Abstract
Stable crystalline phosphorus ylides were obtained in excellent yields from the 1:1:1 addition reaction between triphenylphosphine (TPP) and dialkyl acetylenedicarboxylates, in the presence of NH-acids, such as benzhydrazide. To determine the kinetic parameters of the reactions, they were monitored by UV spectrophotometery. The second order fits were automatically drawn and the values of the second order rate constant (k2) were calculated using standard equations within the program. At the temperature range studied the dependence of the second order rate constant (Ln k2) on reciprocal temperature was compatible with Arrhenius equation. This provided the relevant plots to calculate the activation energy of all reactions. Furthermore, useful information were obtained from studies of the effect of solvent, structure of reactants (different alkyl groups within the dialkyl acetylenedicarboxylates) and also concentration of reactants on the rate of reactions. On the basis of experimental data the proposed mechanism was confirmed according to the obtained results and a steady state approximation and the first step (k2) and third (k3) steps of the reactions were recognized as the rate determining steps, respectively. In addition, three speculative proposed mechanisms were theoretically investigated using quantum mechanical calculation. The results, arising from the second and third speculative mechanisms, were far from the experimental data. Nevertheless, there was a good agreement between the theoretical kinetic data, emerge from the first speculative mechanism, and experimental kinetic data of proposed mechanism.

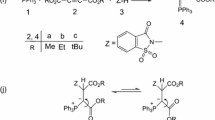
Kinetics and mechanistic study in a synthesis reaction between triphenylphosphine (1) and dialkyl acetylenedicarboxylates (2) in the presence of benzhydrazide (3) Theoretical and Experimental investigation



















Similar content being viewed by others
References
Laszo P (1995) Organic reaction, simplicity and logic. Wiley, New York
Johnson AW (1966) Ylied chemistry. Academic, London
Cadogan JIG (1979) Organophosphorus reagents in organic synthesis. Academic, New York
Engel R (1988) Synthesis of carbon-phosphors bonds. CRC, Boca Raton
Hudson HR (1990) In: Hartley FR (ed) The chemistry of organophosphorus compounds; primary, secondary, and tertiary phosphates and heterocyclic organophosphorus (3) compounds. Wiley, New York, pp 382–472
Corbridge DEC (1995) Phosphorus: an outline of chemistry, biochemistry and uses. Elsevier, Amsterdam
Pietrusiewiz KM, Zablocka M (1994) Chem Rev 94:1375–1411
Shen Y (1998) Acc Chem Res 31:584–592
Gilchrist TL (1985) Heterocyclic Chemistry. Wiley, NewYork
Yavari I, Alizadeh A, Anary-Abbasinejad M (2002) Tetrahedron Lett 43:4503–4505
Yavari I, Adib M, Hojabri L (2002) Tetrahedron 58:7213–7219
Yavari I, Alizadeh A, Anary-Abbasinejad M (2002) Tetrahedron Lett 43:9449–9452
Yavari I, Adib M, Jahani-Moghaddam F, Sayahi MH (2002) Phosphorus Sulfur Silicon Relat Elem 177:545–553
Ramazani A, Shajari N, Gouranlou F (2001) Phosphorus Sulfur Silicon Relat Elem 174:223–227
Maghsoodlou MT, Habibi-Khorassani SM, Heydari R, Rostami-Charati F (2006) J Chem Res 364-365
Maghsoodlou MT, Rostami-Charati F, Habibi-Khorassani SM, Khosroshahrodi M, Makha M (2008) Iran J Chem Chem Eng 27:105–113
Maghsoodlou MT, Heydari R, Hazeri N, Habibi-Khorassani SM, Nassiri M, Ghasemzadeh M, Salehzadeh J, Gharechaei Z (2009) Heteroatom Chem 20:240–245
Maghsoodlou MT, Hazeri N, Habibi-Khorassani SM, Heydari R, Marandi G, Lashkari M, Bagherpour K, Gharechaei Z (2010) Monatsh Chem 141:351–356
Aminkhani A, Kabiri R, Habibi-Khorassani SM, Heydari R, Maghsoodlou MT, Marandi G, Lashkari M, Rostamizadeh M (2009) J Sulfur Chem 30:500–506
Habibi-Khorassani SM, Maghsoodlou MT, Zakarianejad M, Nassiri M, Kazemian MA, Karimi P (2008) Heteroatom Chem 19:723–732
Ortiz de Montellano PR (1995) Biochimie 77:581–593
DePillis GD, Wariishi H, Gold MH, Ortiz de Montellano PR (1990) Arch Biochem Biophys 280:217–223
Harris RZ, Wariishi H, Gold MH, Ortiz de Montellano PR (1991) J Biol Chem 266:8751–8758
Samokyszyn VM, Ortiz de Montellano PR (1991) Biochemistry 30:11646–11653
Burner U, Obinger C, Paumann M, Furtmuller PG, Kettle AJ (1999) J Biol Chem 274:9494–9502
Wengenack NL, Rusnak F (2001) Biochemistry 40:8990–8996
Torffvit O, Thysell H, Nassberger L (1994) Hum Exp Toxicol 13:563–567
Reilly CA, Aust SD (1997) Chem Res Toxicol 10:328–334
Silverman RB (1988) Mechanism-based enzyme inactivation: Chemistry and enzymology, vol. 1. CRC, Boca Raton
Furtmuller PG, Burner U, Regelsberger G (2000) Biochemistry 39:15578–15584
Nielsen OJ, Sehsted J, Langer S, Ljungström E, Wängberg I (1995) Chem Phys Lett 238:359–364
Langer S, Ljungström E, Ellemann T, Sehsted J, Nielsen OJ (1995) Chem Phys Lett 53:240–255
Petersson GA, Al-Laham MA (1991) J Chem Phys 94:6081–6090
Petersson GA, Bennett A, Tensfeldt TG, Al-Laham MA, Shirley WA, Mantzaris J (1988) J Chem Phys 89:2193–2218
Becke AD (1988) Phys Rev A 38:3098–3100
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Schmidt MW, Baldridge KK, Boatz JA, Elbert ST, Gordon MS, Jensen JH, Koseki S, Matsunaga N, Nguyen KA, Su SJ, Windus TL, Dupuis M, Montgomery JA (1993) General atomic and molecular electronic structure system. J Comput Chem 14:1347–1363
Acknowledgments
Authors sincerely thank the University of Sistan & Baluchestan for providing financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kazemian, M.A., Habibi-Khorassani, S.M., Ebrahimi, A. et al. A joint experimental and theoretical investigation of kinetics and mechanistic study in a synthesis reaction between triphenylphosphine and dialkyl acetylenedicarboxylates in the presence of benzhydrazide. J Mol Model 18, 5075–5088 (2012). https://doi.org/10.1007/s00894-012-1503-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-012-1503-3