Abstract
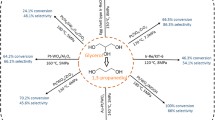
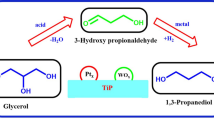
Biodiesel is one of the most common biofuels, and its production yields a large amount of glycerol as a by-product. It is necessary to develop new technologies for the use of this by-product, adding value to the biodiesel production chain. In this work we investigated glycerol oxidation under mild reaction conditions (air as oxidizing agent and base-free medium) promoted by suitable catalysts. We prepared mono- and bimetallic catalysts of platinum, copper and nickel in the form of nanoparticles by conventional heating and by an alternative method using microwave heating. The nanoparticles were dispersed in activated carbon and tested in glycerol oxidation aiming its valorization into molecules with high added value. Copper and nickel monometallic materials were not active in glycerol oxidation. Platinum monometallic and platinum–copper and platinum–nickel bimetallic materials showed catalytic activity, with platinum–nickel prepared by microwave heating being the most active material in reactions tested. This catalyst presented glycerol conversion of approximately 20% with a turnover number of 9465 in a reaction time of 6 h and 58% of selectivity to glyceric acid, the main product obtained. The best performance of platinum–nickel prepared by microwave heating catalyst was attributed to the probable formation of a metallic alloy between Pt and Ni, as evidenced by the decrease in the lattice parameter for PtNi bimetallic nanoparticles. The results showed that it was possible to obtain an active catalyst in glycerol oxidation reaction under mild conditions via a simple methodology using microwave heating, which demands 94% less time in comparison with conventional heating.





Similar content being viewed by others
References
Alexander Naumkin V, Kraut-Vass A, Stephen Gaarenstroom W, CJP (2012) NIST X-ray photoelectron spectroscopy database. In: Measurement Services Division of the National Institute of Standards and Technology (NIST) Technology Services. https://srdata.nist.gov/xps/Default.aspx. Accessed 27 Feb 2018
Ambat I, Srivastava V, Sillanpää M (2018) Recent advancement in biodiesel production methodologies using various feedstock: a review. Renew Sustain Energy Rev 90:356–369. https://doi.org/10.1016/J.RSER.2018.03.069
Behr A, Eilting J, Irawadi K et al (2008) Improved utilisation of renewable resources: new important derivatives of glycerol. Green Chem 10:13–30. https://doi.org/10.1039/B710561D
Bianchi CL, Canton P, Dimitratos N et al (2005) Selective oxidation of glycerol with oxygen using mono and bimetallic catalysts based on Au, Pd and Pt metals. Catal Today 102–103:203–212. https://doi.org/10.1016/j.cattod.2005.02.003
Bock C, Paquet C, Couillard M et al (2004) Size-selected synthesis of PtRu nano-catalysts: reaction and size control mechanism. J Am Chem Soc 126:8028–8037. https://doi.org/10.1021/ja0495819
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60:309–319. https://doi.org/10.1021/ja01269a023
Chang F-W, Kuo W-Y, Lee K-C (2003) Dehydrogenation of ethanol over copper catalysts on rice husk ash prepared by incipient wetness impregnation. Appl Catal A Gen 246:253–264. https://doi.org/10.1016/S0926-860X(03)00050-4
Chen S-S, Yang Z-Z, Wang A-J et al (2018) Facile synthesis of bimetallic gold–palladium nanocrystals as effective and durable advanced catalysts for improved electrocatalytic performances of ethylene glycol and glycerol oxidation. J Colloid Interface Sci 509:10–17. https://doi.org/10.1016/j.jcis.2017.08.063
Dodekatos G, Schü Nemann S, Tü Ysü H (2018) Recent advances in thermo-, photo-, and electrocatalytic glycerol oxidation. ACS Catal. https://doi.org/10.1021/acscatal.8b01317
Dou J, Zhang B, Liu H et al (2016) Carbon supported Pt9Sn1 nanoparticles as an efficient nanocatalyst for glycerol oxidation. Appl Catal B Environ 180:78–85. https://doi.org/10.1016/j.apcatb.2015.06.007
Fuskele V, Sarviya RM (2017) Recent developments in nanoparticles synthesis, preparation and stability of nanofluids. Mater Today Proc 4:4049–4060. https://doi.org/10.1016/j.matpr.2017.02.307
Galhardo TS, Simone N, Gonçalves M et al (2013) Preparation of sulfonated carbons from rice husk and their application in catalytic conversion of glycerol. ACS Sustain Chem Eng 1:1381–1389. https://doi.org/10.1021/sc400117t
Gan Z, Zheng X, Wei D et al (2010) One-pot synthesis of one-dimensional array Pt nanoparticles on carbon nanotubes via a facile microwave polyol method. Superlattices Microstruct 47:705–709. https://doi.org/10.1016/j.spmi.2010.04.001
Gao J, Liang D, Chen P et al (2009) Oxidation of glycerol with oxygen in a base-free aqueous solution over Pt/AC and Pt/MWNTs catalysts. Catal Lett 130:185–191. https://doi.org/10.1007/s10562-009-9849-6
Garcia R, Besson M, Gallezot P (1995) Chemoselective catalytic oxidation of glycerol with air on platinum metals. Appl Catal A Gen 127:165–176. https://doi.org/10.1016/0926-860X(95)00048-8
Gebremariam SN, Marchetti JM (2018) Economics of biodiesel production: review. Energy Convers Manag 168:74–84. https://doi.org/10.1016/J.ENCONMAN.2018.05.002
Geng Z, Wang D, Zhang C et al (2014) Spillover enhanced hydrogen uptake of Pt/Pd doped corncob-derived activated carbon with ultra-high surface area at high pressure. Int J Hydrog Energy 39:13643–13649. https://doi.org/10.1016/j.ijhydene.2014.02.065
Ghodselahi T, Vesaghi MA, Shafiekhani A et al (2008) XPS study of the Cu@Cu2O core-shell nanoparticles. Appl Surf Sci 255:2730–2734. https://doi.org/10.1016/j.apsusc.2008.08.110
Gholami Z, Abdullah AZ, Lee K-T (2014) Dealing with the surplus of glycerol production from biodiesel industry through catalytic upgrading to polyglycerols and other value-added products. Renew Sustain Energy Rev 39:327–341. https://doi.org/10.1016/j.rser.2014.07.092
Gonçalves M, Souza VC, Galhardo TS et al (2013) Glycerol conversion catalyzed by carbons prepared from agroindustrial wastes. Ind Eng Chem Res 52:2832–2839. https://doi.org/10.1021/ie303072d
Gupta SK (2006) Hydroxy acid complexes for antiaging and skin renovation, US20090209642A1
Haider MH, Dummer NF, Knight DW et al (2015) Efficient green methanol synthesis from glycerol. Nat Chem 7:1028–1032. https://doi.org/10.1038/nchem.2345
Harish S, Baranton S, Coutanceau C, Joseph J (2012) Microwave assisted polyol method for the preparation of Pt/C, Ru/C and PtRu/C nanoparticles and its application in electrooxidation of methanol. J Power Sources 214:33–39. https://doi.org/10.1016/j.jpowsour.2012.04.045
Hejna A, Kosmela P, Formela K et al (2016) Potential applications of crude glycerol in polymer technology–current state and perspectives. Renew Sustain Energy Rev 66:449–475. https://doi.org/10.1016/J.RSER.2016.08.020
Hsieh C-T, Chen W-Y, Chen IL, Roy AK (2012) Deposition and activity stability of Pt–Co catalysts on carbon nanotube-based electrodes prepared by microwave-assisted synthesis. J Power Sources 199:94–102. https://doi.org/10.1016/j.jpowsour.2011.10.055
Jiang Y, Kang Q, Zhang J et al (2015) High-performance nickel–platinum nanocatalyst supported on mesoporous alumina for hydrogen generation from hydrous hydrazine. J Power Sources 273:554–560. https://doi.org/10.1016/j.jpowsour.2014.09.119
Kapkowski M, Bartczak P, Korzec M et al (2014) SiO2-, Cu-, and Ni-supported Au nanoparticles for selective glycerol oxidation in the liquid phase. J Catal 319:110–118. https://doi.org/10.1016/j.jcat.2014.08.003
Katryniok B, Kimura H, Skrzyńska E et al (2011) Selective catalytic oxidation of glycerol: perspectives for high value chemicals. Green Chem 13:1960. https://doi.org/10.1039/c1gc15320j
Katryniok B, Paul S, Dumeignil F (2013) Recent developments in the field of catalytic dehydration of glycerol to acrolein. ACS Catal 3:1819–1834. https://doi.org/10.1021/cs400354p
Kondrat SA, Miedziak PJ, Douthwaite M et al (2014) Base-Free oxidation of glycerol using titania-supported trimetallic Au–Pd–Pt nanoparticles. Chemsuschem 7:1326–1334. https://doi.org/10.1002/cssc.201300834
Kunz S (2018) Colloidal synthesis methods for the preparation of supported nanoparticles. In: Wandelt K (ed) Encyclopedia of interfacial chemistry. Elsevier, Oxford, pp 428–433. https://doi.org/10.1016/B978-0-12-409547-2.12931-2
Lebègue E, Baranton S, Coutanceau C (2011) Polyol synthesis of nanosized Pt/C electrocatalysts assisted by pulse microwave activation. J Power Sources 196:920–927. https://doi.org/10.1016/j.jpowsour.2010.08.107
Lee S, Kim HJ, Choi SM et al (2012) The promotional effect of Ni on bimetallic PtNi/C catalysts for glycerol electrooxidation. Appl Catal A Gen 429–430:39–47. https://doi.org/10.1016/j.apcata.2012.04.002
Lei J, Dong H, Duan X et al (2016) Insights into activated carbon-supported platinum catalysts for base-free oxidation of glycerol. Ind Eng Chem Res 55:420–427. https://doi.org/10.1021/acs.iecr.5b03076
Liang D, Gao J, Wang J et al (2011) Bimetallic Pt–Cu catalysts for glycerol oxidation with oxygen in a base-free aqueous solution. Catal Commun 12:1059–1062. https://doi.org/10.1016/j.catcom.2011.03.019
Marousek J, Itoh S et al (2012) The use of underwater high-voltage discharges to improve the efficiency of Jatropha curcas L. biodiesel production. Biotechnol Appl Biochem 59:451–456. https://doi.org/10.1002/bab.1045
Maroušek J, Maroušková A, Myšková K et al (2015) Techno-economic assessment of collagen casings waste management. Int J Environ Sci Technol 12:3385–3390. https://doi.org/10.1007/s13762-015-0840-z
Martin A, Richter M (2011) Oligomerization of glycerol—a critical review. Eur J Lipid Sci Technol 113:100–117. https://doi.org/10.1002/ejlt.201000386
Mathe NR, Scriba MR, Coville NJ (2014) Methanol oxidation reaction activity of microwave-irradiated and heat-treated Pt/Co and Pt/Ni nano-electrocatalysts. Int J Hydrog Energy 39:18871–18881. https://doi.org/10.1016/j.ijhydene.2014.09.070
Min M, Cho J, Cho K, Kim H (2000) Particle size and alloying effects of Pt-based alloy catalysts for fuel cell applications. Electrochim Acta 45:4211–4217. https://doi.org/10.1016/S0013-4686(00)00553-3
Misra DN (1969) Adsorption on heterogeneous surfaces: a Dubinin–Radushkevich equation. Surf Sci 18:367–372. https://doi.org/10.1016/0039-6028(69)90179-4
Olmos CM, Chinchilla LE, Rodrigues EG et al (2016) Synergistic effect of bimetallic Au–Pd supported on ceria–zirconia mixed oxide catalysts for selective oxidation of glycerol. Appl Catal B Environ 197:222–235. https://doi.org/10.1016/j.apcatb.2016.03.050
Ravindran R, Jaiswal AK (2016) Exploitation of food industry waste for high-value products. Trends Biotechnol 34:58–69. https://doi.org/10.1016/j.tibtech.2015.10.008
Sharma G, Kumar A, Sharma S et al (2017) Novel development of nanoparticles to bimetallic nanoparticles and their composites: a review. J King Saud Univ Sci. https://doi.org/10.1016/j.jksus.2017.06.012
Sheldon RA, Wallau M, Arends IWCE, Schuchardt U (1998) Heterogeneous catalysts for liquid-phase oxidations: philosophers’ stones or trojan horses? Acc Chem Res 31:485–493. https://doi.org/10.1021/ar9700163
Shen Y, Xiao K, Xi J, Qiu X (2015) Comparison study of few-layered graphene supported platinum and platinum alloys for methanol and ethanol electro-oxidation. J Power Sources 278:235–244. https://doi.org/10.1016/j.jpowsour.2014.12.062
Silva JM, Soria MA, Madeira LM (2015) Challenges and strategies for optimization of glycerol steam reforming process. Renew Sustain Energy Rev 42:1187–1213. https://doi.org/10.1016/J.RSER.2014.10.084
Sing KSW, Everett DH, Haul RAW et al (1985) Reporting physisorption data for gas solid systems with special reference to the determination of surface-area and porosity (Recommendations 1984). Pure Appl Chem 57:603–619. https://doi.org/10.1351/pac198557040603
Skrzyńska E, Ftouni J, Mamede A-S et al (2014) Glycerol oxidation over gold supported catalysts–“Two faces” of sulphur based anchoring agent. J Mol Catal A: Chem 382:71–78. https://doi.org/10.1016/J.MOLCATA.2013.11.007
Stockel R (2007) Method of treating dermatological conditions. United States Patent, US 2007/0086977 A1
Su L, Shrestha S, Zhang Z et al (2013) Platinum–copper nanotube electrocatalyst with enhanced activity and durability for oxygen reduction reactions. J Mater Chem A 1:12293–12301. https://doi.org/10.1039/C3TA13097E
Sun D, Yamada Y, Sato S, Ueda W (2016) Glycerol hydrogenolysis into useful C3 chemicals. Appl Catal B Environ 193:75–92. https://doi.org/10.1016/J.APCATB.2016.04.013
Szumełda T, Drelinkiewicz A, Lalik E et al (2018) Carbon-supported Pd100-XAuX alloy nanoparticles for the electrocatalytic oxidation of formic acid: Influence of metal particles composition on activity enhancement. Appl Catal B Environ 221:393–405. https://doi.org/10.1016/j.apcatb.2017.09.039
Thommes M, Kaneko K, Neimark AV et al (2015) Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl Chem 87:1051–1069. https://doi.org/10.1515/pac-2014-1117
Vajíček S, Štolcová M, Kaszonyi A et al (2016) Gel-type ion exchange resin stabilized Pd–Bi nanoparticles for the glycerol oxidation in liquid phase. J Ind Eng Chem 39:77–86. https://doi.org/10.1016/j.jiec.2016.05.010
Wang Y, Ren J, Deng K et al (2000) Preparation of tractable platinum, rhodium, and ruthenium nanoclusters with small particle size in organic media. Chem Mater 12:1622–1627. https://doi.org/10.1021/cm0000853
Wang F-F, Shao S, Liu C-L et al (2015) Selective oxidation of glycerol over Pt supported on mesoporous carbon nitride in base-free aqueous solution. Chem Eng J 264:336–343. https://doi.org/10.1016/j.cej.2014.11.115
Yahya MA, Al-Qodah Z, Ngah CWZ (2015) Agricultural bio-waste materials as potential sustainable precursors used for activated carbon production: a review. Renew Sustain Energy Rev 46:218–235. https://doi.org/10.1016/J.RSER.2015.02.051
Yang Z-Z, Lin X-X, Zhang X-F et al (2018) Bimetallic PtPd alloyed core-shell nanodendrites supported on reduced graphene oxide: one-pot green synthesis and efficient electrocatalytic performances for glycerol oxidation and hydrogen evolution. J Alloys Compd 735:2123–2132. https://doi.org/10.1016/j.jallcom.2017.11.322
Yu W, Porosoff MD, Chen JG (2012) Review of Pt-based bimetallic catalysis: from model surfaces to supported catalysts. Chem Rev 112:5780–5817. https://doi.org/10.1021/cr300096b
Zaid S, Skrzyńska E, Addad A et al (2017) Development of silver based catalysts promoted by noble metal M (M = Au, Pd or Pt) for glycerol oxidation in liquid phase. Top Catal 60:1072–1081. https://doi.org/10.1007/s11244-017-0800-6
Zhang M, Nie R, Wang L et al (2015a) Selective oxidation of glycerol over carbon nanofibers supported Pt catalysts in a base-free aqueous solution. Catal Commun 59:5–9. https://doi.org/10.1016/j.catcom.2014.09.036
Zhang M, Shi J, Sun Y et al (2015b) Selective oxidation of glycerol over nitrogen-doped carbon nanotubes supported platinum catalyst in base-free solution. Catal Commun 70:72–76. https://doi.org/10.1016/j.catcom.2015.08.002
Zhang C, Wang T, Liu X, Ding Y (2016) Cu-promoted Pt/activated carbon catalyst for glycerol oxidation to lactic acid. J Mol Catal A: Chem 424:91–97. https://doi.org/10.1016/j.molcata.2016.08.018
Zhou W, Zhou Z, Song S et al (2003) Pt based anode catalysts for direct ethanol fuel cells. Appl Catal B Environ 46:273–285. https://doi.org/10.1016/S0926-3373(03)00218-2
Acknowledgements
This work was supported by FAPESP (Project Numbers 2016/05006-5, 2013/15783-0). The authors are thankful to Dr. Ângela A. T. Neto (LNNano/CNPEM) and Rigaku Latin America for the facilities and the Multi-User Central Facilities (CEM/UFABC) for the experimental support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Galhardo, T.S., Gonçalves, M., Mandelli, D. et al. Glycerol valorization by base-free oxidation with air using platinum–nickel nanoparticles supported on activated carbon as catalyst prepared by a simple microwave polyol method. Clean Techn Environ Policy 20, 2075–2088 (2018). https://doi.org/10.1007/s10098-018-1593-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-018-1593-4