Abstract
Purpose
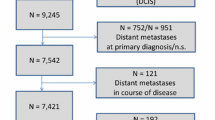
Endocrine therapy (ET) is the mainstream adjuvant treatment for ER-positive breast cancer (BC). We analysed 9293 ER-positive BC patients diagnosed in nine European countries in 2009–2013 to investigate how comorbidities at diagnosis, age, stage and subtype affected ET use over time, and relapse.
Methods
Adjusted odds ratios (ORs) and 95% confidence intervals (95%CIs) of receiving ET were estimated according to Charlson comorbidity, age, stage and subtype using logistic regression. The 2-year cumulative incidence and adjusted sub-hazard ratios (SHRs) of relapse were estimated using competing risk analysis, with all-cause death as the competing event. The z-test was used to assess differences in the proportion of patients receiving ET in 1996–1998 and 2009–2013.
Results
Ninety percent of the patients started adjuvant ET, range 96% (Belgium, Estonia, Slovenia, Spain)—75% (Switzerland). ORs of starting ET were lower for women aged > 75 years, with severe comorbidities, or luminal B HER2-positive cancer. The factors independently increasing the risk of relapse were: not receiving ET (SHR 2.26, 95%CI 1.02–5.03); severe comorbidity (SHR 1.94, 95%CI 1.06–3.55); luminal B, either HER2 negative (SHR 3.06, 95%CI 1.61–5.79) or positive (SHR 3.10, 95%CI 1.36–7.07); stage II (SHR 3.20, 95%CI 1.56–6.57) or stage III (SHR 7.41, 95%CI 3.48–15.73). ET use increased significantly but differently across countries from 51–85% in 1996–1998 to 86–96% in 2009–2013.
Conclusions
ER-positive BC patients in Europe are increasingly prescribed ET but between-country disparities persist. Older women and women with severe comorbidity less frequently receive ET. ET omission and severe comorbidity independently predict early disease relapse.

Similar content being viewed by others
Abbreviations
- BC:
-
Breast cancer
- CCI:
-
Charlson Comorbidity Index
- CI:
-
Confidence interval
- CR:
-
Cancer registry
- ER:
-
Oestrogen receptor
- ET:
-
Endocrine therapy
- HER2:
-
Human epidermal growth factor receptor 2
- HR:
-
High resolution
- OR:
-
Odds ratio
- SHR:
-
Sub-hazard ratio
References
Harvey JM, Clark GM, Osborne CK, Allred DC (1999) Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J Clin Oncol 17:1474–1481. https://doi.org/10.1200/JCO.1999.17.5.1474
Puig-Vives M, Sánchez MJ, Sánchez-Cantalejo J, Torrella-Ramos A, Martos C, Ardanaz E, Chirlaque MD, Perucha J, Díaz JM, Mateos A, Machón M, Marcos-Gragera R (2013) Distribution and prognosis of molecular breast cancer subtypes defined by immunohistochemical biomarkers in a Spanish population-based study. Gynecol Oncol 130(3):609–614. https://doi.org/10.1016/j.ygyno.2013.05.039
Biganzoli L, Marotti L, Hart CD et al (2017) Quality indicators in breast cancer care: an update from the EUSOMA working group. Eur J Cancer 86:59–68. https://doi.org/10.1016/j.ejca.2017.08.017
Sant M, Chirlaque Lopez MD, Agresti R et al (2015) Survival of women with cancers of breast and genital organs in Europe 1999–2007: results of the EUROCARE-5 study. Eur J Cancer 51:2191–2205. https://doi.org/10.1016/j.ejca.2015.07.022
Allemani C, Storm H, Voogd AC et al (2010) Variation in ‘standard care’ for breast cancer across Europe: a EUROCARE-3 high resolution study. Eur J Cancer 46:1528–1536. https://doi.org/10.1016/j.ejca.2010.02.016
Minicozzi P, Van Eycken L, Molinie F et al; European HR Working Group on breast cancer (2019) Comorbidities, age and period of diagnosis influence treatment and outcomes in early breast cancer. Int J Cancer 144(9):2118–2127. https://doi.org/10.1002/ijc.31974
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373–383
Fritz A, Percy C, Jack A et al (2000) International classification of diseases for oncology, 3rd edn. World Health Organisation, Geneva
Carli PM, Izarzugaza I, Koscianska B (2004) International Rules For Multiple Primary Cancers. https://www.encr.eu/sites/default/files/pdf/MPrules_july2004.pdf. Accessed 18 Nov 2019
Goldhirsch A, Wood WC, Coates AS, Gelber RD, Thürlimann B, Senn HJ; Panel members (2011) Strategies for subtypes–dealing with the diversity of breast cancer: highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann Oncol 22:1736–1747. https://doi.org/10.1093/annonc/mdr304
Curigliano G, Burstein HJ, P Winer E et al; St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer (2017) De-escalating and escalating treatments for early-stage breast cancer: the St. Gallen International Expert Consensus Conference on the Primary Therapy of Early Breast Cancer 2017. Ann Oncol 28(8): 1700–1712. https://doi.org/10.1093/annonc/mdx308
Menard SW (2002) Applied logistic regression analysis, 2nd edn. Sage Publications, Thousand Oaks
Fine JP, Gray RJ (1999) A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc 94:496–509
Austin PC, Fine JP (2017) Practical recommendations for reporting Fine-Gray model analyses for competing risk data. Stat Med 36(27):4391–4400. https://doi.org/10.1002/sim.7501
StataCorp. Stata statistical software, 12th release. College Station: StataCorp LP, 2011
Sheppard VB, de Mendoza AH, He J, Jennings Y, Edmonds MC, Oppong BA, Tadesse MG (2018) Initiation of adjuvant endocrine therapy in black and white women with breast cancer. Clin Breast Cancer 18(5):337-346.e1. https://doi.org/10.1016/j.clbc.2017.12.002
Neugut AI, Hillyer GC, Kushi LH et al (2012) Non-initiation of adjuvant hormonal therapy in women with hormone receptor-positive breast cancer: the Breast Cancer Quality of Care Study (BQUAL). Breast Cancer Res Treat 134(1):419–428. https://doi.org/10.1007/s10549-012-2066-9
Grau C, Defourny N, Malicki J et al (2014) Radiotherapy equipment and departments in the European countries: final results from the ESTRO-HERO survey. Radiother Oncol 112:155–164. https://doi.org/10.1016/j.radonc.2014.08.029
Liu Y, Malin JL, Diamant AL, Thind A, Maly RC (2013) Adherence to adjuvant hormone therapy in low-income women with breast cancer: the role of provider-patient communication. Breast Cancer Res Treat 137(3):829–836. https://doi.org/10.1007/s10549-012-2387-8
Ess SM, Herrmann C, Bouchardy C, Neyroud I, Rapiti E, Konzelmann I, Bordoni A, Ortelli L, Rohrmann S, Frick H, Mousavi M, Thürlimann B (2018) Impact of subtypes and comorbidities on breast cancer relapse and survival in population-based studies. Breast 41:151–158. https://doi.org/10.1016/j.breast.2018.07.011
Font R, Espinas JA, Barnadas A, Izquierdo A, Galceran J, Saladie F, Marcos-Gragera R, Torrent A, Manchon-Walsh P, Borras JM (2019) Influence of adherence to adjuvant endocrine therapy on disease-free and overall survival: a population-based study in Catalonia, Spain. Breast Cancer Res Treat 175(3):733–740. https://doi.org/10.1007/s10549-019-05201-3
Karmakar M, Pinto SL, Jordan TR, Mohamed I, Holiday-Goodman M (2017) Predicting adherence to aromatase inhibitor therapy among breast cancer survivors: an application of the protection motivation theory. Breast Cancer (Auckl). https://doi.org/10.1177/1178223417694520
Nabieva N, Kellner S, Fehm T et al (2018) Influence of patient and tumor characteristics on early therapy persistence with letrozole in postmenopausal women with early breast cancer: results of the prospective Evaluate-TM study with 3941 patients. Ann Oncol 29(1):186–192. https://doi.org/10.1093/annonc/mdx630
Hershman DL, Kushi LH, Shao T, Buono D, Kershenbaum A, Tsai WY, Fehrenbacher L, Gomez SL, Miles S, Neugut AI (2010) Early discontinuation and nonadherence to adjuvant hormonal therapy in a cohort of 8,769 early-stage breast cancer patients. J Clin Oncol 28(27):4120–4128. https://doi.org/10.1200/JCO.2009.25.9655
Wulaningsih W, Garmo H, Ahlgren J, Holmberg L, Folkvaljon Y, Wigertz A, Van Hemelrijck M, Lambe M (2018) Determinants of non-adherence to adjuvant endocrine treatment in women with breast cancer: the role of comorbidity. Breast Cancer Res Treat 172(1):167–177. https://doi.org/10.1007/s10549-018-4890-z
Tinari N, Fanizza C, Romero M et al (2015) Identification of subgroups of early breast cancer patients at high risk of nonadherence to adjuvant hormone therapy: results of an Italian survey. Clin Breast Cancer 15(2):e131–e137. https://doi.org/10.1016/j.clbc.2014.10.005
Neugut AI, Zhong X, Wright JD, Accordino M, Yang J, Hershman DL (2016) Nonadherence to medications for chronic conditions and nonadherence to adjuvant hormonal therapy in women with breast cancer. JAMA Oncol 2(10):1326–1332. https://doi.org/10.1001/jamaoncol.2016.1291
Sheppard VB, Faul LA, Luta G et al (2014) Frailty and adherence to adjuvant hormonal therapy in older women with breast cancer: CALGB protocol 369901. J Clin Oncol 32(22):2318–2327. https://doi.org/10.1200/JCO.2013.51.7367
van Hellemond IEG, Geurts SME, Tjan-Heijnen VCG (2018) Current status of extended adjuvant endocrine therapy in early stage breast cancer. Curr Treat Options Oncol 19(5):26. https://doi.org/10.1007/s11864-018-0541-1
Bracken-Clarke DB, Lucas MW, Higgins MJ (2017) Extended adjuvant endocrine therapy in hormone receptor-positive early breast cancer. Breast Care (Basel) 12(3):138–144. https://doi.org/10.1159/000477956
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Aromatase inhibitors versus tamoxifen in early breast cancer: patient-level meta-analysis of the randomised trials. Lancet. 2015;386(10001):1341–1352. https://doi.org/10.1016/s0140-6736(15)61074-1
Mathew J, Asgeirsson KS, Jackson LR, Cheung KL, Robertson JF (2009) Neoadjuvant endocrine treatment in primary breast cancer—review of literature. Breast 18(6):339–344. https://doi.org/10.1016/j.breast.2009.09.012
Hershman DL, Shao T, Kushi LH et al (2011) Early discontinuation and non-adherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer. Breast Cancer Res Treat 126(2):529–537. https://doi.org/10.1007/s10549-010-1132-4
Dispinzieri M, La Rocca E, Meneghini E et al (2018) Discontinuation of hormone therapy for elderly breast cancer patients after hypofractionated whole-breast radiotherapy. Med Oncol 35(7):107. https://doi.org/10.1007/s12032-018-1165-9
Murphy CC, Bartholomew LK, Carpentier MY, Bluethmann SM, Vernon SW (2012) Adherence to adjuvant hormonal therapy among breast cancer survivors in clinical practice: a systematic review. Breast Cancer Res Treat 134(2):459–478. https://doi.org/10.1007/s10549-012-2114-5
Acknowledgements
The authors would like to thank Camilla Amati and Kevin Smart for their help with the English, and the participating cancer registries for collecting and preparing the data as part of their essential role in cancer control.
European High Resolution Working Group on Breast: Belgium: L. Van Eycken, K. Henau (Belgian CR, Flanders); Estonia: K. Innos, M. Mägi (Estonia CR); France: F. Binder-Foucard, M. Velten, (Bas-Rhin CR); A.V. Guizard (Calvados general CR); P. Arveux (Côte d’Or gynaecologic CR); A.S. Woronoff (Doubs CR); B. Amadeo, G. Coureau (Gironde CR); E. Marrer, S. Boyer (Haut-Rhin CR); N. Léone (Haute-Vienne CR); B. Trétarre (Hérault CR); M. Colonna, P. Delafosse (Isère CR); V. Démaret, K. Ligier (Lille et sa région CR); S. Ayrault-Piault (Loire-Atlantique CR); S. Bara (Manche CR); G. Defossez (Poitou–Charentes CR); B. Lapotre-Ledoux (Somme CR); L. Daubisse-Marliac, P. Grosclaude (Tarn CR); A. Cowppli-Bony (Vendée CR); F Molinié (Loire-Atlantique/Vendée CR, FRANCIM);Germany: V. Babaev, A. Katalinic (Germany-Schleswig–Holstein CR); Italy: C. Amati, P. Baili, S. Bonfarnuzzo, E. Meneghini, P. Minicozzi, G. Moretti, M. Sant (Fondazione IRCCS Istituto Nazionale dei Tumori, Milan); Silvia Iacovacci, Susanna Busco (Latina CR); E. Marani, C. Casella, R. (Liguria CR, Ospedale Policlinico San Martino IRCCS); G. Carrozzi, C. Cirilli (Modena CR); R. Amodio, R. Cusimano (Palermo CR); C. Nicita, R. Tumino (Ragusa CR, ASP); F. Ferrari, L. Mangone (Reggio Emilia CR, IRCCS AUSL ASMN); F. Stracci, F. Bianconi (Umbria CR); P. Contiero, G. Tagliabue (Varese Province CR, Fondazione IRCCS Istituto Nazionale dei Tumori); Poland: L. Molong, J. Rachtan (Cracow CR); J. Janowski, A. Kubiak (Greater Poland CR); S. Góźdź, P. Macek (Kielce CR); M. Bielska-Lasota (National Institute of Public Health-NIH, Warsaw); J. Błaszczyk, K. Kępska (Lower Silesia CR, Wroclaw); Portugal: J. Bastos, B. Carrito (Central Portugal CR); M. J. Bento, C. Castro (Northern Portugal CR); A. Mayer-da-Silva, A. Miranda, (Southern Portugal CR); Slovenia: V. Zadnig (National CR), G. Plavc (Institute of Oncology Ljubljana); Spain: L. Gil Majuelo, N. Larrañaga (Basque Country CR, CIBERESP); C. Sabater, A. Torella (Castellón-Valencia CR); R. Marcos-Gragera, M. Puigdemont (Girona CR, CIBERESP, ICO,IDIBGI); E. Molina-Portillo, M. J. Sánchez-Pérez (Granada CR, EASP, CIBERESP, ibs.Granada); E. Ardanaz, M. Guevara (Navarra CR, CIBERESP, IdiSNa); J. Galceran, F. Saladié (Tarragona CR);Switzerland: C. Bouchardy, E. Fournier (Geneva CR, University of Geneva).
Funding
These results have been achieved within the framework of the call on “Translational research on tertiary prevention in cancer patients (TRANSCAN)” carried out by ERA-NET, with funding from: Ministero della Salute, Dipartimento della Sanità Pubblica e dell’Innovazione, Direzione Generale della Ricerca Sanitaria e Biomedica e della Vigilanza sugli Enti (MoH), Viale Giorgio Ribotta 5, Roma, Italy; Fonds voor Wetenschappelijk Onderzoek Vlaanderen, FWO (Research Foundation Flanders), Egmonstraat 5 B-1000 Brussels, Belgium; Institut National du Cancer (INCa), 52 avenue André Morizet, 92513 Boulogne Billancourt Cedex, France; Federal Ministry of Education and Research (BMBF), Heinrich-Konen-Str. 1, D-53227 Bonn, Germany; Narodowego Centrum Badań i Rozwoju (NCBR) ul. Nowogrodzka 47a, 00695 Warszawa Poland; Ministrstvo za izobraževanje, znanost in šport (Ministry of Education, Science and Sport, MIZS), Masarykova 16, 1000 Ljubljana, Slovenia; The Instituto de Salud Carlos III (ISCIII), C/Sinesio Delgado 4, 28029 Madrid, Spain (Grant No. AC14/00036); Fundación Bancaria Caixa d’Estalvis i Pensions de Barcelona, Plaza Weyler, 3, 07001 Palma, Spain. Andalusian Department of Health, Spain (Grant No. PI-0152/2017).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards (N. INT 197/14).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A list of the members of the European High Resolution Working Group on Breast Cancer is given in the Acknowledgment.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sant, M., Meneghini, E., Bastos, J. et al. Endocrine treatment and incidence of relapse in women with oestrogen receptor-positive breast cancer in Europe: a population-based study. Breast Cancer Res Treat 183, 439–450 (2020). https://doi.org/10.1007/s10549-020-05761-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05761-9