Abstract
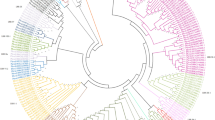
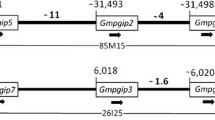
Disease-resistant genes (R genes) encode proteins that are involved in protecting plants from their pathogens and pests. Availability of complete genome sequences from soybean and common bean allowed us to perform a genome-wide identification and analysis of the Toll interleukin-1 receptor-like nucleotide-binding site leucine-rich repeat (TNL) proteins. Hidden Markov model (HMM) profiling of all protein sequences resulted in the identification of 117 and 77 regular TNL genes in soybean and common bean, respectively. We also identified TNL gene homologs with unique domains, and signal peptides as well as nuclear localization signals. The TNL genes in soybean formed 28 clusters located on 10 of the 20 chromosomes, with the majority found on chromosome 3, 6 and 16. Similarly, the TNL genes in common bean formed 14 clusters located on five of the 11 chromosomes, with the majority found on chromosome 10. Phylogenetic analyses of the TNL genes from Arabidopsis, soybean and common bean revealed less divergence within legumes relative to the divergence between legumes and Arabidopsis. Syntenic blocks were found between chromosomes Pv10 and Gm03, Pv07 and Gm10, as well as Pv01 and Gm14. The gene expression data revealed basal level expression and tissue specificity, while analysis of available microRNA data showed 37 predicted microRNA families involved in targeting the identified TNL genes in soybean and common bean.




Similar content being viewed by others
References
Ameline-Torregrosa C, Wang BB, O’Bleness MS, Deshpande S, Zhu H, Roe B, Young ND, Cannon SB (2008) Identification and characterization of nucleotide-binding site-leucine-rich repeat genes in the model plant Medicago truncatula. Plant Physiol. https://doi.org/10.1104/pp.107.104588
Andersen EJ, Nepal MP (2017) Genetic diversity of disease resistance genes in foxtail millet (Setaria italica L.). Plant Gene 10:8–16
Andersen EJ, Ali S, Reese RN, Yen Y, Neupane S, Nepal MP (2016) Diversity and evolution of disease resistance genes in Barley (Hordeum vulgare L.). Evol Bioinfor Online 12:99
Anderson PA, Lawrence GJ, Morrish BC, Ayliffe MA, Finnegan EJ, Ellis JG (1997) Inactivation of the flax rust resistance gene M associated with loss of a repeated unit within the leucine-rich repeat coding region. Plant Cell 9:641–651
Andolfo G, Ercolano MR (2015) Plant innate immunity multicomponent model. Front Plant Sci 6:987
Andolfo G, Sanseverino W, Aversano R, Frusciante L, Ercolano M (2014) Genome-wide identification and analysis of candidate genes for disease resistance in tomato. Mol Breed 33:227–233
Arumuganathan K, Earle E (1991) Nuclear DNA content of some important plant species. Plant Mol Biol Report 9:208–218
Arya P, Kumar G, Acharya V, Singh AK (2014) Genome-wide identification and expression analysis of NBS-encoding genes in Malus x domestica and expansion of NBS genes family in Rosaceae. PLoS ONE 9:e107987. https://doi.org/10.1371/journal.pone.0107987
Asai T, Tena G, Plotnikova J, Willmann MR, Chiu WL, Gomez-Gomez L, Boller T, Ausubel FM, Sheen J (2002) MAP kinase signalling cascade in Arabidopsis innate immunity. Nature 415:977–983. https://doi.org/10.1038/415977a
Ashfield T, Egan AN, Pfeil BE, Chen NW, Podicheti R, Ratnaparkhe MB, Ameline-Torregrosa C, Denny R, Cannon S, Doyle JJ (2012) Evolution of a complex disease resistance gene cluster in diploid Phaseolus and tetraploid Glycine. Plant Physiol 159:336–354
Axtell MJ (2013) Classification and comparison of small RNAs from plants. Annu Rev Plant Biol 64:137–159
Ba ANN, Pogoutse A, Provart N, Moses AM (2009) NLStradamus: a simple Hidden Markov Model for nuclear localization signal prediction. BMC Bioinformatics 10:1
Bailey TL, Elkan C (1994) Fitting a mixture model by expectation maximization to discover motifs in bipolymers. Proc Int Syst Mol Biol 2:28–36
Bales C, Zhang G, Liu M, Mensah C, Gu C, Song Q, Hyten D, Cregan P, Wang D (2013) Mapping soybean aphid resistance genes in PI 567598B. Theor Appl Genet 126:2081–2091
Bennett MD, Smith J (1976) Nuclear DNA amounts in angiosperms. Phil Trans Soc B 274:227–274
Benson B (2014) Disease resistance genes and their evolutionary history in six plant species. South Dakota State University, Brookings, SD
Bonierbale MW, Plaisted RL, Tanksley SD (1988) RFLP maps based on a common set of clones reveal modes of chromosomal evolution in potato and tomato. Genetics 120:1095–1103
Botella MA, Parker JE, Frost LN, Bittner-Eddy PD, Beynon JL, Daniels MJ, Holub EB, Jones JD (1998) Three genes of the Arabidopsis RPP1 complex resistance locus recognize distinct Peronospora parasitica avirulence determinants. Plant Cell 10:1847–1860
Caicedo AL, Purugganan MD (2005) Comparative plant genomics. Frontiers and prospects. Plant Physiol 138:545–547
Cesari S, Bernoux M, Moncuquet P, Kroj T, Dodds PN (2014) A novel conserved mechanism for plant NLR protein pairs: the “integrated decoy” hypothesis. Front Plant Sci 5(10):3389
Chen NW, Sévignac M, Thareau V, Magdelenat G, David P, Ashfield T, Innes RW, Geffroy V (2010) Specific resistances against Pseudomonas syringae effectors AvrB and AvrRpm1 have evolved differently in common bean (Phaseolus vulgaris), soybean (Glycine max), and Arabidopsis thaliana. New Phytol 187:941–956
Choi H-K, Mun J-H, Kim D-J, Zhu H, Baek J-M, Mudge J, Roe B, Ellis N, Doyle J, Kiss GB (2004) Estimating genome conservation between crop and model legume species. Proc Natl Acad Sci USA 101:15289–15294
Christie N, Tobias PA, Naidoo S, Külheim C (2016) The Eucalyptus grandis NBS-LRR gene family: physical clustering and expression hotspots. Front Plant Sci. https://doi.org/10.3389/fpls.2015.01238
Collier SM, Hamel L-P, Moffett P (2011) Cell death mediated by the N-terminal domains of a unique and highly conserved class of NB-LRR protein. Mol Plant Microbe Interact 24:918–931
Cui X, Yan Q, Gan S, Xue D, Dou D, Guo N, Xing H (2017) Overexpression of gma-miR1510a/b suppresses the expression of a NB-LRR domain gene and reduces resistance to Phytophthora sojae. Gene 621:32–39
Dai X, Zhao PX (2011) psRNATarget: a plant small RNA target analysis server. Nucleic Acids Res 39:W155–W159
Dangl JL, Jones JD (2001) Plant pathogens and integrated defense responses to infection. Nature 411:826–833
DeYoung BJ, Innes RW (2006) Plant NBS-LRR proteins in pathogen sensing and host defense. Nat Immunol 7:1243–1249
Dodds PN, Lawrence GJ, Ellis JG (2001) Six amino acid changes confined to the leucine-rich repeat β-strand/β-turn motif determine the difference between the P and P2 rust resistance specificities in flax. Plant Cell 13:163–178
Du J, Tian Z, Sui Y, Zhao M, Song Q, Cannon SB, Cregan P, Ma J (2012) Pericentromeric effects shape the patterns of divergence, retention, and expression of duplicated genes in the paleopolyploid soybean. Plant Cell 24:21–32
Duc G, Agrama H, Bao S, Berger J, Bourion V, De Ron AM, Gowda CL, Mikic A, Millot D, Singh KB (2015) Breeding annual grain legumes for sustainable agriculture: new methods to approach complex traits and target new cultivar ideotypes. Crit Rev Plant Sci 34:381–411
Ellis JG, Lawrence GJ, Luck JE, Dodds PN (1999) Identification of regions in alleles of the flax rust resistance gene L that determine differences in gene-for-gene specificity. Plant cell 11:495–506
Emanuelsson O, Brunak S, von Heijne G, Nielsen H (2007) Locating proteins in the cell using TargetP, SignalP and related tools. Nat Protoc 2:953–971
Fei Q, Xia R, Meyers BC (2013) Phased, secondary, small interfering RNAs in posttranscriptional regulatory networks. Plant Cell 25:2400–2415
Fei Q, Li P, Teng C, Meyers BC (2015) Secondary siRNAs from Medicago NB-LRRs modulated via miRNA–target interactions and their abundances. Plant J 83:451–465
Finn RD, Bateman A, Clements J, Coggill P, Eberhardt RY, Eddy SR, Heger A, Hetherington K, Holm L, Mistry J (2013) Pfam: the protein families database. Nucleic Acids Res 42:D222–D231
Finn RD, Clements J, Arndt W, Miller BL, Wheeler TJ, Schreiber F, Bateman A, Eddy SR (2015) HMMER web server: 2015 update. Nucleic Acids Res 43:W30–W38
Friedman AR, Baker BJ (2007) The evolution of resistance genes in multi-protein plant resistance systems. Curr Opin Genet Dev. https://doi.org/10.1016/j.gde.2007.08.014
Gao H, Bhattacharyya MK (2008) The soybean-Phytophthora resistance locus Rps1-k encompasses coiled coil-nucleotide binding-leucine rich repeat-like genes and repetitive sequences. BMC Plant Biol 8:29
Gao H, Narayanan NN, Ellison L, Bhattacharyya MK (2005) Two classes of highly similar coiled coil-nucleotide binding-leucine rich repeat genes isolated from the Rps1-k locus encode Phytophthora resistance in soybean. Mol Plant Microbe Interact 18:1035–1045
Gassmann W, Hinsch ME, Staskawicz BJ (1999) The Arabidopsis RPS4 bacterial-resistance gene is a member of the TIR-NBS-LRR family of disease-resistance genes. Plant J 20:265–277
Glazebrook J (2001) Genes controlling expression of defense responses in Arabidopsis—2001 status. Curr Opin Plant Biol 4:301–308
Goellner K, Loehrer M, Langenbach C, Conrath U, Koch E, Schaffrath U (2010) Phakopsora pachyrhizi, the causal agent of Asian soybean rust. Mol Plant Pathol 11:169–177. https://doi.org/10.1111/j.1364-3703.2009.00589.x
Goff SA, Ricke D, Lan T-H, Presting G, Wang R, Dunn M, Glazebrook J, Sessions A, Oeller P, Varma H (2002) A draft sequence of the rice genome (Oryza sativa L. ssp. japonica). Science 296:92–100
González VM, Müller S, Baulcombe D, Puigdomènech P (2015) Evolution of NBS-LRR gene copies among dicot plants and its regulation by members of the miR482/2118 superfamily of miRNAs. Mol Plant 8:329–331
Guo Y-L, Fitz J, Schneeberger K, Ossowski S, Cao J, Weigel D (2011) Genome-wide comparison of nucleotide-binding site-leucine-rich repeat-encoding genes in Arabidopsis. Plant Physiol 157:757–769
Gururani MA, Venkatesh J, Upadhyaya CP, Nookaraju A, Pandey SK, Park SW (2012) Plant disease resistance genes: current status and future directions. Physiol Mol Plant Pathol 78:51–65. https://doi.org/10.1016/j.pmpp.2012.01.002
Hartman G, Domier L, Wax L, Helm C, Onstad D, Shaw J, Solter L, Voegtlin D, d’Arcy C, Gray M (2001) Occurrence and distribution of Aphis glycines on soybeans in Illinois in 2000 and its potential control. Plant Health Progr 74:33–37
He C-Y, Tian A-G, Zhang J-S, Zhang Z-Y, Gai J-Y, Chen S-Y (2003) Isolation and characterization of a full-length resistance gene homolog from soybean. Theor Appl Genet 106:786–793
Hill C, Chirumamilla A, Hartman G (2012) Resistance and virulence in the soybean-Aphis glycines interaction. Euphytica 186:635–646
Howe EA, Sinha R, Schlauch D, Quackenbush J (2011) RNA-Seq analysis in MeV. Bioinformatics 27:3209–3210
Hulbert SH, Webb CA, Smith SM, Sun Q (2001) Resistance gene complexes. Annu Rev Phytopathol 39:285–312
Jain S, Chittem K, Brueggeman R, Osorno JM, Richards J, Nelson BD Jr (2016) Comparative transcriptome analysis of resistant and susceptible common bean genotypes in response to soybean cyst nematode infection. PLoS ONE 11:e0159338
Jones JD, Dangl JL (2006) The plant immune system. Nature 444:323–329. https://doi.org/10.1038/nature05286
Jones P, Binns D, Chang H-Y, Fraser M, Li W, McAnulla C, McWilliam H, Maslen J, Mitchell A, Nuka G (2014) InterProScan 5: genome-scale protein function classification. Bioinformatics 30:1236–1240
Jun T-H, Mian MR, Michel AP (2012) Genetic mapping revealed two loci for soybean aphid resistance in PI 567301B. Theor Appl Genet 124:13–22
Jupe F, Pritchard L, Etherington GJ, MacKenzie K, Cock PJ, Wright F, Sharma SK, Bolser D, Bryan GJ, Jones JD (2012) Identification and localisation of the NB-LRR gene family within the potato genome. BMC Genomics 13:1
Kang YJ, Kim KH, Shim S, Yoon MY, Sun S, Kim MY, Van K, Lee S-H (2012) Genome-wide mapping of NBS-LRR genes and their association with disease resistance in soybean. BMC Plant Biol 12:1
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C (2012) Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649
Kim K-S, Bellendir S, Hudson KA, Hill CB, Hartman GL, Hyten DL, Hudson ME, Diers BW (2010a) Fine mapping the soybean aphid resistance gene Rag1 in soybean. Theor Appl Genet 120:1063–1071
Kim K-S, Hill CB, Hartman GL, Hyten DL, Hudson ME, Diers BW (2010b) Fine mapping of the soybean aphid-resistance gene Rag2 in soybean PI 200538. Theor Appl Genet 121:599–610
Kohler A, Rinaldi C, Duplessis S, Baucher M, Geelen D, Duchaussoy F, Meyers BC, Boerjan W, Martin F (2008) Genome-wide identification of NBS resistance genes in Populus trichocarpa. Plant Mol Biol. https://doi.org/10.1007/s11103-008-9293-9
Kozomara A, Griffiths-Jones S (2014) miRBase: annotating high confidence microRNAs using deep sequencing data. Nucleic Acids Res 42:D68–D73
Kuang H, Caldwell KS, Meyers BC, Michelmore RW (2008) Frequent sequence exchanges between homologs of RPP8 in Arabidopsis are not necessarily associated with genomic proximity. Plant J 54:69–80
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874
Kuwabara C, Arakawa K, Yoshida S (1999) Abscisic acid-induced secretory proteins in suspension-cultured cells of winter wheat. Plant Cell Physiol 40:184–191
Lahaye T (2002) The Arabidopsis RRS1-R disease resistance gene–uncovering the plant’s nucleus as the new battlefield of plant defense? Trends Plant Sci 7:425–427
Larkin MA, Blackshields G, Brown N, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948
Lavin M, Herendeen PS, Wojciechowski MF (2005) Evolutionary rates analysis of Leguminosae implicates a rapid diversification of lineages during the Tertiary. Syst Biol 54:575–594
Lawrence GJ, Finnegan EJ, Ayliffe MA, Ellis JG (1995) The L6 gene for flax rust resistance is related to the Arabidopsis bacterial resistance gene RPS2 and the tobacco viral resistance gene N. Plant Cell 7:1195–1206
Leister D (2004) Tandem and segmental gene duplication and recombination in the evolution of plant disease resistance genes. Trends Genet 20:116–122
Li J, Ding J, Zhang W, Zhang Y, Tang P, Chen JQ, Tian D, Yang S (2010) Unique evolutionary pattern of numbers of gramineous NBS-LRR genes. Mol Genet Genomics 283:427–438. https://doi.org/10.1007/s00438-010-0527-6
Li X, Kapos P, Zhang Y (2015) NLRs in plants. Curr Opin Immunol 32:114–121. https://doi.org/10.1016/j.coi.2015.01.014
Lozano R, Ponce O, Ramirez M, Mostajo N, Orjeda G (2012) Genome-wide identification and mapping of NBS-encoding resistance genes in Solanum tuberosum group phureja. PLoS ONE 7:e34775
Lozano R, Hamblin MT, Prochnik S, Jannink JL (2015) Identification and distribution of the NBS-LRR gene family in the Cassava genome. BMC Genomics 16:360. https://doi.org/10.1186/s12864-015-1554-9
Luo S, Zhang Y, Hu Q, Chen J, Li K, Lu C, Liu H, Wang W, Kuang H (2012) Dynamic nucleotide-binding site and leucine-rich repeat-encoding genes in the grass family. Plant Physiol 159:197–210. https://doi.org/10.1104/pp.111.192062
Marchler-bauer A, Derbyshire MK, Gonzales NR, Lu S, Chitsaz F, Geer LY, Geer RC, He J, Gwadz M, Hurwitz DI (2015) CDD: NCBI’s conserved domain database. Nucleic Acids Res 43:D222–D226
Marone D, Russo MA, Laido G, De Leonardis AM, Mastrangelo AM (2013) Plant nucleotide binding site-leucine-rich repeat (NBS-LRR) genes: active guardians in host defense responses. Int J Mol Sci 14:7302–7326. https://doi.org/10.3390/ijms14047302
McClean PE, Mamidi S, McConnell M, Chikara S, Lee R (2010) Synteny mapping between common bean and soybean reveals extensive blocks of shared loci. BMC Genomics 11:184
Meyers BC, Morgante M, Michelmore RW (2002) TIR-X and TIR-NBS proteins: two new families related to disease resistance TIR-NBS-LRR proteins encoded in Arabidopsis and other plant genomes. Plant J 32:77–92
Meyers BC, Kozik A, Griego A, Kuang H, Michelmore RW (2003) Genome-wide analysis of NBS-LRR-encoding genes in Arabidopsis. Plant Cell. https://doi.org/10.1105/tpc.009308
Michelmore RW, Meyers BC (1998) Clusters of resistance genes in plants evolve by divergent selection and a birth-and-death process. Genome Res 8:1113–1130
Milos V, Vuk D, Kristina P, Jegor M (2013) Review of soybean resistance to pathogens. Ratarstvo i povrtarstvo 50:52–61. https://doi.org/10.5937/ratpov50-4038
Mun J-H, Yu H-J, Park S, Park B-S (2009) Genome-wide identification of NBS-encoding resistance genes in Brassica rapa. Mol Genet Genomics 282:617–631
Namayanja A, Buruchara R, Mahuku G, Rubaihayo P, Kimani P, Mayanja S, Eyedu H (2006) Inheritance of resistance to angular leaf spot in common bean and validation of the utility of resistance linked markers for marker assisted selection out side the mapping population. Euphytica 151:361–369
Nandety RS, Caplan JL, Cavanaugh K, Perroud B, Wroblewski T, Michelmore RW, Meyers BC (2013) The role of TIR-NBS and TIR-X proteins in plant basal defense responses. Plant Physiol 162:1459–1472
Nepal MP, Benson BV (2015) CNL disease resistance genes in soybean and their evolutionary divergence. Evol Bioinform Online 11:49–63. https://doi.org/10.4137/EBO.S21782
Noël L, Moores TL, Van Der Biezen EA, Parniske M, Daniels MJ, Parker JE, Jones JD (1999) Pronounced intraspecific haplotype divergence at the RPP5 complex disease resistance locus of Arabidopsis. Plant Cell 11:2099–2111
O’Rourke JA, Iniguez LP, Fu F, Bucciarelli B, Miller SS, Jackson SA, McClean PE, Li J, Dai X, Zhao PX (2014) An RNA-Seq based gene expression atlas of the common bean. BMC Genomics 15:866
Orgil U, Araki H, Tangchaiburana S, Berkey R, Xiao S (2007) Intraspecific genetic variations, fitness cost and benefit of RPW8, a disease resistance locus in Arabidopsis thaliana. Genetics 176:2317–2333
Pan Q, Liu Y-S, Budai-Hadrian O, Sela M, Carmel-Goren L, Zamir D, Fluhr R (2000) Comparative genetics of nucleotide binding site-leucine rich repeat resistance gene homologues in the genomes of two dicotyledons: tomato and Arabidopsis. Genetics 155:309–322
Peart JR, Mestre P, Lu R, Malcuit I, Baulcombe DC (2005) NRG1, a CC-NB-LRR protein, together with N, a TIR-NB-LRR protein, mediates resistance against tobacco mosaic virus. Curr Biol 15:968–973
Peele HM, Guan N, Fogelqvist J, Dixelius C (2014) Loss and retention of resistance genes in five species of the Brassicaceae family. BMC Plant Biol 14:1
Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786
Pfeil B, Schlueter JA, Shoemaker R, Doyle J (2005) Placing paleopolyploidy in relation to taxon divergence: a phylogenetic analysis in legumes using 39 gene families. Sys Biol 54:441–454
Plocik A, Layden J, Kesseli R (2004) Comparative analysis of NBS domain sequences of NBS-LRR disease resistance genes from sunflower, lettuce, and chicory. Mol Phylogenet Evol 31:153–163
Pontes O, Neves N, Silva M, Lewis MS, Madlung A, Comai L, Viegas W, Pikaard CS (2004) Chromosomal locus rearrangements are a rapid response to formation of the allotetraploid Arabidopsis suecica genome. Proc Natl Acad Sci USA 101:18240–18245
Rathi D, Gayen D, Gayali S, Chakraborty S, Chakraborty N (2016) Legume proteomics: progress, prospects, and challenges. Proteomics 16:310–327
Ratnaparkhe MB, Wang X, Li J, Compton RO, Rainville LK, Lemke C, Kim C, Tang H, Paterson AH (2011) Comparative analysis of peanut NBS-LRR gene clusters suggests evolutionary innovation among duplicated domains and erosion of gene microsynteny. New Phytol 192:164–178
Reams AB, Neidle EL (2004) Selection for gene clustering by tandem duplication. Annu Rev Microbiol 58:119–142
Rozas J (2009) DNA sequence polymorphism analysis using DnaSP. Bioinformatics for DNA sequence analysis. Mehods Mol Biol 537:337–350
Sartorato A, Nietsche S, Barros EG, Moreira MA (2000) RAPD and SCAR markers linked to resistance gene to angular leaf spot in common beans. Fitopatol Bras 25:637–642
Savard L, Li P, Strauss SH, Chase MW, Michaud M, Bousquet J (1994) Chloroplast and nuclear gene sequences indicate late Pennsylvanian time for the last common ancestor of extant seed plants. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.91.11.5163
Schatz MC, Witkowski J, McCombie WR (2012) Current challenges in de novo plant genome sequencing and assembly. Genome Biol 13:243
Schlueter JA, Dixon P, Granger C, Grant D, Clark L, Doyle JJ, Shoemaker RC (2004) Mining EST databases to resolve evolutionary events in major crop species. Genome 47:868–876
Schmutz J, Cannon SB, Schlueter J, Ma J, Mitros T, Nelson W, Hyten DL, Song Q, Thelen JJ, Cheng J (2010) Genome sequence of the palaeopolyploid soybean. Nature 463:178–183
Schmutz J, McClean PE, Mamidi S, Wu GA, Cannon SB, Grimwood J, Jenkins J, Shu S, Song Q, Chavarro C (2014) A reference genome for common bean and genome-wide analysis of dual domestications. Nat Genet 46:707–713
Schulze-Lefert P, Panstruga R (2011) A molecular evolutionary concept connecting nonhost resistance, pathogen host range, and pathogen speciation. Trends Plant Sci 16:117–125
Severin AJ, Woody JL, Bolon Y-T, Joseph B, Diers BW, Farmer AD, Muehlbauer GJ, Nelson RT, Grant D, Specht JE (2010) RNA-Seq Atlas of Glycine max: a guide to the soybean transcriptome. BMC Plant Biol 10:160
Shao Z-Q, Zhang Y-M, Hang Y-Y, Xue J-Y, Zhou G-C, Wu P, Wu X-Y, Wu X-Z, Wang Q, Wang B (2014) Long-term evolution of nucleotide-binding site-leucine-rich repeat genes: understanding gained from and beyond the legume family. Plant Physiol 166:217–234
Shao Z-Q, Xue J-Y, Wu P, Zhang Y-M, Wu Y, Hang Y-Y, Wang B, Chen J-Q (2016) Large-scale analyses of angiosperm nucleotide-binding site-leucine-rich repeat (NBS-LRR) genes reveal three anciently diverged classes with distinct evolutionary patterns. Plant Physiol 170:15
Shivaprasad PV, Chen H-M, Patel K, Bond DM, Santos BA, Baulcombe DC (2012) A microRNA superfamily regulates nucleotide binding site–leucine-rich repeats and other mRNAs. Plant Cell 24:859–874
Soderlund C, Bomhoff M, Nelson WM (2011) SyMAP v3. 4: a turnkey synteny system with application to plant genomes. Nucleic Acids Res 39:e68–e76
Souza TLP, Faleiro FG, Dessaune SN, Paula-Junior TJd, Moreira MA, Barros EGd (2013) Breeding for common bean (Phaseolus vulgaris L.) rust resistance in Brazil. Trop Plant Pathol 38:361–374
Sprent JI (2007) Evolving ideas of legume evolution and diversity: a taxonomic perspective on the occurrence of nodulation. New Phytol 174:11–25
Stokes TL, Kunkel BN, Richards EJ (2002) Epigenetic variation in Arabidopsis disease resistance. Genes Dev 16:171–182
Takemoto D, Rafiqi M, Hurley U, Lawrence GJ, Bernoux M, Hardham AR, Ellis JG, Dodds PN, Jones DA (2012) N-terminal motifs in some plant disease resistance proteins function in membrane attachment and contribute to disease resistance. Mol Plant-Microbe Interact 25:379–392
Taylor JS, Raes J (2004) Duplication and divergence: the evolution of new genes and old ideas. Annu Rev Genet 38:615–643
Tian D, Traw M, Chen J, Kreitman M, Bergelson J (2003) Fitness costs of R-gene-mediated resistance in Arabidopsis thaliana. Nature 423:74–77
Tian Y, Fan L, Thurau T, Jung C, Cai D (2004) The absence of TIR-type resistance gene analogues in the sugar beet (Beta vulgaris L.) genome. J Mol Evol 58:40–53
Van Der Biezen EA, Freddie CT, Kahn K, Jones JD (2002) Arabidopsis RPP4 is a member of the RPP5 multigene family of TIR-NB-LRR genes and confers downy mildew resistance through multiple signalling components. Plant J 29:439–451
Wan H, Yuan W, Bo K, Shen J, Pang X, Chen J (2013) Genome-wide analysis of NBS-encoding disease resistance genes in Cucumis sativus and phylogenetic study of NBS-encoding genes in Cucurbitaceae crops. BMC Genomics 14:1
Wei H, Li W, Sun X, Zhu S, Zhu J (2013) Systematic analysis and comparison of nucleotide-binding site disease resistance genes in a diploid cotton Gossypium raimondii. PLoS ONE 8:e68435. https://doi.org/10.1371/journal.pone.0068435
Whitham S, Dinesh-Kumar S, Choi D, Hehl R, Corr C, Baker B (1994) The product of the tobacco mosaic virus resistance gene N: similarity to toll and the interleukin-1 receptor. Cell 78:1101–1115
Whitkus R, Doebley J, Lee M (1992) Comparative genome mapping of Sorghum and maize. Genetics 132:1119–1130
Yang S, Zhang X, Yue J-X, Tian D, Chen J-Q (2008) Recent duplications dominate NBS-encoding gene expansion in two woody species. Mol Genet Genomics 280:187–198
Yang S, Tang F, Gao M, Krishnan HB, Zhu H (2010) R gene-controlled host specificity in the legume–rhizobia symbiosis. Proc Natl Acad Sci USA 107:18735–18740
Yang L, Li D, Li Y, Gu X, Huang S, Garcia-Mas J, Weng Y (2013) A 1,681-locus consensus genetic map of cultivated cucumber including 67 NB-LRR resistance gene homolog and ten gene loci. BMC Plant Biol 13:1
Yu J, Tehrim S, Zhang F, Tong C, Huang J, Cheng X, Dong C, Zhou Y, Qin R, Hua W, Liu S (2014) Genome-wide comparative analysis of NBS-encoding genes between Brassica species and Arabidopsis thaliana. BMC Genomics 15:1–18. https://doi.org/10.1186/1471-2164-15-3
Yue JX, Meyers BC, Chen JQ, Tian D, Yang S (2012) Tracing the origin and evolutionary history of plant nucleotide-binding site–leucine-rich repeat (NBS-LRR) genes. New Phytol 193:1049–1063
Zhai J, Jeong D-H, De Paoli E, Park S, Rosen BD, Li Y, González AJ, Yan Z, Kitto SL, Grusak MA (2011) MicroRNAs as master regulators of the plant NB-LRR defense gene family via the production of phased, trans-acting siRNAs. Genes Dev 25:2540–2553
Zhang G, Gu C, Wang D (2009) Molecular mapping of soybean aphid resistance genes in PI 567541B. Theor Appl Genet 118:473–482
Zhang G, Gu C, Wang D (2010) A novel locus for soybean aphid resistance. Theor Appl Genet 120:1183–1191
Zhang Y, Xia R, Kuang H, Meyers BC (2016a) The diversification of plant NBS-LRR defense genes directs the evolution of microRNAs that target them. Mol Biol Evol 33:2692–2705
Zhang YM, Shao ZQ, Wang Q, Hang YY, Xue JY, Wang B, Chen JQ (2016b) Uncovering the dynamic evolution of nucleotide-binding site-leucine-rich repeat (NBS-LRR) genes in Brassicaceae. J Integr Plant Biol 58:165–177
Zheng F, Wu H, Zhang R, Li S, He W, Wong F-L, Li G, Zhao S, Lam H-M (2016) Molecular phylogeny and dynamic evolution of disease resistance genes in the legume family. BMC Genomics 17:1
Zhu H, Choi H-K, Cook DR, Shoemaker RC (2005) Bridging model and crop legumes through comparative genomics. Plant Physiol 137:1189–1196
Acknowledgements
This project was supported by South Dakota Agricultural Experiment Station (SDAES) USDA-NIFA hatch Project to M. Nepal (SD00H469-13) and South Dakota Soybean Research and Promotion Council (SDSRPC-SA1800238).
Author information
Authors and Affiliations
Contributions
SN conducted gene identification and analysis. MPN conceived and supervised the project as well as helped SN draft the manuscript. QM, FM, EJA and AV contributed to data analysis, interpretation and revision of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Neupane, S., Ma, Q., Mathew, F.M. et al. Evolutionary Divergence of TNL Disease-Resistant Proteins in Soybean (Glycine max) and Common Bean (Phaseolus vulgaris). Biochem Genet 56, 397–422 (2018). https://doi.org/10.1007/s10528-018-9851-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-018-9851-z