Abstract
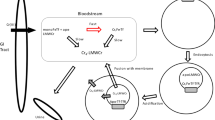
The mechanism and effectiveness of iron removal from transferrin by three series of new potential therapeutic iron sequestering agents have been analyzed with regard to the structures of the chelators. All compounds are hexadentate ligands composed of a systematically varied combination of methyl-3,2-hydroxypyridinone (Me-3,2-HOPO) and 2,3-dihydroxyterephthalamide (TAM) binding units linked to a polyamine scaffold through amide linkers; each series is based on a specific backbone: tris(2-aminoethyl)amine, spermidine, or 5-LIO(TAM), where 5-LIO is 2-(2-aminoethoxy)ethylamine. Rates of iron removal from transferrin were determined spectrophotometrically for the ten ligands, which all efficiently acquire ferric ion from diferric transferrin with a hyperbolic dependence on ligand concentration (saturation kinetics). The effect of the two iron-binding subunits Me-3,2-HOPO and TAM and of the scaffold structures on iron removal ability is discussed. At the low concentrations corresponding to therapeutic dose, TAM-containing ligands exhibit the fastest rates of iron removal, which correlates with their high affinity for ferric ion and suggests the insertion of such binding units into future therapeutic chelating agents. In addition, urea polyacrylamide gel electrophoresis was used to measure the individual microscopic rates of iron removal from the three iron-bound transferrin species (diferric transferrin, N-terminal monoferric transferrin, C-terminal monoferric transferrin) by the representative chelators 5-LIO(Me-3,2-HOPO)2(TAM) and 5-LIO(TAMmeg)2(TAM), where TAMmeg is 2,3-dihydroxy-1-(methoxyethylcarbamoyl)terephthalamide. Both ligands show preferential removal from the C-terminal site of the iron-binding protein. However, cooperative effects between the two binding sites differ with the chelator. Replacement of hydroxypyridinone moieties by terephthalamide groups renders the N-terminal site more accessible to the ligand and may represent an advantage for iron chelation therapy.








Similar content being viewed by others
Notes
The pM value is the negative logarithm of the free iron concentration in equilibrium with complexed and free ligand, at physiological pH (pH 7.4) with 1 μM total iron concentration and 10 μM total ligand concentration.
We repeat here the detailed solutions of the system of equations used in our analysis, since the mathematical model used in a recent study by Hissen and Moore [38] was based on erroneous solutions to the same integral equations. Caution should be used in interpreting the results of Hissen and Moore.
References
Lippard SJ, Berg JM (eds) (1994) Principles of bioinorganic chemistry. University Science Books, Mill Valley
Koppenol WH (2000) In: Badman DG, Bergeron RJ, Brittenham GM (eds) Iron chelators: new development strategies. The Saratoga Group, Ponte Vedra Beach, pp 3–10
Andrews NC (2000) Rev Clin Exp Hematol 4:283–301
Ponka P (1999) Kidney Int 55:S2–S11
Liu XF, Theil EC (2005) Acc Chem Res 38:167–175
Dertz EA, Raymond KN (2003) In: McCleverty J, Meyer T (eds) Comprehensive coordination chemistry II. Pergamon, Oxford, pp 141–168
Baker EN (1994) Adv Inorg Chem 41:389–463
Harris DC, Aisen P (1989) In: Loehr T (ed) Iron carriers and iron proteins. VCH, New York, pp 239–352
Otto B, Verweij-van Vught A, Maclaren D (1992) Crit Rev Microbiol 18:217–233
Pietrangelo A (2004) N Engl J Med 350:2383–2397
Thein SL (2004) Br J Haematol 124:264–274
Ballas SK (2002) Drugs 62:1143–1172
Olivieri NF, Brittenham GM (1997) Blood 89:739–761
Olivieri NF (2001) Semin Hematol 38:57–62
Porter JB, Huehns ER, Hider RC (1989) Baillieres Clin Haematol 2:257–292
Kontoghiorghes GJ, Pattichi K, Hadjigavriel M, Kolnagou A (2000) Transfus Sci 23:211–223
Kalinowski DS, Richardson DR (2005) Pharmaco Rev 57:547–583
O’Sullivan B, Xu J, Raymond Kenneth N (2000) In: Badman DG, Bergeron RJ, Brittenham GM (eds) Iron chelators: new development strategies. The Saratoga Group, Ponte Vedra Beach, pp 177–208
Hider RC, Zhou T (2005) Ann N Y Acad Sci 1054:141–154
O’Sullivan B, Xu J, Raymond KN (2000) In: Badman DG, Bergeron RJ, Brittenham GM (eds) Iron chelators: new development strategies. The Saratoga Group, Ponte Vedra Beach, pp 177–208
Cappellini MD, Cohen A, Piga A, Bejaoui M, Perrotta S, Agaoglu L, Aydinok Y, Kattamis A, Kilinc Y, Porter J, Capra M, Galanello R, Fattoum S, Drelichman G, Magnano C, Verissimo M, Athanassiou-Metaxa M, Giardina P, Kourakli-Symeonidis A, Janka-Schaub G, Coates T, Vermylen C, Olivieri N, Thuret I, Opitz H, Ressayre-Djaffer C, Marks P, Alberti D (2006) Blood 107:3455–3462
Motekaitis RJ, Martell AE (1991) Inorg Chim Acta 183:71–80
Pippard MJ (1994) In: Bergeron RJ , Brittenham GM (eds) The development of iron chelators for clinical use. CRC, Boca Raton, pp 57–74
Abergel RJ, Raymond KN (2006) Inorg Chem 45:3622–3631
Jurchen KMC, Raymond KN (2005) J Coord Chem 58:55–80
Jurchen KMC, Raymond KN (2006) Inorg Chem 45:1078–1090
Yokel RA, Fredenburg AM, Durbin PW, Xu J, Rayens MK, Raymond KN (2000) J Pharm Sci 89:545–555
Carrano CJ, Raymond KN (1979) J Am Chem Soc 101:5401–5404
Kretchmar S, Raymond KN (1986) J Am Chem Soc 108:6212–6218
Turcot I, Stintzi A, Xu JD, Raymond KN (2000) J Biol Inorg Chem 5:634–641
Kontoghiorghes GJ, Evans RW (1985) FEBS Lett 189:141–144
Harris WR (1984) J Inorg Biochem 21:263–276
Konopka K, Bindereif A, Neilands JB (1982) Biochemistry 21:6503–6508
Harris WR, Rezvani AB, Bali PK (1987) Inorg Chem 26:2711–2716
Aisen P, Leibman A (1972) Biochim Biophys Acta 257:311–323
Stintzi A, Raymond KN (2000) J Biol Inorg Chem 5:57–66
Ford S, Cooper RA, Evans RW, Hider RC, Williams PH (1988) Eur J Biochem 178:477–481
Hissen AHT, Moore MM (2005) J Biol Inorg Chem 10:211–220
Baldwin D (1980) Biochim Biophys Acta 623:183–198
Hamilton DH, Turcot I, Stintzi A, Raymond KN (2004) J Biol Inorg Chem 9:936–944
Thompson CP, Grady JK, Chasteen ND (1986) J Biol Chem 261:13128–13134
Raymond KN, Telford JR (1995) In: Kessissoglou DP (ed) Bioinorganic chemistry an inorganic perspective of life. Kluwer, Dordrecht, pp 25–37
Raymond KN, Bryan BL (1995) In: Kessissoglou DP (ed) Bioinorganic chemistry an inorganic perspective of life. Kluwer, Dordrecht, pp 13–24
Hershko C, Link G, Konijn AM (2002) In: Templeton DM (ed) Molecular and cellular iron transport. Dekker, New York, pp 775–816
Turcot I, Stintzi A, Xu J, Raymond KN (2000) J Biol Inorg Chem 5:634–641
Bali PK, Harris WR, Nesset-Tollefson D (1991) Inorg Chem 30:502–508
Bali PK, Harris WR (1989) J Am Chem Soc 111:4457–4461
Leibman A, Aisen P (1979) Blood 53:1058–1065
Baker EN, Baker HM, Kidd RD (2002) Biochem Cell Biol 80:27–34
He QY, Mason AB (2002) In: Templeton DM (ed) Molecular and cellular iron transport. Dekker, New York, pp 95–124
He QY, Mason AB, Tam BM, MacGillivray RTA, Woodworth RC (1999) Biochemistry 38:9704–9711
Zak O, Tam BM, MacGillivray RTA, Aisen P (1997) Biochemistry 36:11036–11043
Bali PK, Aisen P (1991) Biochemistry 30:9947–9952
Xu JD, Raymond KN (1994) In: Bergeron RJ, Brittenham JM (eds) The development of iron chelators for clinical use. CRC, Boca Raton, pp 307–327
Schwarzenbach G, Schwarzenbach K (1963) Helv Chim Acta 46:1390–1400
Motekaitis RJ, Martell AE (1991) Inorg Chim Acta 183:71–80
Harris WR, Carrano CJ, Cooper SR, Sofen SR, Avdeef A, McArdle JV, Raymond KN (1979) J Am Chem Soc 101:6097–6104
Rodgers SJ, Lee C, Ng CY, Raymond KN (1987) Inorg Chem 26:1622–1625
Acknowledgement
This research was supported by the National Institutes of Health (grant DK057814).
Author information
Authors and Affiliations
Corresponding author
Additional information
Paper number 43 in the series “Ferric ion sequestering agents.” For the previous paper, see [24].
Rights and permissions
About this article
Cite this article
Abergel, R.J., Raymond, K.N. Terephthalamide-containing ligands: fast removal of iron from transferrin. J Biol Inorg Chem 13, 229–240 (2008). https://doi.org/10.1007/s00775-007-0314-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-007-0314-y