Abstract
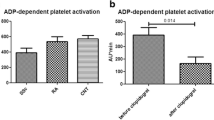
The purpose of this study is to assess the effect of sulfasalazine and its metabolites on platelet function in patients with inflammatory arthritis (IA). One hundred thirty-five consecutive patients with an established diagnosis of IA were screened. Those with a history of cardiovascular disease (CVD), taking anti-platelet agents or non-steroidal anti-inflammatory drugs (NSAIDs) were excluded. A total of 32 patients were investigated, 15 taking sulfasalazine and 17 taking other disease-modifying anti-rheumatic drugs (DMARDs) and no sulfasalazine. These two cohorts were compared to 15 patients with stable CVD on long-term aspirin. The effect of sulfasalazine and its metabolites on arachidonic acid (AA)-induced platelet aggregation was also tested in vitro in samples from healthy donors (n = 18). Demographics, CVD risk factors and disease activity indices were similar in the sulfasalazine and other DMARD groups. AA-induced platelet aggregation was significantly inhibited in the sulfasalazine group (9 ± 7 %) and comparable to that in the aspirin group (10 ± 6 %). In contrast, there was no effect on AA-induced platelet aggregation in the other DMARDs group (77 ± 12 %) (p < 0.001). Furthermore, sulfasalazine therapy had no effect on platelet aggregation in response to multiple other agonists. Sulfasalazine and its metabolites (5-aminosalicylic acid and sulfapyridine) exerted an additive and dose-dependent inhibitory effect on AA-induced platelet aggregation in vitro (p < 0.001). The inhibition of AA-induced platelet aggregation by sulfasalazine is comparable to that achieved by aspirin and is dependent on both sulfasalazine and its metabolites. This represents a potential mechanism that may contribute to the known cardioprotective effect of sulfasalazine in patients with IA.



Similar content being viewed by others
References
Wahl C, Liptay S, Adler G, Schmid RM (1998) Sulfasalazine: a potent and specific inhibitor of nuclear factor kappa B. J Clin Invest 101(5):1163–1174
Naranjo A, Sokka T, Descalzo MA, Calvo-Alen J, Horslev-Petersen K, Luukkainen RK et al (2008) Cardiovascular disease in patients with rheumatoid arthritis: results from the QUEST-RA study. Arthritis Res Ther 10(2):R30
Lee HJ, Waller RD, Stebbings S, Highton J, Orlovich DA, Schmierer D et al (2010) The effects of an orally administered probiotic on sulfasalazine metabolism in individuals with rheumatoid arthritis: a preliminary study. Int J Rheum Dis 13(1):48–54
Stenson WF, Lobos E (1983) Inhibition of platelet thromboxane synthetase by sulfasalazine. Biochem Pharmacol 32(14):2205–2209
Gurbel PA, Becker RC, Mann KG, Steinhubl SR, Michelson AD (2007) Platelet function monitoring in patients with coronary artery disease. J Am Coll Cardiol 50(19):1822–1834
Davi G, Patrono C (2007) Platelet activation and atherothrombosis. N Engl J Med 357(24):2482–2494
Peace AJ, Tedesco AF, Foley DP, Dicker P, Berndt MC, Kenny D (2008) Dual antiplatelet therapy unmasks distinct platelet reactivity in patients with coronary artery disease. J Thromb Haemost 6:2027–2034
Catella-Lawson F, Reilly MP, Kapoor SC, Cucchiara AJ, DeMarco S, Tournier B et al (2001) Cyclooxygenase inhibitors and the antiplatelet effects of aspirin. N Engl J Med 345(25):1809–1817
Peace AJ, Egan K, Kavanagh GF, Tedesco AF, Foley DP, Dicker P et al (2009) Reducing intra-individual variation in platelet aggregation: implications for platelet function testing. J Thromb Haemost 7(11):1941–1943
Smolen JS, Breedveld FC, Eberl G, Jones I, Leeming M, Wylie GL et al (1995) Validity and reliability of the twenty-eight-joint count for the assessment of rheumatoid arthritis activity. Arthritis Rheum 38(1):38–43
Moran N, Kiernan A, Dunne E, Edwards RJ, Shields DC, Kenny D (2006) Monitoring modulators of platelet aggregation in a microtiter plate assay. Anal Biochem 357(1):77–84
Baber N (1994) International conference on harmonisation of technical requirements for registration of pharmaceuticals for human use (ICH). Br J Clin Pharmacol 37(5):401–404
Coppinger JA, O’Connor R, Wynne K, Flanagan M, Sullivan M, Maguire PB et al (2007) Moderation of the platelet released response by aspirin. Blood 109(11):4786–4792
Smock KJ, Rodgers GM (2010) Laboratory evaluation of aspirin responsiveness. Am J Hematol 85(5):358–360
Harrison P, Frelinger AL 3rd, Furman MI, Michelson AD (2007) Measuring antiplatelet drug effects in the laboratory. Thromb Res 120(3):323–336
Del Rincon I, O’Leary DH, Freeman GL, Escalante A (2007) Acceleration of atherosclerosis during the course of rheumatoid arthritis. Atherosclerosis 195(2):354–360
van Halm VP, Peters MJ, Voskuyl AE, Boers M, Lems WF, Visser M et al (2009) Rheumatoid arthritis versus diabetes as a risk factor for cardiovascular disease: a cross-sectional study, the CARRE Investigation. Ann Rheum Dis 68(9):1395–1400
Van Doornum S, Brand C, King B, Sundararajan V (2006) Increased case fatality rates following a first acute cardiovascular event in patients with rheumatoid arthritis. Arthritis Rheum 54(7):2061–2068
Peters MJ, Symmons DP, McCarey D, Dijkmans BA, Nicola P, Kvien TK et al (2010) EULAR evidence-based recommendations for cardiovascular risk management in patients with rheumatoid arthritis and other forms of inflammatory arthritis. Ann Rheum Dis 69(2):325–331
Barrett NE, Holbrook L, Jones S, Kaiser WJ, Moraes LA, Rana R et al (2008) Future innovations in anti-platelet therapies. Br J Pharmacol 154(5):918–939
Wang F, Wang NS, Yan CG, Li JH, Tang LQ (2007) The significance of platelet activation in rheumatoid arthritis. Clin Rheumatol 26(5):768–771
Ferguson JJ (2006) The role of oral antiplatelet agents in atherothrombotic disease. Am J Cardiovasc Drugs 6(3):149–157
Buse JB, Ginsberg HN, Bakris GL, Clark NG, Costa F, Eckel R et al (2007) Primary prevention of cardiovascular diseases in people with diabetes mellitus: a scientific statement from the American Heart Association and the American Diabetes Association. Circulation 115(1):114–126
Dixon WG, Watson KD, Lunt M, Hyrich KL, Silman AJ, Symmons DP (2007) Reduction in the incidence of myocardial infarction in patients with rheumatoid arthritis who respond to anti-tumor necrosis factor alpha therapy: results from the British Society for Rheumatology Biologics Register. Arthritis Rheum 56(9):2905–2912
Mani H, Luxembourg B, Klaffling C, Erbe M, Lindhoff-Last E (2005) Use of native or platelet count adjusted platelet rich plasma for platelet aggregation measurements. J Clin Pathol 58(7):747–750
Cattaneo M (2004) Aspirin and clopidogrel: efficacy, safety, and the issue of drug resistance. Arterioscler Thromb Vasc Biol 24(11):1980–1987
Gurbel PA, Bliden KP, Kreutz RP, Dichiara J, Antonino MJ, Tantry US (2009) The link between heightened thrombogenicity and inflammation: pre-procedure characterization of the patient at high risk for recurrent events after stenting. Platelets 20(2):97–104
Bernlochner I, Steinhubl S, Braun S, Morath T, Jaitner J, Stegherr J et al (2010) Association between inflammatory biomarkers and platelet aggregation in patients under chronic clopidogrel treatment. Thromb Haemost 13:104(6)
Mac Mullan PA, Peace AJ, Madigan AM, Tedesco AF, Kenny D, McCarthy GM (2010) Platelet hyper-reactivity in active inflammatory arthritis is unique to the adenosine diphosphate pathway: a novel finding and potential therapeutic target. Rheumatology (Oxford) 49(2):240–245
Harrison P, Keeling D (2006) Platelet hyperactivity and risk of recurrent thrombosis. J Thromb Haemost 4(12):2544–2546
Gurbel PA, Bliden KP, Guyer K, Cho PW, Zaman KA, Kreutz RP et al (2005) Platelet reactivity in patients and recurrent events post-stenting: results of the PREPARE POST-STENTING Study. J Am Coll Cardiol 46(10):1820–1826
Espinola RG, Pierangeli SS, Gharavi AE, Harris EN (2002) Hydroxychloroquine reverses platelet activation induced by human IgG antiphospholipid antibodies. Thromb Haemost 87(3):518–522
Acknowledgments
The authors would like to acknowledge Dr. Conor McCarthy and Dr. Suzanne Donnelly of the Mater Hospital’s Rheumatology Department for their assistance in recruiting participants for this study.
Authors’ contributions
Paul MacMullan was involved in all aspects of this research, including study design, data collection, analyses and manuscript preparation. Anne Madigan’s main contribution centred around patient recruitment, disease activity assessment, data collection and write-up. Nevin Paul performed the experiments for the in vitro work and subsequent data collection and analysis. Aaron Peace was involved in study design, adaptation of the appropriate platelet function testing procedures, data analysis and write-up. Ahmed Alagha helped to design the in vitro experiments, prepared the agents to be tested and collected data. Kevin Nolan oversaw the in vitro work and contributed to the write-up. As the senior authors, both Geraldine McCarthy and Dermot Kenny were involved in all aspects of this work, including study design, data analyses and preparation of final submission.
Authors’ information
The first author, Dr. Paul MacMullan, is a graduate of the Irish SpR Program in Rheumatology and has recently taken up an appointment as Clinical Assistant Professor at the University of Calgary in Alberta, Canada. His main areas of research interest include mechanisms of cardiovascular disease and platelet function in the inflammatory arthritis, crystal arthritis and the potential pathogenic role of basic calcium phosphate crystals in osteoarthritis. He has several publications in international peer-reviewed journals and is completing a higher degree in research.
As one of the two senior authors, Professor Geraldine McCarthy is a world-renowned rheumatologist with an international research reputation. She has published widely in many different areas and is a sought-after speaker with a particular expertise in gout.
Prof. Dermot Kenny is a cardiologist and clinician scientist based at the RCSI in Dublin. He is an international expert and authority on platelet function and is a member of the International Society for Thrombosis and Haemostasis (ISTH) committee for recommendations and guidelines surrounding platelet function testing.
Conflict of interest
All authors have completed the Unified Competing Interest form and declare no financial relationships with any organisations that might have an interest in the submitted work and no other relationships or activities that could appear to have influenced the submitted work.
Grant support
This work was not funded by any formal grant, but was partially supported by indirect funding from the Health Research Board (Ireland) and by unrestricted training allowances from Abbott Laboratories (Ireland) Ltd. and Pfizer (Ireland) Ltd.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
MacMullan, P.A., Madigan, A.M., Paul, N. et al. Sulfasalazine and its metabolites inhibit platelet function in patients with inflammatory arthritis. Clin Rheumatol 35, 447–455 (2016). https://doi.org/10.1007/s10067-014-2769-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-014-2769-x