Abstract
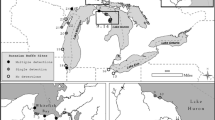
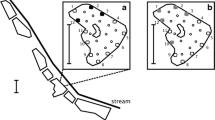
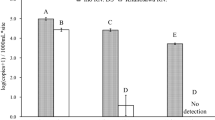
Containing the spread of established invasive species is critical for minimizing their ecological impact. Effective containment requires sensitive sampling methods capable of detecting new introductions when invaders are at low density. Here we explore whether environmental DNA (eDNA) sampling could be used as a surveillance tool to detect new incursions of aquatic invasive species on offshore islands. We develop an eDNA molecular assay for invasive cane toads (Rhinella marina) in Australia, validate our assay on the mainland, and apply it to an offshore island (Moreton Island) that is a target of ongoing cane toad surveillance. Our eDNA assay correctly identified four mainland sites at which cane toads were observed, as well as a fifth site within 1 km of known populations. Five additional sites outside the cane toad’s current distribution tested negative for cane toad eDNA. Site occupancy detection models indicated that two water samples and three qPCR replicates were sufficient to achieve a cumulate detection probability > 0.95. Applying our eDNA assay to samples from 19 sites on an offshore island over a 2-year period revealed the absence of cane toad eDNA, in line with our current understanding of cane toad distribution. Our results suggest that eDNA sampling could be strategically applied to meet the Australian Commonwealth’s objective of maintaining cane toad-free offshore islands.


Similar content being viewed by others
References
Adrian-Kalchhauser I, Burkhardt-Holm P (2016) An eDNA assay to monitor a globally invasive fish species from flowing freshwater. PLoS ONE 11:e0147558. https://doi.org/10.1371/journal.pone.0147558
Commonwealth of Australia (2011) Threat abatement plan for the biological effects, including lethal toxic ingestion, caused by cane toads. Department of Sustainability, Environment, Water, Population and Communities, Canberra, Australia
Dejean T, Valentini A, Miquel C et al (2012) Improved detection of an alien invasive species through environmental DNA barcoding: the example of the American bullfrog Lithobates catesbeianus. J Appl Ecol 49:953–959. https://doi.org/10.1111/j.1365-2664.2012.02171.x
Dougherty MM, Larson ER, Renshaw MA et al (2016) Environmental DNA (eDNA) detects the invasive rusty crayfish Orconectes rusticus at low abundances. J Appl Ecol 53:722–732. https://doi.org/10.1111/1365-2664.12621
Epanchin-Niell RS, Hastings A (2010) Controlling established invaders: integrating economics and spread dynamics to determine optimal management. Ecol Lett 13:528–541. https://doi.org/10.1111/j.1461-0248.2010.01440.x
Kearney M, Phillips BL, Tracy CR et al (2008) Modelling species distributions without using species distributions: the cane toad in Australia under current and future climates. Ecography 31:423–434. https://doi.org/10.1111/j.0906-7590.2008.05457.x
Lahoz-Monfort JJ, Guillera-Arroita G, Tingley R (2016) Statistical approaches to account for false-positive errors in environmental DNA samples. Mol Ecol Resour 16:673–685. https://doi.org/10.1111/1755-0998.12486
Lugg WH, Griffiths J, van Rooyen AR et al (2018) Optimal survey designs for environmental DNA sampling. Methods Ecol Evol 9:1049–1059. https://doi.org/10.1111/2041-210X.12951
Pilliod DS, Goldberg CS, Arkle RS et al (2013) Estimating occupancy and abundance of stream amphibians using environmental DNA from filtered water samples. Can J Fish Aquat Sci 70:1123–1130. https://doi.org/10.1139/cjfas-2013-0047
Plummer M (2003) JAGS: A program for analysis of Bayesian graphical models using Gibbs sampling. Proc Int Workshop Distrib stat Comput 124:125
R Core Team (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Schmidt BR, Kery M, Ursenbacher S et al (2013) Site occupancy models in the analysis of environmental DNA presence/absence surveys: a case study of an emerging amphibian pathogen. Methods Ecol Evol 4:646–653. https://doi.org/10.1111/2041-210X.12052
Slade RW, Moritz C (1998) Phylogeography of Bufo marinus from its natural and introduced ranges. Proc Royal Soc B: Biol Sci 265(1398):769–777
Smart AS, Weeks AR, van Rooyen AR et al (2015) Environmental DNA sampling is more sensitive than a traditional survey technique for detecting an aquatic invader. Ecol Appl 25:1944–1952
Spens J, Evans AR, Halfmaerten D et al (2016) Comparison of capture and storage methods for aqueous macrobial eDNA using an optimized extraction protocol: advantage of enclosed filter. Methods Ecol Evol 8:635–645. https://doi.org/10.1111/2041-210X.12683
Su Y-S, Yajima M (2015) R2jags: using R to Run ‘JAGS’. R package version 0.5-7. https://CRAN.R-project.org/package=R2jags
Tingley R, Ward-Fear G, Schwarzkopf L et al (2017) New Weapons in the Toad Toolkit: A review of methods to control and mitigate the biodiversity impacts of invasive cane toads (Rhinella marina). Q Rev Biol 92:123–149. https://doi.org/10.1086/692167
Xia Z, Zhan A, Gao Y et al (2018) Early detection of a highly invasive bivalve based on environmental DNA (eDNA). Biol Invasions 20:437–447. https://doi.org/10.1007/s10530-017-1545-7
Acknowledgements
RT and ARW were funded by the Australian Research Council Linkage Scheme (LP140100731). MG would like to thank Sutherland Shire Council and Sydney Metropolitan Local Land Services for funding, and Trent McKenna and George Madani for assistance in the field.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tingley, R., Greenlees, M., Oertel, S. et al. Environmental DNA sampling as a surveillance tool for cane toad Rhinella marina introductions on offshore islands. Biol Invasions 21, 1–6 (2019). https://doi.org/10.1007/s10530-018-1810-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-018-1810-4