Abstract
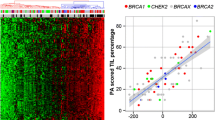
Extensive expression profiling studies have shown that sporadic breast cancer is composed of five clinically relevant molecular subtypes. However, although BRCA1-related tumours are known to be predominantly basal-like, there are few published data on other classes of familial breast tumours. We analysed a cohort of 75 BRCA1, BRCA2 and non-BRCA1/2 breast tumours by gene expression profiling and found that 74% BRCA1 tumours were basal-like, 73% of BRCA2 tumours were luminal A or B, and 52% non-BRCA1/2 tumours were luminal A. Thirty-four tumours were also analysed by single nucleotide polymorphism–comparative genomic hybridization (SNP-CGH) arrays. Copy number data could predict whether a tumour was basal-like or luminal with high accuracy, but could not predict its mutation class. Basal-like BRCA1 and basal-like non-BRCA1 tumours were very similar, and contained the highest number of chromosome aberrations. We identified regions of frequent gain containing potential driver genes in the basal (8q and 12p) and luminal A tumours (1q and 17q). Regions of homozygous loss associated with decreased expression of potential tumour suppressor genes were also detected, including in basal tumours (5q and 9p), and basal and luminal tumours (10q). This study highlights the heterogeneity of familial tumours and the clinical consequences for treatment and prognosis.







Similar content being viewed by others
References
(2004) CHEK2*1100delC and susceptibility to breast cancer: a collaborative analysis involving 10,860 breast cancer cases and 9,065 controls from 10 studies. Am J Hum Genet 74:1175–1182
Abd El-Rehim DM, Ball G, Pinder SE, Rakha E, Paish C, Robertson JF et al (2005) High-throughput protein expression analysis using tissue microarray technology of a large well-characterised series identifies biologically distinct classes of breast cancer confirming recent cDNA expression analyses. Int J Cancer 116:340–350
Adelaide J, Finetti P, Bekhouche I, Repellini L, Geneix J, Sircoulomb F et al (2007) Integrated profiling of basal and luminal breast cancers. Cancer Res 67:11565–11575
Alvarez S, Diaz-Uriarte R, Osorio A, Barroso A, Melchor L, Paz MF et al (2005) A predictor based on the somatic genomic changes of the BRCA1/BRCA2 breast cancer tumors identifies the non-BRCA1/BRCA2 tumors with BRCA1 promoter hypermethylation. Clin Cancer Res 11:1146–1153
Assie G, LaFramboise T, Platzer P, Bertherat J, Stratakis CA, Eng C (2008) SNP arrays in heterogeneous tissue: highly accurate collection of both germline and somatic genetic information from unpaired single tumor samples. Am J Hum Genet 82:903–915
Bane AL, Beck JC, Bleiweiss I, Buys SS, Catalano E, Daly MB et al (2007) BRCA2 mutation-associated breast cancers exhibit a distinguishing phenotype based on morphology and molecular profiles from tissue microarrays. Am J Surg Pathol 31:121–128
Bane AL, Pinnaduwage D, Colby S, Bull SB, O’Malley FP, Andrulis IL (2009) Expression profiling of familial breast cancers demonstrates higher expression of FGFR2 in BRCA2-associated tumors. Breast Cancer Res Treat 117:183–191
BCLC (1997) Pathology of familial breast cancer: differences between breast cancers in carriers of BRCA1 or BRCA2 mutations and sporadic cases. Lancet 349:1505–1510
Beger C, Pierce LN, Kruger M, Marcusson EG, Robbins JM, Welcsh P et al (2001) Identification of Id4 as a regulator of BRCA1 expression by using a ribozyme-library-based inverse genomics approach. Proc Natl Acad Sci USA 98:130–135
Benito M, Parker J, Du Q, Wu J, Xiang D, Perou CM et al (2004) Adjustment of systematic microarray data biases. Bioinformatics 20:105–114
Bergamaschi A, Kim YH, Wang P, Sorlie T, Hernandez-Boussard T, Lonning PE et al (2006) Distinct patterns of DNA copy number alteration are associated with different clinicopathological features and gene-expression subtypes of breast cancer. Genes Chromosomes Cancer 45:1033–1040
Bianco T, Chenevix-Trench G, Walsh DC, Cooper JE, Dobrovic A (2000) Tumour-specific distribution of BRCA1 promoter region methylation supports a pathogenetic role in breast and ovarian cancer. Carcinogenesis 21:147–151
Birgisdottir V, Stefansson OA, Bodvarsdottir SK, Hilmarsdottir H, Jonasson JG, Eyfjord JE (2006) Epigenetic silencing and deletion of the BRCA1 gene in sporadic breast cancer. Breast Cancer Res 8:R38
Budroni M, Cesaraccio R, Coviello V, Sechi O, Pirino D, Cossu A et al (2009) Role of BRCA2 mutation status on overall survival among breast cancer patients from Sardinia. BMC Cancer 9:62
Carter SL, Eklund AC, Kohane IS, Harris LN, Szallasi Z (2006) A signature of chromosomal instability inferred from gene expression profiles predicts clinical outcome in multiple human cancers. Nat Genet 38:1043–1048
Catteau A, Harris WH, Xu CF, Solomon E (1999) Methylation of the BRCA1 promoter region in sporadic breast and ovarian cancer: correlation with disease characteristics. Oncogene 18:1957–1965
Chin K, DeVries S, Fridlyand J, Spellman PT, Roydasgupta R, Kuo WL et al (2006) Genomic and transcriptional aberrations linked to breast cancer pathophysiologies. Cancer Cell 10:529–541
Claus EB, Schildkraut JM, Thompson WD, Risch NJ (1996) The genetic attributable risk of breast and ovarian cancer. Cancer 77:2318–2324
Colella S, Yau C, Taylor JM, Mirza G, Butler H, Clouston P et al (2007) QuantiSNP: an Objective Bayes Hidden-Markov Model to detect and accurately map copy number variation using SNP genotyping data. Nucleic Acids Res 35:2013–2025
Colombo M, Giarola M, Mariani L, Ripamonti CB, De Benedetti V, Sardella M et al. (2008) Cyclin D1 expression analysis in familial breast cancers may discriminate BRCAX from BRCA2-linked cases. Mod Pathol 21:1262–1270
Cox A, Dunning AM, Garcia-Closas M, Balasubramanian S, Reed MW, Pooley KA et al (2007) A common coding variant in CASP8 is associated with breast cancer risk. Nat Genet 39:352–358
Du P, Kibbe WA, Lin SM (2008) lumi: a pipeline for processing Illumina microarray. Bioinformatics 24:1547–1548
Dworkin AM, Spearman AD, Tseng SY, Sweet K, Toland AE (2009). Methylation not a frequent “second hit” in tumors with germline BRCA mutations. Fam Cancer 8:339–346
Easton DF, Pooley KA, Dunning AM, Pharoah PD, Thompson D, Ballinger DG et al (2007) Genome-wide association study identifies novel breast cancer susceptibility loci. Nature 447:1087–1093
Edwards SL, Brough R, Lord CJ, Natrajan R, Vatcheva R, Levine DA et al (2008) Resistance to therapy caused by intragenic deletion in BRCA2. Nature 451:1111–1115
Engelman JA, Luo J, Cantley LC (2006) The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat Rev Genet 7:606–619
Esteller M, Silva JM, Dominguez G, Bonilla F, Matias-Guiu X, Lerma E et al (2000) Promoter hypermethylation and BRCA1 inactivation in sporadic breast and ovarian tumors. J Natl Cancer Inst 92:564–569
Fong PC, Boss DS, Yap TA, Tutt A, Wu P, Mergui-Roelvink M et al (2009) Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N Engl J Med 361:123–134
Foulkes WD, Stefansson IM, Chappuis PO, Begin LR, Goffin JR, Wong N et al (2003) Germline BRCA1 mutations and a basal epithelial phenotype in breast cancer. J Natl Cancer Inst 95:1482–1485
Gronwald J, Jauch A, Cybulski C, Schoell B, Bohm-Steuer B, Lener M et al (2005) Comparison of genomic abnormalities between BRCAX and sporadic breast cancers studied by comparative genomic hybridization. Int J Cancer 114:230–236
Harris LN, You F, Schnitt SJ, Witkiewicz A, Lu X, Sgroi D et al (2007) Predictors of resistance to preoperative trastuzumab and vinorelbine for HER2-positive early breast cancer. Clin Cancer Res 13:1198–1207
Haverty PM, Fridlyand J, Li L, Getz G, Beroukhim R, Lohr S et al (2008) High-resolution genomic and expression analyses of copy number alterations in breast tumors. Genes Chromosomes Cancer 47:530–542
Hedenfalk I, Duggan D, Chen Y, Radmacher M, Bittner M, Simon R et al (2001) Gene-expression profiles in hereditary breast cancer. N Engl J Med 344:539–548
Hedenfalk I, Ringner M, Ben-Dor A, Yakhini Z, Chen Y, Chebil G et al (2003) Molecular classification of familial non-BRCA1/BRCA2 breast cancer. Proc Natl Acad Sci USA 100:2532–2537
Hollestelle A, Nagel JH, Smid M, Lam S, Elstrodt F, Wasielewski M et al. (2009). Distinct gene mutation profiles among luminal-type and basal-type breast cancer cell lines. Breast Cancer Res Treat. doi:10.1007/s10549-009-0460-8
Honrado E, Benitez J, Palacios J (2006) Histopathology of BRCA1- and BRCA2-associated breast cancer. Crit Rev Oncol Hematol 59:27–39
Honrado E, Osorio A, Milne RL, Paz MF, Melchor L, Cascon A et al (2007) Immunohistochemical classification of non-BRCA1/2 tumors identifies different groups that demonstrate the heterogeneity of BRCAX families. Mod Pathol 20:1298–1306
Honrado E, Osorio A, Palacios J, Milne RL, Sanchez L, Diez O et al (2005) Immunohistochemical expression of DNA repair proteins in familial breast cancer differentiate BRCA2-associated tumors. J Clin Oncol 23:7503–7511
Hu Z, Fan C, Oh DS, Marron JS, He X, Qaqish BF et al (2006) The molecular portraits of breast tumors are conserved across microarray platforms. BMC Genomics 7:96
Hu X, Stern HM, Ge L, O’Brien C, Haydu L, Honchell CD et al (2009) Genetic alterations and oncogenic pathways associated with breast cancer subtypes. Mol Cancer Res 7:511–522
Jacquemier J, Ginestier C, Rougemont J, Bardou VJ, Charafe-Jauffret E, Geneix J et al (2005) Protein expression profiling identifies subclasses of breast cancer and predicts prognosis. Cancer Res 65:767–779
Jonsson G, Naylor TL, Vallon-Christersson J, Staaf J, Huang J, Ward MR et al (2005) Distinct genomic profiles in hereditary breast tumors identified by array-based comparative genomic hybridization. Cancer Res 65:7612–7621
Joosse SA, van Beers EH, Tielen IH, Horlings H, Peterse JL, Hoogerbrugge N et al (2009) Prediction of BRCA1-association in hereditary non-BRCA1/2 breast carcinomas with array-CGH. Breast Cancer Res Treat 116:479–489
Kauraniemi P, Hedenfalk I, Persson K, Duggan DJ, Tanner M, Johannsson O et al (2000) MYB oncogene amplification in hereditary BRCA1 breast cancer. Cancer Res 60:5323–5328
Kreike B, van Kouwenhove M, Horlings H, Weigelt B, Peterse H, Bartelink H et al (2007) Gene expression profiling and histopathological characterization of triple-negative/basal-like breast carcinomas. Breast Cancer Res 9:R65
Lakhani SR, Jacquemier J, Sloane JP, Gusterson BA, Anderson TJ, van de Vijver MJ et al (1998) Multifactorial analysis of differences between sporadic breast cancers and cancers involving BRCA1 and BRCA2 mutations. J Natl Cancer Inst 90:1138–1145
Lakhani SR, Van De Vijver MJ, Jacquemier J, Anderson TJ, Osin PP, McGuffog L et al (2002) The pathology of familial breast cancer: predictive value of immunohistochemical markers estrogen receptor, progesterone receptor, HER-2, and p53 in patients with mutations in BRCA1 and BRCA2. J Clin Oncol 20:2310–2318
Lakhani SR, Reis-Filho JS, Fulford L, Penault-Llorca F, van der Vijver M, Parry S et al (2005) Prediction of BRCA1 status in patients with breast cancer using estrogen receptor and basal phenotype. Clin Cancer Res 11:5175–5180
Lingjaerde OC, Baumbusch LO, Liestol K, Glad IK, Borresen-Dale AL (2005) CGH-Explorer: a program for analysis of array-CGH data. Bioinformatics 21:821–822
Mackay A, Tamber N, Fenwick K, Iravani M, Grigoriadis A, Dexter T et al (2009) A high-resolution integrated analysis of genetic and expression profiles of breast cancer cell lines. Breast Cancer Res Treat 118:481–498
Mann GJ, Thorne H, Balleine RL, Butow PN, Clarke CL, Edkins E et al (2006) Analysis of cancer risk and BRCA1 and BRCA2 mutation prevalence in the kConFab familial breast cancer resource. Breast Cancer Res 8:R12
Marcus JN, Watson P, Page DL, Narod SA, Tonin P, Lenoir GM et al (1997) BRCA2 hereditary breast cancer pathophenotype. Breast Cancer Res Treat 44:275–277
Martin RW, Orelli BJ, Yamazoe M, Minn AJ, Takeda S, Bishop DK (2007) RAD51 up-regulation bypasses BRCA1 function and is a common feature of BRCA1-deficient breast tumors. Cancer Res 67:9658–9665
Marty B, Maire V, Gravier E, Rigaill G, Vincent-Salomon A, Kappler M et al (2008) Frequent PTEN genomic alterations and activated phosphatidylinositol 3-kinase pathway in basal-like breast cancer cells. Breast Cancer Res 10:R101
Matros E, Wang ZC, Lodeiro G, Miron A, Iglehart JD, Richardson AL (2005) BRCA1 promoter methylation in sporadic breast tumors: relationship to gene expression profiles. Breast Cancer Res Treat 91:179–186
Matsuda M, Miyagawa K, Takahashi M, Fukuda T, Kataoka T, Asahara T et al (1999) Mutations in the RAD54 recombination gene in primary cancers. Oncogene 18:3427–3430
Melchor L, Benitez J (2008) An integrative hypothesis about the origin and development of sporadic and familial breast cancer subtypes. Carcinogenesis 29:1475–1482
Melchor L, Honrado E, Garcia MJ, Alvarez S, Palacios J, Osorio A et al. (2008) Distinct genomic aberration patterns are found in familial breast cancer associated with different immunohistochemical subtypes. Oncogene 27:3165–3175
Natrajan R, Lambros MB, Rodriguez-Pinilla SM, Moreno-Bueno G, Tan DS, Marchio C et al (2009) Tiling path genomic profiling of grade 3 invasive ductal breast cancers. Clin Cancer Res 15:2711–2722
Nielsen TO, Hsu FD, Jensen K, Cheang M, Karaca G, Hu Z et al (2004) Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res 10:5367–5374
Nordgard SH, Johansen FE, Alnaes GI, Bucher E, Syvanen AC, Naume B et al (2008) Genome-wide analysis identifies 16q deletion associated with survival, molecular subtypes, mRNA expression, and germline haplotypes in breast cancer patients. Genes Chromosomes Cancer 47:680–696
Oldenburg RA, Kroeze-Jansema K, Meijers-Heijboer H, van Asperen CJ, Hoogerbrugge N, van Leeuwen I et al (2006) Characterization of familial non-BRCA1/2 breast tumors by loss of heterozygosity and immunophenotyping. Clin Cancer Res 12:1693–1700
Palacios J, Honrado E, Osorio A, Cazorla A, Sarrio D, Barroso A et al (2003) Immunohistochemical characteristics defined by tissue microarray of hereditary breast cancer not attributable to BRCA1 or BRCA2 mutations: differences from breast carcinomas arising in BRCA1 and BRCA2 mutation carriers. Clin Cancer Res 9:3606–3614
Palacios J, Honrado E, Osorio A, Cazorla A, Sarrio D, Barroso A et al (2005) Phenotypic characterization of BRCA1 and BRCA2 tumors based in a tissue microarray study with 37 immunohistochemical markers. Breast Cancer Res Treat 90:5–14
Perou CM, Jeffrey SS, van de Rijn M, Rees CA, Eisen MB, Ross DT et al (1999) Distinctive gene expression patterns in human mammary epithelial cells and breast cancers. Proc Natl Acad Sci USA 96:9212–9217
Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA et al (2000) Molecular portraits of human breast tumours. Nature 406:747–752
Radpour R, Kohler C, Haghighi MM, Fan AX, Holzgreve W, Zhong XY (2009). Methylation profiles of 22 candidate genes in breast cancer using high-throughput MALDI-TOF mass array. Oncogene 28:2969–2978
Rauta J, Alarmo EL, Kauraniemi P, Karhu R, Kuukasjarvi T, Kallioniemi A (2006) The serine-threonine protein phosphatase PPM1D is frequently activated through amplification in aggressive primary breast tumours. Breast Cancer Res Treat 95:257–263
Rayter S, Elliott R, Travers J, Rowlands MG, Richardson TB, Boxall K et al (2008) A chemical inhibitor of PPM1D that selectively kills cells overexpressing PPM1D. Oncogene 27:1036–1044
Saal LH, Gruvberger-Saal SK, Persson C, Lovgren K, Jumppanen M, Staaf J et al (2008) Recurrent gross mutations of the PTEN tumor suppressor gene in breast cancers with deficient DSB repair. Nat Genet 40:102–107
Shih HA, Couch FJ, Nathanson KL, Blackwood MA, Rebbeck TR, Armstrong KA et al (2002) BRCA1 and BRCA2 mutation frequency in women evaluated in a breast cancer risk evaluation clinic. J Clin Oncol 20:994–999
Smyth GK (2004). Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol 3:Article3
Smyth GK (2005) Limma: linear models for microarray data. Springer, New York, pp 397–420
Sorlie T (2004) Molecular portraits of breast cancer: tumour subtypes as distinct disease entities. Eur J Cancer 40:2667–2675
Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H et al (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98:10869–10874
Sorlie T, Tibshirani R, Parker J, Hastie T, Marron JS, Nobel A et al (2003) Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci USA 100:8418–8423
Sorlie T, Wang Y, Xiao C, Johnsen H, Naume B, Samaha RR et al (2006) Distinct molecular mechanisms underlying clinically relevant subtypes of breast cancer: gene expression analyses across three different platforms. BMC Genomics 7:127
Sotiriou C, Neo SY, McShane LM, Korn EL, Long PM, Jazaeri A et al (2003) Breast cancer classification and prognosis based on gene expression profiles from a population-based study. Proc Natl Acad Sci USA 100:10393–10398
Stefansson OA, Jonasson JG, Johannsson OT, Olafsdottir K, Steinarsdottir M, Valgeirsdottir S et al (2009) Genomic profiling of breast tumours in relation to BRCA abnormalities and phenotypes. Breast Cancer Res 11:R47
Tavtigian SV, Oefner PJ, Babikyan D, Hartmann A, Healey S, Le Calvez-Kelm F et al. (2009) Rare evolutionarily unlikely missense substitutions in ATM confer increased risk of breast cancer. Am J Hum Genet 85:427–446
Tibshirani R, Hastie T, Narasimhan B, Chu G (2002) Diagnosis of multiple cancer types by shrunken centroids of gene expression. Proc Natl Acad Sci USA 99:6567–6572
Tirkkonen M, Johannsson O, Agnarsson BA, Olsson H, Ingvarsson S, Karhu R et al (1997) Distinct somatic genetic changes associated with tumor progression in carriers of BRCA1 and BRCA2 germ-line mutations. Cancer Res 57:1222–1227
Tischkowitz M, Xia B, Sabbaghian N, Reis-Filho JS, Hamel N, Li G et al (2007) Analysis of PALB2/FANCN-associated breast cancer families. Proc Natl Acad Sci USA 104:6788–6793
Turner NC, Reis-Filho JS, Russell AM, Springall RJ, Ryder K, Steele D et al (2007) BRCA1 dysfunction in sporadic basal-like breast cancer. Oncogene 26:2126–2132
van’t Veer LJ, Dai H, van de Vijver MJ, He YD, Hart AA, Mao M et al (2002) Gene expression profiling predicts clinical outcome of breast cancer. Nature 415:530–536
van Beers EH, van Welsem T, Wessels LF, Li Y, Oldenburg RA, Devilee P et al (2005) Comparative genomic hybridization profiles in human BRCA1 and BRCA2 breast tumors highlight differential sets of genomic aberrations. Cancer Res 65:822–827
Vincent-Salomon A, Gruel N, Lucchesi C, MacGrogan G, Dendale R, Sigal-Zafrani B et al (2007) Identification of typical medullary breast carcinoma as a genomic sub-group of basal-like carcinomas, a heterogeneous new molecular entity. Breast Cancer Res 9:R24
Wang K, Li M, Hadley D, Liu R, Glessner J, Grant SF et al (2007) PennCNV: an integrated hidden Markov model designed for high-resolution copy number variation detection in whole-genome SNP genotyping data. Genome Res 17:1665–1674
Weber F, Shen L, Fukino K, Patocs A, Mutter GL, Caldes T et al (2006) Total-genome analysis of BRCA1/2-related invasive carcinomas of the breast identifies tumor stroma as potential landscaper for neoplastic initiation. Am J Hum Genet 78:961–972
Wei M, Grushko TA, Dignam J, Hagos F, Nanda R, Sveen L et al (2005) BRCA1 promoter methylation in sporadic breast cancer is associated with reduced BRCA1 copy number and chromosome 17 aneusomy. Cancer Res 65:10692–10699
Wessels LF, van Welsem T, Hart AA, van’t Veer LJ, Reinders MJ, Nederlof PM (2002) Molecular classification of breast carcinomas by comparative genomic hybridization: a specific somatic genetic profile for BRCA1 tumors. Cancer Res 62:7110–7117
Yan M, kConFab Investigators, Fox SB (2009) Does cyclin D1 discriminate between BRCA2 and BRCAx breast cancers? Histolopathology 55:625–626
Acknowledgements
We wish to thank Heather Thorne, Eveline Niedermayr, all the kConFab research nurses and staff, the heads and staff of the Family Cancer Clinics, and the Clinical Follow Up Study (funded by NHMRC grants 145684, 288704 and 454508) for their contributions to this resource, and the many families who contributed to kConFab. kConFab is supported by grants from the National Breast Cancer Foundation, the National Health and Medical Research Council (NHMRC) and by the Queensland Cancer Fund, the Cancer Councils of New South Wales, Victoria, Tasmania and South Australia, and the Cancer Foundation of Western Australia. GCT and KKK are NHMRC Research Fellows. CE holds the Sondra J. and Stephen R. Hardis Chair of Cancer Genomic Medicine at the Cleveland Clinic, is a Doris Duke Distinguished Clinical Scientist and an American Cancer Society Clinical Research Professor. We also wish to thank Jelle Wesseling, pathologist, and Petra Kristel, technician, for helping with the selection of BRCA2 tumours from the Netherlands Cancer Institute. This study was supported by grants from the National Breast Cancer Foundation and the NHMRC.
Author information
Authors and Affiliations
Consortia
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Waddell, N., Arnold, J., Cocciardi, S. et al. Subtypes of familial breast tumours revealed by expression and copy number profiling. Breast Cancer Res Treat 123, 661–677 (2010). https://doi.org/10.1007/s10549-009-0653-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-009-0653-1