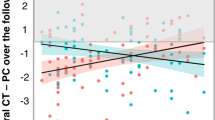
Abstract
Progressive loss of cortical gray matter (GM) and increase of cerebrospinal fluid (CSF) have been reported in early-onset psychosis (EOP). EOP typically begins during adolescence, a time when developmental brain trajectories differ by gender. This study aimed to determine gender differences in progression of brain changes in this population. A sample of 61 (21 females) adolescents with a first psychotic episode and a matched sample of 70 (23 females) controls underwent both baseline and 2-year follow-up anatomical brain imaging assessments. Regional GM and CSF volumes were obtained using automated methods based on the Talairach’s proportional grid system. At baseline, only male patients showed a clear pattern of alterations in the frontal lobe relative to controls (smaller GM and larger CSF volumes). However, parallel longitudinal changes for male and female patients relative to controls were observed, resulting in a common pattern of brain changes across both genders: rate of left frontal lobe GM volume loss was larger in male (−3.8 %) and female patients (−4.2 %) than in controls (−0.7 % males; −0.4 % females). The reverse was found for the CSF volume in the left frontal lobe. While the GM and CSF volumes of females with EOP appear to be within the normal range at initial illness onset, our results point to a similar trajectory of increased/accelerated brain changes in both male and female patients with EOP. The pattern of progression of brain changes in psychosis appears to be independent of gender or structural alterations on appearance of psychotic symptoms.


Similar content being viewed by others
References
Aleman A, Kahn RS, Selten JP (2003) Sex differences in the risk of schizophrenia: evidence from meta-analysis. Arch Gen Psychiatry 60(6):565–571
Hafner H (2003) Gender differences in schizophrenia. Psychoneuroendocrinology 28(Suppl 2):17–54
McGrath JJ (2007) The surprisingly rich contours of schizophrenia epidemiology. Arch Gen Psychiatry 64(1):14–16
Keshavan MS et al (2008) Schizophrenia, “just the facts”: what we know in 2008. Part 3: neurobiology. Schizophr Res 106(2–3):89–107
Leung A, Chue P (2000) Sex differences in schizophrenia, a review of the literature. Acta Psychiatr Scand Suppl 401:3–38
Huber TJ et al (2005) Sex hormones in psychotic men. Psychoneuroendocrinology 30(1):111–114
Gogos A, Kwek P, van den Buuse M (2012) The role of estrogen and testosterone in female rats in behavioral models of relevance to schizophrenia. Psychopharmacology 219(1):213–224
Bora E et al (2011) The effects of gender on grey matter abnormalities in major psychoses: a comparative voxelwise meta-analysis of schizophrenia and bipolar disorder. Psychol Med 42(2):295–307
Nopoulos P et al (2000) Sexual dimorphism in the human brain: evaluation of tissue volume, tissue composition and surface anatomy using magnetic resonance imaging. Psychiatry Res-Neuroimaging 98(1):1–13
Lenroot RK et al (2007) Sexual dimorphism of brain developmental trajectories during childhood and adolescence. Neuroimage 36(4):1065–1073
Blakemore SJ (2012) Imaging brain development: the adolescent brain. Neuroimage 61(2):397–406
Raznahan A et al (2011) How does your cortex grow? J Neurosci 31(19):7174–7177
Paus T, Keshavan M, Giedd JN (2008) Why do many psychiatric disorders emerge during adolescence? Nat Rev Neurosci 9(12):947–957
Goldstein JM et al (2002) Impact of normal sexual dimorphisms on sex differences in structural brain abnormalities in schizophrenia assessed by magnetic resonance imaging. Arch Gen Psychiatry 59(2):154–164
Thompson PM et al (2001) Mapping adolescent brain change reveals dynamic wave of accelerated gray matter loss in very early-onset schizophrenia. Proc Natl Acad Sci USA 98(20):11650–11655
Rapoport JL et al (1999) Progressive cortical change during adolescence in childhood-onset schizophrenia—a longitudinal magnetic resonance imaging study. Arch Gen Psychiatry 56(7):649–654
Sporn AL et al (2003) Progressive brain volume loss during adolescence in childhood-onset schizophrenia. Am J Psychiatry 160(12):2181–2189
Vidal CN et al (2006) Dynamically spreading frontal and cingulate deficits mapped in adolescents with schizophrenia. Arch Gen Psychiatry 63(1):25–34
Keller A et al (2003) Progressive loss of cerebellar volume in childhood-onset schizophrenia. Am J Psychiatry 160(1):128–133
Frazier JA et al (2008) Diagnostic and sex effects on limbic volumes in early-onset bipolar disorder and schizophrenia. Schizophr Bull 34(1):37–46
James ACD et al (2002) Evidence for non-progressive changes in adolescent-onset schizophrenia—follow-up magnetic resonance imaging study. Br J Psychiatry 180:339–344
James AC et al (2004) Cerebellar, prefrontal cortex, and thalamic volumes over two time points in adolescent-onset schizophrenia. Am J Psychiatry 161(6):1023–1029
Weisinger B et al (2013) Lack of gender influence on cortical and subcortical gray matter development in childhood-onset schizophrenia. Schizophr Bull 39(1):52–58
Hafner H et al (1995) When and how does schizophrenia produce social deficits. Eur Arch Psychiatry Clin Neurosci 246(1):17–28
Huber G (1997) The heterogeneous course of schizophrenia. Schizophr Res 28:177–185
Gogtay N et al (2011) Age of onset of schizophrenia: perspectives from structural neuroimaging studies. Schizophr Bull 37(3):504–513
Reig S et al (2009) Progression of brain volume changes in adolescent-onset psychosis. Schizophr Bull 35(1):233–243
Reig S et al (2010) Multicenter study of brain volume abnormalities in children and adolescent-onset psychosis. Schizophr Bull 37(6):1270–1280
Arango C et al (2012) Progressive brain changes in children and adolescents with first-episode psychosis. Arch Gen Psychiatry 69(1):16–26
Lenroot RK, Giedd JN (2010) Sex differences in the adolescent brain. Brain Cogn 72(1):46–55
Castro-Fornieles J et al (2007) The child and adolescent first-episode psychosis study (CAFEPS): design and baseline results. Schizophr Res 91(1–3):226–237
Soutullo C (2007) Traducción al Español de la Entrevista Diagnóstica: Kiddie-Schedule for affective disorders and schizophrenia, present and lifetime version (K-SADS-PL, 1996). http://www.cun.es/la-clinica/departamentos-yservicios-medicos/psiquiatria-y-psicologia-medica/mas-sobreel-departamento/unidades/psiquiatria-infantil-y-adolescente, 1999
Geller B et al (2001) Reliability of the Washington University in St. Louis Kiddie schedule for affective disorders and schizophrenia (WASH-U-KSADS) mania and rapid cycling sections. J Am Acad Child Adolesc Psychiatry 40(4):450–455
Kay SR, Fiszbein A, Opler LA (1987) The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull 13(2):261–276
Peralta V, Cuesta MJ (1994) Validation of positive and negative symptom scale (PANSS) in a sample of Spanish schizophrenia patients. Actas Luso Españolas de Neurología Psiquiatría y Ciencias Afines 22:171–177
Shaffer D et al (1983) A children’s global assessment scale (CGAS). Arch Gen Psychiatry 40:1228–1231
Fraguas D et al (2014) Duration of untreated psychosis predicts functional and clinical outcome in children and adolescents with first episode psychosis: a 2-year longitudinal study. Schizophr Res 152(1):130–138
Rey MJ et al (1989) Guidelines for the dosage of neuroleptics. I: chlorpromazine equivalents of orally-administered neuroleptics. Int Clin Psychopharmacol 4(2):95–104
Woods SW (2003) Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry 64(6):663–667
Reig S et al (2009) Assessment of the increase in variability when combining volumetric data from different scanners. Hum Brain Mapp 30(2):355–368
Andreasen NC et al (1996) Automatic atlas-based volume estimation of human brain regions from MR images. J Comput Assist Tomogr 20(1):98–106
Kates WR et al (1999) Automated Talairach atlas-based parcellation and measurement of cerebral lobes in children. Psychiatry Res-Neuroimaging 91(1):11–30
Desco M et al (2001) Multimodality image quantification using Talairach grid. In: Sonka M, Hason KM (eds) In: Proceedings from the international society for optical engineering, 18–22 February 2001, San Diego CA
Ashburner J, Friston KJ (1997) Multimodal image coregistration and partitioning—a unified framework. Neuroimage 6:209–217
Talairach J, Tournoux P (1988) Co-planar stereotaxic atlas of the human brain. Thieme Medical, New York
Huber TJ et al (2001) Estradiol levels in psychotic disorders. Psychoneuroendocrinology 26(1):27–35
Kulkarni J et al (2008) Estrogen in severe mental illness: a potential new treatment approach. Arch Gen Psychiatry 65(8):955–960
Viveros MP et al (2012) A comparative, developmental, and clinical perspective of neurobehavioral sexual dimorphisms. Front Neurosci 6:84
Nopoulos P, Flaum M, Andreasen N (1997) Sex differences in brain morphology in schizophrenia. Am J Psychiatry 154:1648–1654
Molina V et al (2005) Association between excessive frontal cerebrospinal fluid and illness duration in males but not in females with schizophrenia. Eur Psychiatry 20:332–338
Beltz AM, Berenbaum SA (2013) Cognitive effects of variations in pubertal timing: is puberty a period of brain organization for human sex-typed cognition? Horm Behav 63(5):823–828
Field EF et al (2004) Neonatal and pubertal, but not adult, ovarian steroids are necessary for the development of female-typical patterns of dodging to protect a food item. Behav Neurosci 118(6):1293–1304
Schulz KM, Molenda-Figueira HA, Sisk CL (2009) Back to the future: the organizational-activational hypothesis adapted to puberty and adolescence. Horm Behav 55(5):597–604
Ismail N, Garas P, Blaustein JD (2011) Long-term effects of pubertal stressors on female sexual receptivity and estrogen receptor-alpha expression in CD-1 female mice. Horm Behav 59(4):565–571
Cropley V, Wood SJ, Pantelis C (2013) Brain structural, neurochemical and neuroinflammatory markers of psychosis onset and relapse: is there evidence for a psychosis relapse signature? Int Clin Psychopharmacol [Epub ahead of print]
Fraguas D et al (2012) Decreased glutathione levels predict loss of brain volume in children and adolescents with first-episode psychosis in a two-year longitudinal study. Schizophr Res 137(1–3):58–65
Cahn W et al (2009) Psychosis and brain volume changes during the first five years of schizophrenia. Eur Neuropsychopharmacol 19(2):147–151
Sun D et al (2009) Brain surface contraction mapped in first-episode schizophrenia: a longitudinal magnetic resonance imaging study. Mol Psychiatry 14(10):976–986
Pantelis C et al (2005) Structural brain imaging evidence for multiple pathological processes at different stages of brain development in schizophrenia. Schizophr Bull 31(3):672–696
McGlashan T (2006) Is active psychosis neurotoxic? Schizophr Bull 32:609–613
Weinberger DR (2002) Biological phenotypes and genetic research on schizophrenia. World Psychiatry 1(1):2–6
Andreasen NC et al (2013) Relapse duration, treatment intensity, and brain tissue loss in schizophrenia: a prospective longitudinal MRI study. Am J Psychiatry 170(6):609–615
Sowell ER et al (2003) Mapping cortical change across the human life span. Nat Neurosci 6:309–315
Jernigan TL et al (1991) Maturation of human cerebrum observed in vivo during adolescence. Brain 114(5):2037–2049
Zipursky RB, Reilly TJ, Murray RM (2012) The myth of schizophrenia as a progressive brain disease. Schizophr Bull 39(6):1363–1372
Cannon TD, Mednick SA, Parnas J (1989) Genetic and perinatal determinants of structural brain deficits in schizophrenia. Arch Gen Psychiatry 46:883–889
Keshavan MS, Anderson S, Pettegrew JW (1994) Is schizophrenia due to excessive synaptic pruning in the prefrontal cortex? The Feinberg hypothesis revisited. J Psychiatr Res 28:239–265
Huttenlocher PR, Dabholkar AS (1997) Regional differences in synaptogenesis in human cerebral cortex. J Comp Neurol 387:167–178
Lenroot RK et al (2009) Differences in genetic and environmental influences on the human cerebral cortex associated with development during childhood and adolescence. Hum Brain Mapp 30(1):163–174
Kulkarni J, Hayes E, Gavrilidis E (2012) Hormones and schizophrenia. Curr Opin Psychiatry 25(2):89–95
Gur RE et al (2000) Reduced dorsal and orbital prefrontal gray matter volumes in schizophrenia. Arch Gen Psychiatry 57(8):761–768
Velakoulis D et al (2006) Hippocampal and amygdala volumes according to psychosis stage and diagnosis: a magnetic resonance imaging study of chronic schizophrenia, first-episode psychosis, and ultra-high-risk individuals. Arch Gen Psychiatry 63(2):139–149
Pantelis C, Velakoulis D, McGorry PD et al (2003) Neuroanatomical abnormalities before and after onset of psychosis: a cross-sectional and longitudinal MRI comparison. Lancet 361:281–288
Cropley VL, Pantelis C (2014) Using longitudinal imaging to map the ‘relapse signature’ of schizophrenia and other psychoses. Epidemiol Psychiatr Sci 23(3):219–225
Giedd JN, Rapoport JL (2010) Structural MRI of pediatric brain development: what have we learned and where are we going? Neuron 67(5):728–734
Hollingshead AB, Redlich FC (1954) Schizophrenia and social structure. Am J Psychiatry 110(9):695–701
Acknowledgments
We thank Jose de Arriba of the Child and Adolescent Psychiatry Department, Hospital General Universitario Gregorio Marañón, CIBERSAM, Madrid, Spain for data management. In memoriam: Santiago Reig, Ph.D. (1958–2011). We thank Dr. Reig for his statistical guidance, mentorship and outstanding contributions, not only to the development of this study but also to the field of neuroimaging studies in early-onset psychosis.
Conflict of interest
This work was supported by the Spanish Ministry of Economy and Competitiveness. Instituto de Salud Carlos III, CIBERSAM, RETICS RD06/0011 (REM-TAP Network), Fundación Alicia Koplowitz, Fundación Mutua Madrileña, by Grants PI02/1248, PI05/0678, PI09/01442, PI12/1303, G03/032, Regional Government (S2010/BMD-2422 AGES) and by the European Union Structural Funds (Madrid, Spain). This work was partially supported by Spain’s Ministry of Science and Innovation through CDTI’s CENIT Program (AMIT Project). Dr. Rapado-Castro has received a Health Research Sara Borrell Fellowship from the Spanish Ministry of Economy and Competitiveness, an Alicia Koplowitz Grant for Short-Term Placements from the Alicia Koplowitz Foundation and an IiSGM Fellowship Award for Short-term Placements from the Health Research Institute from the Hospital Gregorio Marañón (IiSGM) (Madrid, Spain). Dr. Rapado-Castro has also received grant support from the Fundación Alicia Koplowitz. Dr. Bartholomeusz has received a National Health and Medical Research Council (NHMRC) Australian-based Clinical Research Fellowship and a John and Betty Lynch Fellowship from the Department of Psychiatry, Faculty of Medicine, Dentistry and Health Sciences, The University of Melbourne. Dr. González-Pinto is employed by the University of the Basque Country and has been a consultant for the Ministry of Science of Spain and the Basque Government. Dr. González-Pinto also gives conferences, acts as consultant to or receives grants from Lilly, Janssen, MSD, Lundbek, AstraZeneca, Almirall, Sanofi Aventis, BMS, Novartis and Pfizer. Dr. Baeza has received support from Otsuka for attending a conference. Dr. Moreno has served as a consultant to AstraZeneca, Otsuka, Bristol-Myers Squibb and Janssen. Dr. Moreno has also received grant support from the Fundación Alicia Koplowitz, Instituto de Salud Carlos III and European Union Structural Funds. Professor Pantelis has received a NHMRC Australian-based Senior Principal Research Fellowship (ID: 628386), NARSAD Distinguished Investigator Award (US; Grant ID: 18722), and NHMRC Program Grant (ID: 566529); he has received grant support from Janssen-Cilag, Eli Lilly, Hospira (Mayne), Astra Zeneca and has provided consultancy to Janssen-Cilag, Eli Lilly, Hospira (Mayne), Astra Zeneca, Pfizer, Schering Plough, Lundbeck. He has undertaken investigator initiated studies supported by Eli Lilly, Hospira, Janssen Cilag and Astra Zeneca. Dr. Arango. has been a consultant to or has received honoraria or grants from Abbot, AMGEN, AstraZeneca, Bristol-Myers Squibb, Caja Navarra, CIBERSAM, Fundación Alicia Koplowitz, Instituto de Salud Carlos III, Janssen Cilag, Lundbeck, Merck, Ministerio de Ciencia e Innovación, Ministerio de Sanidad, Ministerio de Economía y Competitividad, Mutua Madrileña, Otsuka, Pfizer, Roche, Servier, Shire, Takeda and Schering Plough. The rest of the authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Partial results of this work were presented as a poster at the 2nd Biennial Schizophrenia International Research Conference, April 12, 2010 in Florence, Italy. The abstract was published in Schizophrenia Research, doi:10.1016/j.schres.2010.02.605. This work was presented as poster at the 3rd Schizophrenia International Research Society Conference “Schizophrenia: The Globalization of research”. Florence, Italy. The abstract was published in Schizophrenia Research, 136 (Supplement 1) pp. 372–373. ISSN 0920-9964.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rapado-Castro, M., Bartholomeusz, C.F., Castro-Fornieles, J. et al. Gender effects on brain changes in early-onset psychosis. Eur Child Adolesc Psychiatry 24, 1193–1205 (2015). https://doi.org/10.1007/s00787-014-0669-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-014-0669-x