Abstract
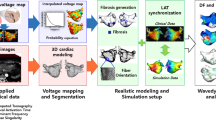
Computational models have made it possible to study the effect of fibrosis and scar on atrial fibrillation (AF) and plan future personalized treatments. Here, we study the effect of area available for fibrillatory waves to sustain AF. Then we use it to plan for AF ablation to improve procedural outcomes. CARPentry was used to create patient-specific models to determine the association between the size of residual contiguous areas available for AF wavefronts to propagate and sustain AF [fibrillatory area (FA)] after ablation with procedural outcomes. The FA was quantified in a novel manner accounting for gaps in ablation lines. We selected 30 persistent AF patients with known ablation outcomes. We divided the atrial surface into five areas based on ablation scar pattern and anatomical landmarks and calculated the FAs. We validated the models based on clinical outcomes and suggested future ablation lines that minimize the FAs and terminate rotor activities in simulations. We also simulated the effects of three common antiarrhythmic drugs. In the patient-specific models, the predicted arrhythmias matched the clinical outcomes in 25 of 30 patients (accuracy 83.33%). The average largest FA (FAmax) in the recurrence group was 8517 ± 1444 vs. 6772 ± 1531 mm2 in the no recurrence group (p < 0.004). The final FAs after adding the suggested ablation lines in the AF recurrence group reduced the average FAmax from 8517 ± 1444 to 6168 ± 1358 mm2 (p < 0.001) and stopped the sustained rotor activity. Simulations also correctly anticipated the effect of antiarrhythmic drugs in 5 out of 6 patients who used drug therapy post unsuccessful ablation (accuracy 83.33%). Sizes of FAs available for AF wavefronts to propagate are important determinants for ablation outcomes. FA size in combination with computational simulations can be used to direct ablation in persistent AF to minimize the critical mass required to sustain recurrent AF.








Similar content being viewed by others
Abbreviations
- AF:
-
Atrial fibrillation
- LGE:
-
Late gadolinium enhancement
- MRI:
-
Magnetic resonance imaging
- FA:
-
Fibrillatory area
- RF:
-
Radiofrequency
References
Andrade, J., P. Khairy, D. Dobrev, and S. Nattel. The clinical profile and pathophysiology of atrial fibrillation: relationships among clinical features, epidemiology, and mechanisms. Circ. Res. 114:1453–1468, 2014.
Boyle, P. M., T. Zghaib, S. Zahid, R. L. Ali, D. Deng, W. H. Franceschi, J. B. Hakim, M. J. Murphy, A. Prakosa, and S. L. Zimmerman. Computationally guided personalized targeted ablation of persistent atrial fibrillation. Nat. Biomed. Eng. 3:870–879, 2019.
Calkins, H., J. Brugada, D. L. Packer, R. Cappato, S.-A. Chen, H. J. Crijns, R. J. Damiano Jr., D. W. Davies, D. E. Haines, and M. Haissaguerre. Hrs/ehra/ecas Expert Consensus Statement on Catheter and Surgical Ablation of Atrial Fibrillation: Recommendations for Personnel, Policy, Procedures and Follow-up: A report of the Heart Rhythm Society (hrs) Task Force on Catheter and Surgical Ablation of Atrial Fibrillation developed in partnership with the European Heart Rhythm Association (ehra) and the European Cardiac Arrhythmia Society (ecas); in collaboration with the American College of Cardiology (acc), American Heart Association (aha), and the Society of Thoracic Surgeons (sts). Endorsed and Approved by the governing bodies of the American College of Cardiology, the American Heart Association, the European Cardiac Arrhythmia Society, the European Heart Rhythm Association, the Society of Thoracic Surgeons, and the Heart Rhythm Society. Europace. 9:335–379, 2007.
Calkins, H., K. H. Kuck, R. Cappato, J. Brugada, A. J. Camm, S.-A. Chen, H. J. Crijns, R. J. Damiano Jr., D. W. Davies, and J. DiMarco. HRS/EHRA/ECAS expert consensus statement on catheter and surgical ablation of atrial fibrillation: recommendations for patient selection, procedural techniques, patient management and follow-up, definitions, endpoints, and research trial design: a report of the Heart Rhythm Society (HRS) Task Force on Catheter and Surgical Ablation of Atrial Fibrillation. Developed in partnership with the European Heart Rhythm Association (EHRA), a registered branch of the European Society of Cardiology (ESC) and the European Cardiac Arrhythmia Society (ECAS); and in collaboration with the American College of Cardiology (ACC), American Heart Association (AHA), the Asia Pacific Heart Rhythm Society (APHRS), and the Society of Thoracic Surgeons (STS). Endorsed by the governing bodies of the American College of Cardiology Foundation, the American Heart Association, the European Cardiac Arrhythmia Society, the European Heart Rhythm Association, the Society of Thoracic Surgeons, the Asia Pacific Heart Rhythm Society, and the Heart Rhythm Society. Europace. 14:528–606, 2012.
Chubb, H., R. Karim, S. Roujol, M. Nuñez-Garcia, S. E. Williams, J. Whitaker, J. Harrison, C. Butakoff, O. Camara, and A. Chiribiri. The reproducibility of late gadolinium enhancement cardiovascular magnetic resonance imaging of post-ablation atrial scar: a cross-over study. J. Cardiovasc. Magn. Reson. 20:21, 2018.
Courtemanche, M., R. J. Ramirez, and S. Nattel. Ionic mechanisms underlying human atrial action potential properties: insights from a mathematical model. Am. J. Physiol.-Heart Circ. Physiol. 275:H301–H321, 1998.
Cox, J. L. Atrial fibrillation II: rationale for surgical treatment. J. Thorac. Cardiovasc. Surg. 126:1693–1699, 2003.
Cox J. L., R. B. Schuessler and J. P. Boineau. The development of the Maze procedure for the treatment of atrial fibrillation. In: Seminars in Thoracic and Cardiovascular Surgery. Elsevier, 2000, pp. 2–14.
Crumb, W. J., Jr., J. Vicente, L. Johannesen, and D. G. Strauss. An evaluation of 30 clinical drugs against the comprehensive in vitro proarrhythmia assay (CiPA) proposed ion channel panel. J. Pharmacol. Toxicol. Methods. 81:251–262, 2016.
Fink, T., M. Schlüter, C.-H. Heeger, C. Lemes, T. Maurer, B. Reissmann, J. Riedl, L. Rottner, F. Santoro, and B. Schmidt. Stand-alone pulmonary vein isolation versus pulmonary vein isolation with additional substrate modification as index ablation procedures in patients with persistent and long-standing persistent atrial fibrillation: the randomized Alster-Lost-AF Trial (Ablation at St. Georg Hospital for Long-Standing Persistent Atrial Fibrillation). Circulation. 10:e005114, 2017.
Haissaguerre, M., M. Hocini, A. Denis, A. J. Shah, Y. Komatsu, S. Yamashita, M. Daly, S. Amraoui, S. Zellerhoff, and M.-Q. Picat. Driver domains in persistent atrial fibrillation. Circulation. 130:530–538, 2014.
Haissaguerre, M., A. J. Shah, H. Cochet, M. Hocini, R. Dubois, I. Efimov, E. Vigmond, O. Bernus, and N. Trayanova. Intermittent drivers anchoring to structural heterogeneities as a major pathophysiological mechanism of human persistent atrial fibrillation. J. Physiol. 594:2387–2398, 2016.
Jaïs, P., B. Cauchemez, L. Macle, E. Daoud, P. Khairy, R. Subbiah, M. Hocini, F. Extramiana, F. Sacher, and P. Bordachar. Catheter ablation versus antiarrhythmic drugs for atrial fibrillation: the A4 study. Circulation. 118:2498–2505, 2008.
Kamali, R., J. Kump, E. Ghafoori, M. Lange, N. Hu, T. J. Bunch, D. J. Dosdall, R. S. Macleod, and R. Ranjan. Area available for atrial fibrillation to propagate is an important determinant of recurrence after ablation. JACC Clin. Electrophysiol. 7:896–908, 2021.
Kamali, R., J. Schroeder, E. DiBella, B. Steinberg, F. Han, D. J. Dosdall, R. S. Macleod, and R. Ranjan. Reproducibility of clinical late gadolinium enhancement magnetic resonance imaging in detecting left atrial scar after atrial fibrillation ablation. J. Cardiovasc. Electrophysiol. 31:2824–2832, 2020.
Kim, B.-S., Y.-H. Kim, G.-S. Hwang, H.-N. Pak, S. C. Lee, W. J. Shim, D. J. Oh, and Y. M. Ro. Action potential duration restitution kinetics in human atrial fibrillation. J. Am. Coll. Cardiol. 39:1329–1336, 2002.
Knecht, S., M. Hocini, M. Wright, N. Lellouche, M. D. O’Neill, S. Matsuo, I. Nault, V. S. Chauhan, K. J. Makati, and M. Bevilacqua. Left atrial linear lesions are required for successful treatment of persistent atrial fibrillation. Eur. Heart J. 29:2359–2366, 2008.
Krueger M. W., V. Schmidt, C. Tobón, F. M. Weber, C. Lorenz, D. U. Keller, H. Barschdorf, M. Burdumy, P. Neher and G. Plank. Modeling atrial fiber orientation in patient-specific geometries: a semi-automatic rule-based approach. In: International Conference on Functional Imaging and Modeling of the Heart. Springer, 2011, pp. 223–232.
McGann, C., N. Akoum, A. Patel, E. Kholmovski, P. Revelo, K. Damal, B. Wilson, J. Cates, A. Harrison, and R. Ranjan. Atrial fibrillation ablation outcome is predicted by left atrial remodeling on MRI. Circulation. 113:000689, 2013.
Oral, H., A. Chugh, E. Good, T. Crawford, J. F. Sarrazin, M. Kuhne, N. Chalfoun, D. Wells, W. Boonyapisit, and N. Gadeela. Randomized evaluation of right atrial ablation after left atrial ablation of complex fractionated atrial electrograms for long-lasting persistent atrial fibrillation. Circulation. 1:6–13, 2008.
Ouyang, F., S. Ernst, J. Chun, D. Bänsch, Y. Li, A. Schaumann, H. Mavrakis, X. Liu, F. T. Deger, and B. Schmidt. Electrophysiological findings during ablation of persistent atrial fibrillation with electroanatomic mapping and double Lasso catheter technique. Circulation. 112:3038–3048, 2005.
Parmar, B. R., T. R. Jarrett, N. S. Burgon, E. G. Kholmovski, N. W. Akoum, N. Hu, R. S. Macleod, N. F. Marrouche, and R. Ranjan. Comparison of left atrial area marked ablated in electroanatomical maps with scar in MRI. J. Cardiovasc. Electrophysiol. 25:457–463, 2014.
Perry D., A. Morris, N. Burgon, C. McGann, R. MacLeod and J. Cates. Automatic classification of scar tissue in late gadolinium enhancement cardiac MRI for the assessment of left-atrial wall injury after radiofrequency ablation. In: Medical Imaging 2012: Computer-Aided DiagnosisInternational Society for Optics and Photonics, 2012, p. 83151D.
Ramos-Mondragón, R., C. A. Galindo, and G. Avila. Role of TGF-β on cardiac structural and electrical remodeling. Vasc. Health Risk Manag. 4:1289, 2008.
Romero, J., L. Di Biase, S. Mohanty, C. Trivedi, K. Patel, M. Parides, I. Alviz, J. C. Diaz, V. Natale, and J. Sanchez. Long-term outcomes of left atrial appendage electrical isolation in patients with nonparoxysmal atrial fibrillation: a propensity score-matched analysis. Circulation. 13:e008390, 2020.
Safavi-Naeini, P., and A. Rasekh. Left atrial appendage closure and pulmonary vein isolation. Texas Heart Inst. J. 47:60–62, 2020.
Schaff H. V., J. A. Dearani, R. G. Daly, T. A. Orszulak and G. K. Danielson. Cox-Maze procedure for atrial fibrillation: Mayo Clinic experience. In: Seminars in Thoracic and Cardiovascular Surgery. Elsevier, 2000, p. 30–37.
Scherr, D., P. Khairy, S. Miyazaki, V. Aurillac-Lavignolle, P. Pascale, S. B. Wilton, K. Ramoul, Y. Komatsu, L. Roten, and A. Jadidi. Five-year outcome of catheter ablation of persistent atrial fibrillation using termination of atrial fibrillation as a procedural endpoint. Circulation. 8:18–24, 2015.
Tilz R. R., B. Schmidt, S. D. Menon, K. Chun, A. Fuernkranz, A. Metzner, B. Koektuerk, M. Konstantinidou, T. Zerm and R. Malesius. Left atrial appendage function and clinical outcome after electrical isolation of left atrial appendage in patients undergoing atrial fibrillation ablation. Am. Heart Assoc., 2008.
Verma, A., C.-Y. Jiang, T. R. Betts, J. Chen, I. Deisenhofer, R. Mantovan, L. Macle, C. A. Morillo, W. Haverkamp, and R. Weerasooriya. Approaches to catheter ablation for persistent atrial fibrillation. N. Engl. J. Med. 372:1812–1822, 2015.
Vigmond, E., R. W. dos Santos, A. J. Prassl, M. Deo, G. Plank, and S. Bauer. Solvers for the cardiac bidomain equations. Prog. Biophys. Mol. Biol. 96:3–18, 2007.
Vigmond, E. J., M. Hughes, G. Plank, and L. J. Leon. Computational tools for modeling electrical activity in cardiac tissue. J. Electrocardiol. 36:69–74, 2003.
Wazni, O. M., N. F. Marrouche, D. O. Martin, A. Verma, M. Bhargava, W. Saliba, D. Bash, R. Schweikert, J. Brachmann, and J. Gunther. Radiofrequency ablation vs antiarrhythmic drugs as first-line treatment of symptomatic atrial fibrillation: a randomized trial. Jama. 293:2634–2640, 2005.
Weimar, T., S. Schena, M. S. Bailey, H. S. Maniar, R. B. Schuessler, J. L. Cox, and R. J. Damiano Jr. The cox-maze procedure for lone atrial fibrillation: a single-center experience over 2 decades. Circulation. 5:8–14, 2012.
Williams, E. V. Classification of antidysrhythmic drugs. Pharmacol. Therap. 1:115–138, 1975.
Yamashita, K., R. Kamali, E. Kwan, R. S. MacLeod, D. J. Dosdall, and R. Ranjan. Effective ablation settings that predict chronic scar after left atrial ablation. JACC. 6:143–152, 2020.
Yorgun, H., U. Canpolat, M. Okşul, Y. Z. Şener, A. H. Ateş, H. J. Crijns, and K. Aytemir. Long-term outcomes of cryoballoon-based left atrial appendage isolation in addition to pulmonary vein isolation in persistent atrial fibrillation. EP Europace. 21:1653–1662, 2019.
Zahid, S., H. Cochet, P. M. Boyle, E. L. Schwarz, K. N. Whyte, E. J. Vigmond, R. Dubois, M. Hocini, M. Haïssaguerre, and P. Jaïs. Patient-derived models link re-entrant driver localization in atrial fibrillation to fibrosis spatial pattern. Cardiovasc. Res. 110:443–454, 2016.
Funding
RR is or reently has been a consultant to Abbott, Biosense Webster and Medtronic. The University of Utah has research grants from Biosense Webster and Abbott with RR as the PI. JB has research grants from Boehringer Ingelheim, Boston Scientific, Altathera. The remaining authors have nothing to disclose. RR is currently supported by NHLBI R01 HL142913.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript.
Additional information
Associate Editor Stefan M. Duma oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (AVI 3030 kb)
Supplementary file3 (AVI 3149 kb)
Supplementary file4 (AVI 3957 kb)
Supplementary file5 (AVI 3768 kb)
Supplementary file6 (AVI 895 kb)
Supplementary file7 (AVI 846 kb)
Rights and permissions
About this article
Cite this article
Kamali, R., Gillete, K., Tate, J. et al. Treatment Planning for Atrial Fibrillation Using Patient-Specific Models Showing the Importance of Fibrillatory-Areas. Ann Biomed Eng 51, 329–342 (2023). https://doi.org/10.1007/s10439-022-03029-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-022-03029-5