Abstract
Recent studies have shown a high level of conservation between Drosophila melanogaster and mammalian copper homeostasis mechanisms. These studies have also demonstrated the efficiency with which this species can be used to characterize novel genes, at both the cellular and whole organism level. As a versatile and inexpensive model organism, Drosophila is also particularly useful for gene discovery applications and thus has the potential to be extremely useful in identifying novel copper homeostasis genes and putative disease genes. In order to assess the suitability of Drosophila for this purpose, three screening approaches have been investigated. These include an analysis of the global transcriptional response to copper in both adult flies and an embryonic cell line using DNA microarray analysis. Two mutagenesis-based screens were also utilized. Several candidate copper homeostasis genes have been identified through this work. In addition, the results of each screen were carefully analyzed to identify any factors influencing efficiency and sensitivity. These are discussed here with the aim of maximizing the efficiency of future screens and the most suitable approaches are outlined. Building on this information, there is great potential for the further use of Drosophila for copper homeostasis gene discovery.







Similar content being viewed by others
References
Armendariz AD, Gonzalez M, Loguinov AV, Vulpe CD (2004) Gene expression profiling in chronic copper overload reveals upregulation of Prnp and App. Physiol Genomics 20:45–54
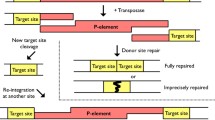
Bentley A, MacLennan B, Calvo J, Dearolf CR (2000) Targeted recovery of mutations in Drosophila. Genetics 156:1169–1173
Camakaris J, Danks DM, Ackland L et al (1980) Altered copper metabolism in cultured cells from human Menkes’ syndrome and mottled mouse mutants. Biochem Genet 18:117–131
Chow KL, Hall DH, Emmons SW (1995) The mab-21 gene of Caenorhabditis elegans encodes a novel protein required for choice of alternate cell fates. Development 121:3615–3626
D’Andrea LD, Regan L (2003) TPR proteins: the versatile helix. Trends Biochem Sci 28:655
Dascher C, Matteson J, Balch WE (1994) Syntaxin 5 regulates endoplasmic reticulum to Golgi transport. J Biol Chem 269:29363–29366
Dimova DK, Stevaux O, Frolov MV, Dyson NJ (2003) Cell cycle-dependent and cell cycle-independent control of transcription by the Drosophila E2F/RB pathway. Genes Dev 17:2308–2320
Durliat M, Bonneton F, Boissonneau E, Andre M, Wegnez M (1995) Expression of metallothionein genes during the post-embryonic development of Drosophila melanogaster. Biometals 8:339–351
Egli D, Selvaraj A, Yepiskoposyan H et al (2003) Knockout of ‘metal-responsive transcription factor’ MTF-1 in Drosophila by homologous recombination reveals its central role in heavy metal homeostasis. EMBO J 22:100–108
Ffrench-Constant RH, Roush RT (1991) Gene mapping and cross-resistance in cyclodiene insecticide-resistant Drosophila melanogaster. Proc Natl Acad Sci USA 57:17
Freedman J, Ciriolo M, Peisach J (1989) The role of glutathione in copper metabolism and toxicity. J Biol Chem 264:5598–5605
Goldberg MA, Dunning SP, Bunn HF (1988) Regulation of the erythropoietin gene: evidence that the oxygen sensor is a heme protein. Science 242:1412–1415
Gunes C, Heuchel R, Georgiev O et al (1998) Embryonic lethality and liver degeneration in mice lacking the metal-responsive transcriptional activator MTF-1. EMBO J 17:2846–2854
Hamza I, Faisst A, Prohaska J et al (2001) The metallochaperone Atox1 plays a critical role in perinatal copper homeostasis. Proc Natl Acad Sci USA 98:6848–6852
Harris ZL, Klomp LW, Gitlin JD (1998) Aceruloplasminemia: an inherited neurodegenerative disease with impairment of iron homeostasis. Am J Clin Nutr 67:972S–977S
Jahn R, Lang T, Sudhof TC (2003) Membrane fusion. Cell 112:519–533
Jin K, Mao XO, Eshoo MW et al (2002) cDNA microarray analysis of changes in gene expression induced by neuronal hypoxia in vitro. Neurochem Res 27:1105–1112
Kanz C, Aldebert P, Althorpe N et al (2005) The EMBL nucleotide sequence database. Nucleic Acids Res 33:D29–D33
Kuo YM, Zhou B, Cosco D, Gitschier J (2001) The copper transporter CTR1 provides an essential function in mammalian embryonic development. Proc Natl Acad Sci USA 98:6836–6841
Levenson CW (1998) Mechanisms of copper conservation in organs. Am J Clin Nutr 67:978S–981S
Li J, Ji C, Chen J et al (2005) Identification and characterization of a novel Cut family cDNA that encodes human copper transporter protein CutC. Biochem Biophys Res Commun 337:179–183
Linder MC (1991) Nutrition and metabolism of trace elements. nutritional biochemistry and metabolism. MC Linder. Elsevier, New York, pp 215–276
Margolis RL, Stine OC, McInnis MG et al (1996) cDNA cloning of a human homologue of the Caenorhabditis elegans cell fate-determining gene mab-21: expression, chromosomal localization and analysis of a highly polymorphic (CAG)n trinucleotide repeat. Hum Mol Genet 5:607–616
Maroni G, Wise J, Young JE, Otto E (1987) Metallothionein gene duplications and metal tolerance in natural populations of Drosophila melanogaster. Genetics 117:739–744
Martin F, Linden T, Katschinski DM et al (2005) Copper-dependent activation of hypoxia-inducible factor (HIF)-1: implications for ceruloplasmin regulation. Blood 105:4613–4619
McCarroll SA, Murphy CT, Zou S et al (2004) Comparing genomic expression patterns across species identifies shared transcriptional profile in aging. Nat Genet 36:197–204
Norgate M, Lee E, Southon A et al (2006) Essential roles in development and pigmentation for the Drosophila copper transporter DmATP7. Mol Biol Cell 17:475–484
Otto E, Young JE, Maroni G (1986) Structure and expression of a tandem duplication of the Drosophila metallothionein gene. Proc Natl Acad Sci USA 83:6025–6029
Petris MJ, Mercer JF, Culvenor JG et al (1996) Ligand-regulated transport of the Menkes copper P-type ATPase efflux pump from the Golgi apparatus to the plasma membrane: a novel mechanism of regulated trafficking. EMBO J 15:6084–6095
Rae TD, Schmidt PJ, Pufahl RA, Culotta VC, O’Halloran TV (1999) Undetectable intracellular free copper: the requirement of a copper chaperone for superoxide dismutase. Science 284:805–808
Rozen S, Skaletsky H (2000) Primer3 on the WWW for general users and for biologist programmers. Methods Mol Biol 132:365–386
Sega GA, Lee WR (1970) Mutagenising Drosophila by vacuum infusion. Drosophila Inf Serv 45:179
Silar P, Theodore L, Mokdad R et al (1990) Metallothionein Mto gene of Drosophila melanogaster: structure and regulation. J Mol Biol 215:217–224
Smyth KA, Parker AG, Yen JL, McKenzie JA (1992) Selection of dieldrin-resistant strains of Lucilia cuprina (Diptera: Calliphoridae) after ethyl methanesulfonate mutagenesis of a susceptible strain. J Econ Entomol 85:352–358
Southon A, Burke R, Norgate M, Batterham P, Camakaris J (2004) Copper homoeostasis in Drosophila melanogaster S2 cells. Biochem J 383:303–309
Stathopoulos A, Van Drenth M, Erives A, Markstein M, Levine M (2002) Whole-genome analysis of dorsal-ventral patterning in the Drosophila embryo. Cell 111:687–701
Suga K, Hattori H, Saito A, Akagawa K (2005) RNA interference-mediated silencing of the syntaxin 5 gene induces Golgi fragmentation but capable of transporting vesicles. FEBS Lett 579:4226–4234
Tai G, Lu L, Wang TL et al (2004) Participation of the syntaxin 5/Ykt6/GS28/GS15 SNARE complex in transport from the early/recycling endosome to the trans-Golgi network. Mol Biol Cell 15:4011–4022
Turner JH, Gardner EJ (1960) The effect of copper and iron salts and tryptophan on head abnormalities and melanotic tumors in different stocks of Drosophila melanogaster. Genetics 45:915–924
Ungar D, Oka T, Brittle EE et al (2002) Characterization of a mammalian Golgi-localized protein complex, COG, that is required for normal Golgi morphology and function. J Cell Biol 157:405–415
Vargas EJ, Shoho AR, Linder MC (1994) Copper transport in the Nagase analbuminemic rat. Am J Physiol 267:G259–G269
Wallace B (1982) Drosophila melanogaster populations selected for resistance to NaCl and CuSo4 in both allopatry and sympatry. J Hered 73:35–42
Xu H, Brill JA, Hsien J et al (2002) Syntaxin 5 Is Required for Cytokinesis and Spermatid Differentiation in Drosophila. Dev Biol 251:294
Zer H, Freedman J, Peisach J, Chevion M (1991) Inverse correlation between resistance towards copper and towards the redox-cycling compound paraquat: a study in copper-tolerant hepatocytes in tissue culture. Free Radic Biol Med 11:9–16
Zhou H, Cadigan KM, Thiele DJ (2003) A Copper-regulated transporter required for copper acquisition, pigmentation, and specific stages of development in Drosophila melanogaster. J Biol Chem 278:48210–48218
Acknowledgments
The authors would like to thank Donald Ingram for generous support with paraquat microarrays and Ben Ong for support with copper microarrays. We are also grateful to John Roote for supplying the deficiency mutants used in mapping the Syx5 region and to John Damiano, Louise Williams, Phoebe Heard, and Sally Coutts who assisted with the various screens. We thank Dr. Richard Burke for helpful discussions. Part of this work was supported by the Intramural Research Program of the NIH, National Institute on Aging (SZ) and grants from the International Copper Association (JC and PB), the Australian Research Council (JC), and the Australian Institute of Nuclear Science and Engineering (JC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Norgate, M., Southon, A., Zou, S. et al. Copper homeostasis gene discovery in Drosophila melanogaster . Biometals 20, 683–697 (2007). https://doi.org/10.1007/s10534-006-9075-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-006-9075-2