shi L, Zhang LR (2017) Neuroprotective effect of carnosine against salsolinol-induced Parkinson’s disease. Exp Ther Med 14:664–670
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
All procedures with animals were approved by the ethical committee of RSN, Moscow.
Consent to participate
No human studies were performed in this work.
Consent for publication
No human studies were performed in this work.
Additional information
Handling editor: W. Derave.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lopachev, A.V., Abaimov, D.A., Filimonov, I.S. et al. An assessment of the transport mechanism and intraneuronal stability of l-carnosine. Amino Acids 54, 1115–1122 (2022). https://doi.org/10.1007/s00726-021-03094-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-021-03094-5