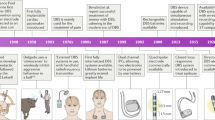
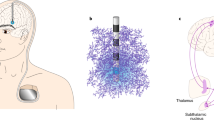
Abstract
High frequency electrical stimulation of deep brain structures such as the subthalamic nucleus in Parkinson’s disease or thalamus for essential tremor is used clinically to reduce symptom severity. Deep brain stimulation activates neurons in specific brain structures and connection pathways, overriding aberrant neural activity associated with symptoms. While optimal deep brain stimulation might activate a particular neural structure precisely, existing deep brain stimulation can only generate roughly-spherical regions of activation that do not overlap with any target anatomy. Additionally, side effects linked to stimulation may be the result of limited control over placement of stimulation and its subsequent spread out of optimal target boundaries. We propose a novel lead with thousands of individually controllable contacts capable of asymmetric stimulation profiles. Here we outline the design motivation, manufacturing process, and initial testing of this new electrode design, placing it on track for further directional stimulation studies.







Similar content being viewed by others
References
L.M. Alba-Ferrara, F. Fernandez, G.A. de Erausquin, The use of neuromodulation in the treatment of cocaine dependence. Addict. Disord. Treat. 13, 1–7 (2014). doi:10.1097/ADT.0b013e31827b5a2c
J.L. Alberts, C. Voelcker-Rehage, K. Hallahan, M. Vitek, R. Bamzai, J.L. Vitek, Bilateral subthalamic stimulation impairs cognitive-motor performance in Parkinson’s disease patients. Brain J. Neurol. 131, 3348–3360 (2008). doi:10.1093/brain/awn238
B.S. Appleby, P.S. Duggan, A. Regenberg, P.V. Rabins, Psychiatric and neuropsychiatric adverse events associated with deep brain stimulation: a meta-analysis of ten years’ experience. Mov. Disord. Off. J. Mov. Disord. Soc. 22, 1722–1728 (2007). doi:10.1002/mds.21551
M. Astrom, E. Diczfalusy, H. Martens, K. Wardell, Relationship between neural activation and electric field distribution during deep brain stimulation. IEEE Trans. Biomed. Eng. 1–1 (2014). doi:10.1109/TBME.2014.2363494
S. Ball, T.L. Gilbert, C.C. Overly, The human brain online: an open resource for advancing brain research. PLoS Biol. 10, (2012). doi:10.1371/journal.pbio.1001453
A.L. Benabid, P. Pollak, C. Gervason, D. Hoffmann, D.M. Gao, M. Hommel, J.E. Perret, J. de Rougemont, Long-term suppression of tremor by chronic stimulation of the ventral intermediate thalamic nucleus. Lancet 337, 403–406 (1991)
C.R. Butson, C.C. McIntyre, Current steering to control the volume of tissue activated during deep brain stimulation. Brain Stimulat. 1, 7–15 (2008). doi:10.1016/j.brs.2007.08.004
A. Chaturvedi, C.R. Butson, S.F. Lempka, S.E. Cooper, C.C. McIntyre, Patient-specific models of deep brain stimulation: Influence of field model complexity on neural activation predictions. Brain Stimulat. 3, 65–67 (2010). doi:10.1016/j.brs.2010.01.003
A. Chopra, B.T. Klassen, M. Stead, Current clinical application of deep-brain stimulation for essential tremor. Neuropsychiatr. Dis. Treat. 9, 1859–1865 (2013). doi:10.2147/NDT.S32342
M.F. Contarino, L.J. Bour, R. Verhagen, M.A.J. Lourens, R.M.A. de Bie, P. van den Munckhof, P.R. Schuurman, Directional steering A novel approach to deep brain stimulation. Neurology 83, 1163–1169 (2014). doi:10.1212/WNL.0000000000000823
Deep-Brain Stimulation for Parkinson’s Disease Study Group, Deep-brain stimulation of the subthalamic nucleus or the pars interna of the globus pallidus in Parkinson’s disease. N. Engl. J. Med. 345, 956–963 (2001). doi:10.1056/NEJMoa000827
P. Doshi, P. Bhargava, Hypersexuality following subthalamic nucleus stimulation for Parkinson’s disease. Neurol. India 56, 474–476 (2008)
T.-M. Ellis, K.D. Foote, H.H. Fernandez, A. Sudhyadhom, R.L. Rodriguez, P. Zeilman, C.E. Jacobson 4th, M.S. Okun, Reoperation for suboptimal outcomes after deep brain stimulation surgery. Neurosurgery 63, 754–760 (2008). doi:10.1227/01.NEU.0000325492.58799.35. discussion 760–761
M.M. Elwassif, A. Datta, A. Rahman, M. Bikson, Temperature control at DBS electrodes using a heat sink: experimentally validated FEM model of DBS lead architecture. J. Neural Eng. 9, 046009 (2012). doi:10.1088/1741-2560/9/4/046009
A.A. Fomani, M. Moradi, S. Assaf, R.R. Mansour, 3D microprobes for deep brain stimulation and recording, in: 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). Presented at the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) (2010), pp. 1808–1811. doi:10.1109/IEMBS.2010.5626410
D. Fontaine, C. Vandersteen, C. Saleh, D. von Langsdorff, G. Poissonnet, Two-step tunneling technique of deep brain stimulation extension wires-a description. Acta Neurochir. (Wien) 155, 2399–2402 (2013). doi:10.1007/s00701-013-1870-2
L. Golestanirad, B. Elahi, A. Molina, J.R. Mosig, C. Pollo, R. Chen, S.J. Graham, Analysis of fractal electrodes for efficient neural stimulation. Front. Neuroengineering 6, 3 (2013). doi:10.3389/fneng.2013.00003
B.D. Greenberg, L.A. Gabriels, D.A. Malone, A.R. Rezai, G.M. Friehs, M.S. Okun, N.A. Shapira, K.D. Foote, P.R. Cosyns, C.S. Kubu, P.F. Malloy, S.P. Salloway, J.E. Giftakis, M.T. Rise, A.G. Machado, K.B. Baker, P.H. Stypulkowski, W.K. Goodman, S.A. Rasmussen, B.J. Nuttin, Deep brain stimulation of the ventral internal capsule/ventral striatum for obsessive-compulsive disorder: worldwide experience. Mol. Psychiatry 15, 64–79 (2010). doi:10.1038/mp.2008.55
K. Hardenacke, E. Shubina, C.P. Bührle, A. Zapf, D. Lenartz, J. Klosterkötter, V. Visser-Vandewalle, J. Kuhn, Deep brain stimulation as a tool for improving cognitive functioning in Alzheimer’s dementia: a systematic review. Front. Psychiatry 4, 159 (2013). doi:10.3389/fpsyt.2013.00159
D. Huys, C. Bartsch, P. Koester, D. Lenartz, M. Maarouf, J. Daumann, J.K. Mai, J. Klosterkötter, S. Hunsche, V. Visser-Vandewalle, C. Woopen, L. Timmermann, V. Sturm, J. Kuhn, Motor improvement and emotional stabilization in patients with Tourette syndrome after deep brain stimulation of the ventral anterior and ventrolateral motor part of the Thalamus. Biol. Psychiatry (2014). doi:10.1016/j.biopsych.2014.05.014
C. Janson, R. Maxwell, A.A. Gupte, A. Abosch, Bowstringing as a complication of deep brain stimulation: case report. Neurosurgery 66, E1205 (2010). doi:10.1227/01.NEU.0000369199.72783.F9. discussion E1205
M. Keane, S. Deyo, A. Abosch, J.A. Bajwa, M.D. Johnson, Improved spatial targeting with directionally segmented deep brain stimulation leads for treating essential tremor. J. Neural Eng. 9, 046005 (2012). doi:10.1088/1741-2560/9/4/046005
O.P. Keifer, J.P. Riley, N.M. Boulis, Deep brain stimulation for chronic pain: intracranial targets, clinical outcomes, and trial design considerations. Neurosurg. Clin. N. Am. 25, 671–692 (2014). doi:10.1016/j.nec.2014.07.009
L. Kirsch-Darrow, L.B. Zahodne, M. Marsiske, M.S. Okun, K.D. Foote, D. Bowers, The trajectory of apathy after deep brain stimulation: from pre-surgery to 6 months post-surgery in Parkinson’s disease. Parkinsonism Relat. Disord. 17, 182–188 (2011). doi:10.1016/j.parkreldis.2010.12.011
B.M. Kluger, K.D. Foote, C.E. Jacobson, M.S. Okun, Lessons learned from a large single center cohort of patients referred for DBS management. Parkinsonism Relat. Disord. 17, 236–239 (2011). doi:10.1016/j.parkreldis.2010.05.003
R.J. Koek, J.-P. Langevin, S.E. Krahl, H.J. Kosoyan, H.N. Schwartz, J.W. Chen, R. Melrose, M.J. Mandelkern, D. Sultzer, Deep brain stimulation of the basolateral amygdala for treatment-refractory combat post-traumatic stress disorder (PTSD): study protocol for a pilot randomized controlled trial with blinded, staggered onset of stimulation. Trials 15, 356 (2014). doi:10.1186/1745-6215-15-356
D. Kondziolka, D. Whiting, A. Germanwala, M. Oh, Hardware-related complications after placement of thalamic deep brain stimulator systems. Stereotact. Funct. Neurosurg. 79, 228–233 (2002)
A.M. Kuncel, S.E. Cooper, B.R. Wolgamuth, M.A. Clyde, S.A. Snyder, E.B. Montgomery, A.R. Rezai, W.M. Grill, Clinical response to varying the stimulus parameters in deep brain stimulation for essential tremor. Mov. Disord. Off. J. Mov. Disord. Soc. 21, 1920–1928 (2006). doi:10.1002/mds.21087
S.-Y. Lim, S.S. O’Sullivan, K. Kotschet, D.A. Gallagher, C. Lacey, A.D. Lawrence, A.J. Lees, D.J. O’Sullivan, R.F. Peppard, J.P. Rodrigues, A. Schrag, P. Silberstein, S. Tisch, A.H. Evans, Dopamine dysregulation syndrome, impulse control disorders and punding after deep brain stimulation surgery for Parkinson’s disease. J. Clin. Neurosci. Off. J. Neurosurg. Soc. Australas. 16, 1148–1152 (2009). doi:10.1016/j.jocn.2008.12.010
K.E. Lyons, S.B. Wilkinson, J. Overman, R. Pahwa, Surgical and hardware complications of subthalamic stimulation A series of 160 procedures. Neurology 63, 612–616 (2004). doi:10.1212/01.WNL.0000134650.91974.1A
A.G. Machado, G.K. Hiremath, F. Salazar, A.R. Rezai, Fracture of subthalamic nucleus deep brain stimulation hardware as a result of compulsive manipulation: case report. Neurosurgery 2005, 57 (2005). doi:10.1227/01.NEU.0000187566.01731.51
C.B. Maks, C.R. Butson, B.L. Walter, J.L. Vitek, C.C. McIntyre, Deep brain stimulation activation volumes and their association with neurophysiological mapping and therapeutic outcomes. J. Neurol. Neurosurg. Psychiatry 80, 659–666 (2009). doi:10.1136/jnnp.2007.126219
H.C.F. Martens, E. Toader, M.M.J. Decré, D.J. Anderson, R. Vetter, D.R. Kipke, K.B. Baker, M.D. Johnson, J.L. Vitek, Spatial steering of deep brain stimulation volumes using a novel lead design. Clin. Neurophysiol. 122, 558–566 (2011). doi:10.1016/j.clinph.2010.07.026
G. Messina, M. Rizzi, I. Dones, A. Franzini, Cosmetic posterior implant of internal pulse generators in deep brain stimulation procedures: technical report. Neuromodulation J. Int. Neuromodulation Soc. (2014). doi:10.1111/ner.12156
T. Morishita, S.M. Fayad, M. Higuchi, K.A. Nestor, K.D. Foote, Deep brain stimulation for treatment-resistant depression: systematic review of clinical outcomes. Neurother. J. Am. Soc. Exp. Neurother. 11, 475–484 (2014). doi:10.1007/s13311-014-0282-1
G. Nair, A. Evans, R.E. Bear, D. Velakoulis, R.G. Bittar, The anteromedial GPi as a new target for deep brain stimulation in obsessive compulsive disorder. J. Clin. Neurosci. Off. J. Neurosurg. Soc. Australas. 21, 815–821 (2014). doi:10.1016/j.jocn.2013.10.003
M.Y. Oh, A.M.D. Abosch, S.H.M.D. Kim, A.E. Lang, A.M.M.D. Lozano, Long-term hardware-related complications of deep brain stimulation. Neurosurgery 2002(50), 1268–1276 (2002)
C. Pollo, A. Kaelin-Lang, M.F. Oertel, L. Stieglitz, E. Taub, P. Fuhr, A.M. Lozano, A. Raabe, M. Schüpbach, Directional deep brain stimulation: an intraoperative double-blind pilot study. Brain 137, 2015–2026 (2014). doi:10.1093/brain/awu102
R.M. Richardson, J.L. Ostrem, P.A. Starr, Surgical repositioning of misplaced subthalamic electrodes in Parkinson’s disease: location of effective and ineffective leads. Stereotact. Funct. Neurosurg. 87, 297–303 (2009). doi:10.1159/000230692
J. Roggendorf, J. Vent, M. Maarouf, C. Haense, A. Thiel, G.R. Fink, R. Hilker, Reversible hypogeusia during bilateral thalamic stimulation for essential tremor. Nervenarzt 79, 454–456 (2008). doi:10.1007/s00115-007-2407-9
W. Sako, Y. Miyazaki, Y. Izumi, R. Kaji, Which target is best for patients with Parkinson’s disease? A meta-analysis of pallidal and subthalamic stimulation. J. Neurol. Neurosurg. Psychiatry 85, 982–986 (2014). doi:10.1136/jnnp-2013-306090
E. Seri-Fainshtat, Z. Israel, A. Weiss, J.M. Hausdorff, Impact of sub-thalamic nucleus deep brain stimulation on dual tasking gait in Parkinson’s disease. J. Neuroengineering Rehabil. 10, 38 (2013). doi:10.1186/1743-0003-10-38
M. Sprengers, K. Vonck, E. Carrette, A.G. Marson, P. Boon, Deep brain and cortical stimulation for epilepsy. Cochrane Database Syst. Rev. 6, CD008497 (2014). doi:10.1002/14651858.CD008497.pub2
A.M. Strutt, R. Simpson, J. Jankovic, M.K. York, Changes in cognitive-emotional and physiological symptoms of depression following STN-DBS for the treatment of Parkinson’s disease. Eur. J. Neurol. Off. J. Eur. Fed. Neurol. Soc. 19, 121–127 (2012). doi:10.1111/j.1468-1331.2011.03447.x
E. Toader, M.M.J. Decré, H.C.F. Martens, Steering deep brain stimulation fields using a high resolution electrode array, in: 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). Presented at the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) (2010), pp. 2061–2064. doi:10.1109/IEMBS.2010.5626472
M. Toft, E. Dietrichs, Aggravated stuttering following subthalamic deep brain stimulation in Parkinson’s disease–two cases. BMC Neurol. 11, 44 (2011). doi:10.1186/1471-2377-11-44
G. Tommasi, P. Krack, V. Fraix, P. Pollak, Effects of varying subthalamic nucleus stimulation on apraxia of lid opening in Parkinson’s disease. J. Neurol. 259, 1944–1950 (2012). doi:10.1007/s00415-012-6447-0
M. Vidailhet, L. Vercueil, J.-L. Houeto, P. Krystkowiak, A.-L. Benabid, P. Cornu, C. Lagrange, S. Tézenas du Montcel, D. Dormont, S. Grand, S. Blond, O. Detante, B. Pillon, C. Ardouin, Y. Agid, A. Destée, P. Pollak, French Stimulation du Pallidum Interne dans la Dystonie (SPIDY) Study Group, Bilateral deep-brain stimulation of the globus pallidus in primary generalized dystonia. N. Engl. J. Med. 352, 459–467 (2005). doi:10.1056/NEJMoa042187
A. Willsie, A.D. Dorval, Charge steering in a novel DBS electrode may accommodate surgical targeting errors, in: 2013 6th International IEEE/EMBS Conference on Neural Engineering (NER). Presented at the 2013 6th International IEEE/EMBS Conference on Neural Engineering (NER) (2013), pp. 152–153. doi:10.1109/NER.2013.6695894
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Willsie, A., Dorval, A. Fabrication and initial testing of the μDBS: a novel Deep Brain Stimulation electrode with thousands of individually controllable contacts. Biomed Microdevices 17, 56 (2015). https://doi.org/10.1007/s10544-015-9961-x
Published:
DOI: https://doi.org/10.1007/s10544-015-9961-x