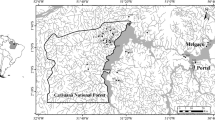
Abstract
Deforestation is a major driver of biodiversity loss in the Tropical region, but the role of upstream refugia and dispersal ability on the community response to this disturbance is unknown. We assessed the relevance of undisturbed upstream patches (“refugia”) on the responses of benthic communities to forest cover loss. We selected four Andean rivers with a well-protected forest in their upstream section and different degree of forest cover loss downstream and evaluated the dissimilarity patterns of three benthic communities (diatoms, Chironomidae, and the assemblage of Ephemeroptera, Plecoptera and Trichoptera—‘EPT’) along their water courses. We evaluated the dissimilarity by using Euclidean (environment), Sørensen (incidence data) and Morisita–Horn (abundance data) pairwise distances. We found that diatom beta-diversity, as organisms with passive but higher dispersal ability, significantly tracked the environmental changes caused by forest loss. However, insect communities, whose a priori are active dispersers and can track for suitable conditions, were weakly affected by deforestation. These results provide evidences that the existence of well-preserved upstream reaches along patched corridors may allow non-tolerant species to remain extant throughout dispersal-driven feedbacks. This being the case in the Andean streams, effects of deforestation on benthic communities were small, and not uniform. Our results reinforce the strategy of preserving upstream sections in order to achieve successful restoration or rehabilitation goals.





Similar content being viewed by others
References
Acosta R, Prat N (2010) Chironomid assemblages in high altitude streams of the Andean region of Peru. Fundam Appl Limnol 177:57–79. https://doi.org/10.1127/1863-9135/2010/0177-0057
Altermatt F, Seymour M, Martinez N (2013) River network properties shape-diversity and community similarity patterns of aquatic insect communities across major drainage basins. J Biogeogr 40:2249–2260. https://doi.org/10.1111/jbi.12178
Anderson MJ, Ellingsen KE, McArdle BH (2006) Multivariate dispersion as a measure of beta diversity. Ecol Lett 9:683–693. https://doi.org/10.1111/j.1461-0248.2006.00926.x
Armenteras D, Rodríguez N, Retana J, Morales M (2011) Understanding deforestation in montane and lowland forests of the Colombian Andes. Reg Environ Chang 11:693–705. https://doi.org/10.1007/s10113-010-0200-y
Barbour MT, Gerritsen J, Snyder BD, Stribling JB (1999) Rapid bioassessment protocols for use in streams and wadeable rivers: periphyton, benthic macroinvertebrates and fish. US Environmental Protection Agency, Office of Water Washington, DC
Barlow J, Lennox GD, Ferreira J et al (2016) Anthropogenic disturbance in tropical forests can double biodiversity loss from deforestation. Nature 535:144–147. https://doi.org/10.1038/nature18326
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 19:134–143. https://doi.org/10.1111/j.1466-8238.2009.00490.x
Baselga A, Orme CDL (2012) Betapart: an R package for the study of beta diversity. Methods Ecol Evol 3:808–812. https://doi.org/10.1111/j.2041-210X.2012.00224.x
Bellinger EG, Sigee DC (2015) Freshwater algae: identification and use as bioindicators. Wiley, New York
Bitušík P, Svitok M, Novikmec M et al (2017) Biological recovery of acidified alpine lakes may be delayed by the dispersal limitation of aquatic insect adults. Hydrobiologia 790:287–298
Bohonak AJ, Jenkins DG (2003) Ecological and evolutionary significance of dispersal by freshwater invertebrates. Ecol Lett 6:783–796
Bojsen BH, Jacobsen D (2003) Effects of deforestation on macroinvertebrate diversity and assemblage structure in Ecuadorian Amazon streams. Arch für Hydrobiol 158:317–342. https://doi.org/10.1127/0003-9136/2003/0158-0317
Booth GD, Niccolucci MJ, Schuster EG (1994) Identifying proxy sets in multiple linear regression: an aid to better coefficient interpretation
Buss DF, Vitorino AS (2010) Rapid bioassessment protocols using benthic macroinvertebrates in Brazil: evaluation of taxonomic sufficiency. J North Am Benthol Soc 29:562–571. https://doi.org/10.1899/09-095.1
Buss DF, Carlisle DM, Chon T-S et al (2015) Stream biomonitoring using macroinvertebrates around the globe: a comparison of large-scale programs. Environ Monit Assess 187:4132
Buytaert W, Célleri R, De Bièvre B et al (2006) Human impact on the hydrology of the Andean páramos. Earth Sci Rev 79:53–72. https://doi.org/10.1016/j.earscirev.2006.06.002
Campbell Grant EH, Lowe WH, Fagan WF (2007) Living in the branches: population dynamics and ecological processes in dendritic networks. Ecol Lett 10:165–175. https://doi.org/10.1111/j.1461-0248.2006.01007.x
Chessel D, Dufour AB, Thioulouse J (2004) The ade4 package-I-one-table methods. R news. 4:5–10
De Bie T, De Meester L, Brendonck L et al (2012) Body size and dispersal mode as key traits determining metacommunity structure of aquatic organisms. Ecol Lett 15:740–747. https://doi.org/10.1111/j.1461-0248.2012.01794.x
Domínguez E, Fernández HR (2009) Macroinvertebrados bentónicos sudamericanos: sistemática y biología. Fundación Miguel Lillo Tucumán
Downes BJ, Lancaster J, Glaister A, Bovill WD (2016) A fresh approach reveals how dispersal shapes metacommunity structure in a human-altered landscape. J Appl Ecol. https://doi.org/10.1111/1365-2664.12759
Elosegi A, Sabater S (2009) Conceptos y técnicas en ecología fluvial. Fundación BBVA, Bilbao
Encalada AC, Peckarsky BL (2006) Selective oviposition of the mayfly Baetis bicaudatus. Oecologia 148:526–537. https://doi.org/10.1007/s00442-006-0376-5
Etter A, Mcalpine C, Possingham H (2008) Historical patterns and drivers of landscape change in Colombia Since 1500: a regionalized spatial approach annals of the association of American geographers. Ann Assoc Am Geogr 98:2–23. https://doi.org/10.1080/00045600701733911
Gaston KJ, Blackburn TM, Greenwood JJD et al (2000) Abundance–occupancy relationships. J Appl Ecol 37:39–59
González-Trujillo JD, Donato-Rondon JC (2016) Changes in invertebrate assemblage structure as affected by the flow regulation of a páramo river. Ann Limnol 52:307–316. https://doi.org/10.1051/limn/2016018
Graham SE, Storey R, Smith B (2017) Dispersal distances of aquatic insects: upstream crawling by benthic EPT larvae and flight of adult Trichoptera along valley floors. New Zeal J Mar Freshw Res 51:146–164. https://doi.org/10.1080/00288330.2016.1268175
Gresens SE, Belt KT, Tang JA et al (2007) Temporal and spatial responses of Chironomidae (Diptera) and other benthic invertebrates to urban stormwater runoff. Hydrobiologia 575:173–190
Gutiérrez-Cánovas C, Millán A, Velasco J et al (2013) Contrasting effects of natural and anthropogenic stressors on beta diversity in river organisms. Glob Ecol Biogeogr 22:796–805. https://doi.org/10.1111/geb.12060
Hawkes HA (1998) Origin and development of the biological monitoring working party score system. Water Res 32:964–968. https://doi.org/10.1016/S0043-1354(97)00275-3
Heino J, Melo AS, Bini LM (2015) Reconceptualising the beta diversity-environmental heterogeneity relationship in running water systems. Freshw Biol 60:223–235. https://doi.org/10.1111/fwb.12502
Hitt NP, Angermeier PL (2008) Evidence for fish dispersal from spatial analysis of stream network topology. J North Am Benthol Soc 27:304–320. https://doi.org/10.1899/07-096.1
Hoffmann A, Resh VH (2003) Oviposition in three species of limnephiloid caddisflies (Trichoptera): hierarchical influences on site selection. Freshw Biol 48:1064–1077. https://doi.org/10.1046/j.1365-2427.2003.01074.x
Horn HS (1966) Measurement of” overlap” in comparative ecological studies. Am Nat 100:419–424
Iñiguez-Armijos C, Leiva A, Frede HG et al (2014) Deforestation and benthic indicators: how much vegetation cover is needed to sustain healthy Andean streams? PLoS ONE. https://doi.org/10.1371/journal.pone.0105869
Jost L, Chao A, Chazdon RL (2011) Compositional similarity and beta diversity. Biological diversity: frontiers in measurement and assessment. Oxford University Press, Oxford, pp 66–84
Kärnä OM, Grönroos M, Antikainen H et al (2015) Inferring the effects of potential dispersal routes on the metacommunity structure of stream insects: as the crow flies, as the fish swims or as the fox runs? J Anim Ecol 84:1342–1353. https://doi.org/10.1111/1365-2656.12397
Kitto JAJ, Gray DP, Greig HS et al (2015) Meta-community theory and stream restoration: evidence that spatial position constrains stream invertebrate communities in a mine impacted landscape. Restor Ecol 23:284–291. https://doi.org/10.1111/rec.12179
Krammer K, Lange-Bertalot H (1986) Bacillariophyceae: Naviculaceae. In: Ettl H, Gerloff J, Heynig H, Mollenhauer D (eds.) Süsswasserflora von Mitteleuropa. Bd. 2, Teil 1. Gustav Fischer, Stuttgart
Krammer K, Lange-Bertalot H (1988) Bacillariophyceae: Bacillariaceae, Epithemiaceae, Surirellaceae. In: Ettl H, Gerloff J, Heynig H, Mollenhauer D (eds.) Süsswasserflora von Mitteleuropa. Bd. 2, Teil 2. Gustav Fischer, Stuttgart
Krammer K, Lange-Bertalot H (1991a) Bacillariophyceae: Achnanthaceae, Kritische Ergänzungen zu Navicula (Lineolatae) und Gomphonema Gesamtliterarurverzeichnis. In: Ettl H, Gerloff J, Heynig H, Mollenhauer D (eds.) Süsswasserflora von Mitteleuropa. Bd. 2, Teil 4. Gustav Fischer, Stuttgart
Krammer K, Lange-Bertalot H (1991b) Bacillariophyceae: Centrales, Fragilariaceae, Eunotiaceae. In: Ettl H, Gerloff J, Heynig H, Mollenhauer D (eds.) Süsswasserflora von Mitteleuropa. Bd. 2, Teil 3. Gustav Fischer, Stuttgart
Lancaster J, Downes BJ (2017) Dispersal traits may reflect dispersal distances, but dispersers may not connect populations demographically. Oecologia 184:171–182. https://doi.org/10.1007/s00442-017-3856-x
Leibold MA, Chase JM (2018) Metacommunity ecology, 1st edn. Princeton University Press, Princeton
Leibold MA, Holyoak M, Mouquet N et al (2004) The metacommunity concept: a framework for multi-scale community ecology. Ecol Lett 7:601–613
Leira M, Sabater S (2005) Diatom assemblages distribution in catalan rivers, NE Spain, in relation to chemical and physiographical factors. Water Res 39:73–82
Lenat DR (1993) A biotic index for the southeastern United States: derivation and list of tolerance values, with criteria for assigning water-quality ratings. J North Am Benthol Soc 12:279–290
Lencioni V, Marziali L, Rossaro B (2012) Chironomids as bioindicators of environmental quality in mountain springs. Freshw Sci 31:525–541. https://doi.org/10.1899/11-038.1
Liu J, Soininen J, Han BP, Declerck SAJ (2013) Effects of connectivity, dispersal directionality and functional traits on the metacommunity structure of river benthic diatoms. J Biogeogr 40:2238–2248. https://doi.org/10.1111/jbi.12160
Lorion CM, Kennedy BP (2009) Relationships between deforestation, riparian forest buffers and benthic macroinvertebrates in neotropical headwater streams. Freshw Biol 54:165–180. https://doi.org/10.1111/j.1365-2427.2008.02092.x
Macneale KH, Peckarsky BL, Likens GE (2005) Stable isotopes identify dispersal patterns of stonefly populations living along stream corridors. Freshw Biol 50:1117–1130. https://doi.org/10.1111/j.1365-2427.2005.01387.x
Malmqvist B (2000) How does wing length relate to distribution patterns of stoneflies (Plecoptera) and mayflies (Ephemeroptera)? Biol Conserv 93:271–276. https://doi.org/10.1016/S0006-3207(99)00139-1
Merritt RW, Cummins KW (2008) An introduction to the aquatic insects of North America. Kendall/Hunt, Dubuque
Metzeltin D, Lange-Bertalot H (2007) Tropical diatoms of South America II. Special remarks on biogeography disjunction. Iconogr Diatomol 18:1–877
Milošević D, Simić V, Stojković M et al (2013) Spatio-temporal pattern of the Chironomidae community: toward the use of non-biting midges in bioassessment programs. Aquat Ecol 47:37–55
Múrria C, Rugenski AT, Whiles MR, Vogler AP (2015) Long-term isolation and endemicity of Neotropical aquatic insects limit the community responses to recent amphibian decline. Divers Distrib 21:938–949. https://doi.org/10.1111/ddi.12343
Myers N, Fonseca GAB, Mittermeier R et al (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Oksanen J, Blanchet FG, Kindt R et al (2013) Package ‘vegan.’ Community Ecol Packag version 2
Padial AA, Ceschin F, Declerck SAJ et al (2014) Dispersal ability determines the role of environmental, spatial and temporal drivers of metacommunity structure. PLoS ONE 9:1–8. https://doi.org/10.1371/journal.pone.0111227
Palmer MA, Ambrose RF, Poff NL (1997) Ecological theory and community restoration ecology. Restor Ecol 5:291–300. https://doi.org/10.1046/j.1526-100X.1997.00543.x
Passy SI (2008) Continental diatom biodiversity in stream benthos declines as more nutrients become limiting. Proc Natl Acad Sci USA 105:9663–9667. https://doi.org/10.1073/pnas.0802542105
Poff NL, Olden JD, Vieira NKM et al (2006) Functional trait niches of North American lotic insects: traits-based ecological applications in light of phylogenetic relationships. J North Am Benthol Soc 25:730–755. https://doi.org/10.1899/0887-3593(2006)025%5b0730:FTNONA%5d2.0.CO;2
Prat N, Rieradevall M, Acosta R, Villamarín C (2011) Guía para el reconocimiento de las larvas de Chironomidae (díptera) de los ríos altoandinos de Ecuador y Perú. Grup Investig FEM Dep Ecol Univ Barcelona, España, p 78
Prat N, González-Trujillo JD, Ospina-Torres R (2014) Key to chironomid pupal exuviae (Diptera: Chironomidae) of tropical high Andean streams. Rev Biol Trop 62:1385–1406
Qian H, Ricklefs RE, White PS (2004) Beta diversity of angiosperms in temperate floras of eastern Asia and eastern North America. Ecol Lett 8:15–22. https://doi.org/10.1111/j.1461-0248.2004.00682.x
R Core Team (2018) R: A language and environment for statistical computing
Richardson JS, Béraud S (2014) Effects of riparian forest harvest on streams: a meta-analysis. J Appl Ecol 51:1712–1721. https://doi.org/10.1111/1365-2664.12332
Rios-Touma B, Encalada AC, Prat N (2012) Oviposition of aquatic insects in a tropical high altitude stream. Environ Entomol 41:1322–1331. https://doi.org/10.1603/EN12037
Ríos-Touma B, Prat N, Encalada AC (2011) Invertebrate drift and colonization processes in a tropical andean stream. Aquat Biol 14:233–246. https://doi.org/10.3354/ab00399
Rodríguez Eraso N, Armenteras Pascual D, Alumbrerosa R (2012) Land use and land cover change in the Colombian Andes: dynamics and future scenarios. J Land Use Sci 8:154–174
Sarremejane R, Mykrä H, Bonada N et al (2017) Habitat connectivity and dispersal ability drive the assembly mechanisms of macroinvertebrate communities in river networks. Freshw Biol 62:1073–1082. https://doi.org/10.1111/fwb.12926
Sedell JR, Reeves GH, Hauer FR et al (1990) Role of refugia in recovery from disturbances: modern fragmented and disconnected river systems. Environ Manag 14:711–724. https://doi.org/10.1007/BF02394720
Serra SRQ, Graça MAS, Dolédec S et al (2017) Chironomidae traits and life history strategies as indicators of anthropogenic disturbance. Environ Monit Assess 189:326. https://doi.org/10.1007/s10661-017-6027-y
Siqueira T, Durães L, de Roque FO (2014) Predictive modelling of insect metacommunities in biomonitoring of aquatic networks. Ecol Model Appl Entomol. Springer, Cham, pp 109–126
Socolar JB, Gilroy JJ, Kunin WE, Edwards DP (2016) How should beta-diversity inform biodiversity conservation? Trends Ecol Evol 31:67–80. https://doi.org/10.1016/j.tree.2015.11.005
Soininen J (2007) Environmental and spatial control of freshwater diatoms: a review. Diatom Res 22:473–490. https://doi.org/10.1080/0269249X.2007.9705724
Sørensen T (1948) A method of establishing groups of equal amplitude in plant sociology based on similarity of species and its application to analyses of the vegetation on Danish commons. Biol Skr 5:1–34
Thompson R, Townsend C (2006) A truce with neutral theory: local deterministic factors, species traits and dispersal limitation together determine patterns of diversity in stream invertebrates. J Anim Ecol 75:476–484. https://doi.org/10.1111/j.1365-2656.2006.01068.x
Tilman D, Kilham SS, Kilham P (1982) Phytoplankton community ecology: the role of limiting nutrients. Annu Rev Ecol Syst 13:349–372. https://doi.org/10.1146/annurev.es.13.110182.002025
Tonkin JD, Stoll S, Sundermann A, Haase P (2014) Dispersal distance and the pool of taxa, but not barriers, determine the colonisation of restored river reaches by benthic invertebrates. Freshw Biol 59:1843–1855. https://doi.org/10.1111/fwb.12387
Tonkin JD, Altermatt F, Finn DS et al (2018) The role of dispersal in river network metacommunities: patterns, processes, and pathways. Freshw Biol 63:141–163. https://doi.org/10.1111/fwb.13037
Tornés E, Ruhí A (2013) Flow intermittency decreases nestedness and specialisation of diatom communities in Mediterranean rivers. Freshw Biol 58:2555–2566. https://doi.org/10.1111/fwb.12232
Tornés E, Mor J-R, Mandaric L, Sabater S (2018) Diatom responses to sewage inputs and hydrological alteration in Mediterranean streams. Environ Pollut 238:369–378. https://doi.org/10.1016/j.envpol.2018.03.037
Trivinho-Strixino S, Strixino G (1995) Larvas de Chironomidae (Diptera) do Estado de São Paulo: guia de identificação e diagnose dos gêneros
Wickham H (2010) ggplot2: elegant graphics for data analysis. J Stat Softw 35:65–88
Williams CJ (1989) Downstream drift of the larvae of Chironomidae (Diptera) in the River Chew, SW England. Hydrobiologia 183:59–72
Winking C, Lorenz AW, Sures B, Hering D (2014) Recolonisation patterns of benthic invertebrates: a field investigation of restored former sewage channels. Freshw Biol 59:1932–1944. https://doi.org/10.1111/fwb.12397
Zuur AF, Ieno EN, Walker NJ et al (2009) In: Gail R, Krickeberg K, Samet JM, Tsiatis A, Wong W (eds) Mixed effects models and extensions in ecology. Springer, New York
Acknowledgements
We would like to thank to the two anonymous reviewers who help to improve substantially our manuscript. We are grateful to Liz Alonso, Katherine Pallares, Laura Fernández and Daniel Amézquita for their help in the field and the laboratory. Special thanks to ‘La Chorrera’ Natural Park for providing access to ‘Chiflón’ stream, and to Miguel Rodriguez and his family for hosting us during our field work. The surveys were financed by the Universidad Nacional de Colombia—Sede Bogotá, through DIB project no. 34856 (CONVOCATORIA NACIONAL DE PROYECTOS PARA EL FORTALECIMIENTO DE LA INVESTIGACIÓN, CREACIÓN E INNOVACIÓN DE LA UNIVERSIDAD NACIONAL DE COLOMBIA 2016–2018).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by David Hawksworth.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
González-Trujillo, J., Petsch, D., Córdoba-Ariza, G. et al. Upstream refugia and dispersal ability may override benthic-community responses to high-Andean streams deforestation. Biodivers Conserv 28, 1513–1531 (2019). https://doi.org/10.1007/s10531-019-01739-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-019-01739-2