Abstract
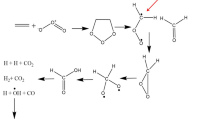
A detailed doublet potential energy surface for the reaction of CH with CH3CCH is investigated at the B3LYP/6-311G(d,p) and G3B3 (single-point) levels. Various possible reaction pathways are probed. It is shown that the reaction is initiated by the addition of CH to the terminal C atom of CH3CCH, forming CH3CCHCH 1 (1a,1b). Starting from 1 (1a,1b), the most feasible pathway is the ring closure of 1a to CH3–cCCHCH 2 followed by dissociation to P 3 (CH3–cCCCH+H), or a 2,3 H shift in 1a to form CH3CHCCH 3 followed by C–H bond cleavage to form P 5 (CH2CHCCH+H), or a 1,2 H-shift in 1 (1a, 1b) to form CH3CCCH2 4 followed by C–H bond fission to form P 6 (CH2CCCH2+H). Much less competitively, 1 (1a,1b) can undergo 3,4 H shift to form CH2CHCHCH 5. Subsequently, 5 can undergo either C–H bond cleavage to form P 5 (CH2CHCCH+H) or C–C bond cleavage to generate P 7 (C2H2+C2H3). Our calculated results may represent the first mechanistic study of the CH + CH3CCH reaction, and may thus lead to a deeper understanding of the title reaction.







Similar content being viewed by others
References
Sanders WA, Lin MC (1986) In chemical kinetic of small organic radical, vol III. CRC, Boca Raton
Miller JA, Kee RJ, Westbrook CK (1990) Annu Rev Phys Chem 41:345–387
Lindqvist M, Sandqvist A, Winnberg A, Johansson L, Nyman LA (1995) Astron Astrophys Suppl Ser 113:257–263
Canosa A, Sims IR, Travers D, Smith IWM, Rowe BR (1997) Astron Astrophys 323:644–651
Amin MY, El-Nawawy MS (1996) Earth Moon Planet 75:25–39
Brownsword RA, Sims IR, Smith IWM (1997) Astrophys J 485:195–201
Herzberg G, Johns JWC (1969) Astrophys J 58:399–406
Bernath PF (1987) J Chem Phys 86:4838–4842
Zachwieja M (1995) J Mol Spectrosc 170:285–289
Kalemos A, Mavridis A, Metropoulos A (1999) J Chem Phys 111:9536–9548
Hirata S, Yanai T, Jong WA, Nakajima T, Hirao K (2004) J Chem Phys 120:3297–3310
Vázquez GJ, Amero JM, Liebermann HP, Buenker RJ, Lefebvre-Brion H (2007) J Chem Phys 126:164302–164315
Ikejiri K, Ohoyama H, Nagamachi Y, Kasai T (2005) Chem Phys Lett 401:465–469
Manaa MR, Yarkony DR (1991) J Chem Phys 95:1808–1817
Seideman T, Walch SP (1994) J Chem Phys 101:3656–3662
Seideman T (1994) J Chem Phys 101:3662–3671
Berman MR, Tsuchiya T, Gregušová A, Perera SA, Bartlett RJ (2007) J Phys Chem A 111:6894–6899
Bergeat A, Calvo T, Dorthe G, Loison JC (1999) J Phys Chem A 103:6360–6365
Jursic BS (1998) J Phys Chem A 102:9255–9260
Fleurat-Lessard P, Rayez JC, Bergeat A, Loison AC (2002) Chem Phys 279:87–99
Daugey N, Caubet P, Retail B, Costes M, Bergeat A, Dorthe G (2005) Phys Chem Chem Phys 7:2921–2927
Loison JC, Bergeat A, Caralp F, Hannachi Y (2006) J Phys Chem A 110:13500–13506
Mckee K, Blitz MA, Hughes KJ, Pilliing MJ, Qian HB, Taylor A, Seakins PW (2003) J Phys Chem A 107:5710–5716
Galland N, Caralp F, Hannachi Y, Bergeat A, Loison JC (2003) J Phys Chem A 107:5419–5426
Loison JC, Bergeat A (2009) Phys Chem Chem Phys 11:655–664
Goulay F, Trevitt AJ, Meloni G, Selby TM, Osborn DL, Taatjes CA, Vereecken L, Leone SR (2009) J Am Chem Soc 131:993–005
Frisch MJ, Trucks GW, Schlegel HB et al (2004) Gaussian 03, revision D.02. Gaussian Inc., Wallingford
Acknowledgments
This work is supported by the National Natural Science Foundation of China (nos. 20773048, 21073075)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, L., Liu, Hl., Yang, GH. et al. Theoretical mechanistic study of the reaction of the methylidyne radical with methylacetylene. J Mol Model 17, 3173–3181 (2011). https://doi.org/10.1007/s00894-011-0979-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-011-0979-6