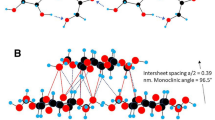
Abstract
One of the most important interactions within the paracrystalline matrix of the plant cell wall occurs between cellulose microfibrils to allow for the formation of larger diameter macrofibrils. Here, we have used computational techniques to investigate how different microfibril surfaces might adsorb onto one another. Molecular dynamics simulations show that limited direct adsorption occurs between non-polar surfaces and free energy of desorption calculations suggest this is due to a high energy barrier for the removal of a single layer of water between these surfaces. Further, it is predicted that when microfibril aggregation occurs, significant conformational changes take place at the surfaces of interaction involving O2 dihedral angles, exocyclic C6 conformation, and microfibril chain tilt. It is more likely that direct interactions initially take place between polar (110) surfaces, and that surface interactions occur between the same types of surface, such as 110 to 110, 1–10 to 1–10 or 200 to 100, where hydrogen bonds can be formed, to stabilise the aggregate. Additionally, we have identified that for the exocyclic group of a glucose residue to change conformation in origin layers, the O2 dihedral in residues before and adjacent to the glucose must rotate to a more cis-like conformation, compared to the trans-like conformation observed in crystalline cellulose. This change in exocyclic conformation occurs due to a slight shift in adjacent chains that preferentially stabilises the exocyclic conformation change in a specific glucose residue of each cellobiose repeat.






Similar content being viewed by others
References
Alekozai EM, GhattyVenkataKrishna PK, Uberbacher EC et al (2014) Simulation analysis of the cellulase Cel7A carbohydrate binding module on the surface of the cellulose Iβ. Cellulose 21:951–971. doi:10.1007/s10570-013-0026-0
Anderson CT, Carroll A, Akhmetova L, Somerville C (2010) Real-time imaging of cellulose reorientation during cell wall expansion in Arabidopsis roots. Plant Physiol 152:787–796. doi:10.1104/pp.109.150128
Baskin TI (2005) Anisotropic expansion of the plant cell wall. Annu Rev Cell Dev Biol 21:203–222. doi:10.1146/annurev.cellbio.20.082503.103053
Beckham GT, Matthews JF, Peters B et al (2011) Molecular-level origins of biomass recalcitrance: decrystallization free energies for four common cellulose polymorphs. J Phys Chem B 115:4118–4127. doi:10.1021/jp1106394
Bellesia G, Asztalos A, Shen T et al (2010) In silico studies of crystalline cellulose and its degradation by enzymes. Acta Crystallogr Sect D: Biol Crystallogr 66:1184–1188. doi:10.1107/S0907444910029483
Bergenstråhle M, Berglund LA, Mazeau K (2007) Thermal response in crystalline Ibeta cellulose: a molecular dynamics study. J Phys Chem B 111:9138–9145. doi:10.1021/jp072258i
Bergenstråhle M, Thormann E, Nordgren N, Berglund LA (2009) Force pulling of single cellulose chains at the crystalline cellulose-liquid interface: a molecular dynamics study. Langmuir 25:4635–4642. doi:10.1021/la803915c
Bergenstråhle M, Wohlert J, Himmel ME, Brady JW (2010) Simulation studies of the insolubility of cellulose. Carbohydr Res 345:2060–2066. doi:10.1016/j.carres.2010.06.017
Boraston AB, Bolam DN, Gilbert HJ, Davies GJ (2004) Carbohydrate-binding modules: fine-tuning polysaccharide recognition. Biochem J 382:769–781. doi:10.1042/BJ20040892
Brett CT (2000) Cellulose microfibrils in plants: biosynthesis, deposition, and integration into the cell wall. Int Rev Cytol 199:161–199. doi:10.1016/s0074-7696(00)99004-1
Bu L, Himmel ME, Crowley MF (2015) The molecular origins of twist in cellulose I-beta. Carbohydr Polym 125:146–152. doi:10.1016/j.carbpol.2015.02.023
Busse-Wicher M, Gomes TCF, Tryfona T et al (2014) The pattern of xylan acetylation suggests xylan may interact with cellulose microfibrils as a twofold helical screw in the secondary plant cell wall of Arabidopsis thaliana. Plant J 79:492–506. doi:10.1111/tpj.12575
Chen P, Nishiyama Y, Putaux J-L, Mazeau K (2014) Diversity of potential hydrogen bonds in cellulose I revealed by molecular dynamics simulation. Cellulose 21:897–908. doi:10.1007/s10570-013-0053-x
Cosgrove DJ (2001) Wall structure and wall loosening. A look backwards and forwards. Plant Physiol 125:131–134. doi:10.1104/pp.125.1.131
Cosgrove DJ, Jarvis MC (2012) Comparative structure and biomechanics of plant primary and secondary cell walls. Front Plant Sci 3:1–6. doi:10.3389/fpls.2012.00204
Dagel DJ, Liu YS, Zhong L et al (2011) In situ imaging of single carbohydrate-binding modules on cellulose microfibrils. J Phys Chem B 115:635–641. doi:10.1021/jp109798p
Darden T, York D, Pedersen L (1993) Particle mesh Ewald: an N·log(N) method for Ewald sums in large systems. J Chem Phys 98:10089. doi:10.1063/1.464397
Davies LM, Harris PJ (2003) Atomic force microscopy of microfibrils in primary cell walls. Planta 217:283–289. doi:10.1007/s00425-003-0979-6
Ding S-Y, Himmel ME (2006) The maize primary cell wall microfibril: a new model derived from direct visualization. J Agric Food Chem 54:597–606. doi:10.1021/jf051851z
Ding S-Y, Liu Y-S, Zeng Y et al (2012) How does plant cell wall nanoscale architecture correlate with enzymatic digestibility? Science 338:1055–1060. doi:10.1126/science.1227491
Ding S-Y, Zhao S, Zeng Y (2014) Size, shape, and arrangement of native cellulose fibrils in maize cell walls. Cellulose 21:863–871. doi:10.1007/s10570-013-0147-5
Doblin MS, Pettolino F, Bacic A (2010) Evans review: plant cell walls: the skeleton of the plant world. Funct Plant Biol 37:357–381. doi:10.1071/FP09279
Donaldson L (2007) Cellulose microfibril aggregates and their size variation with cell wall type. Wood Sci Technol 41:443–460. doi:10.1007/s00226-006-0121-6
Fernandes AN, Thomas LH, Altaner CM et al (2011) Nanostructure of cellulose microfibrils in spruce wood. Proc Natl Acad Sci USA 108:E1195–E1203. doi:10.1073/pnas.1108942108
Fiorin G, Klein ML, Hénin J (2013) Using collective variables to drive molecular dynamics simulations. Mol Phys 111:3345–3362. doi:10.1080/00268976.2013.813594
Foston M (2014) Advances in solid-state NMR of cellulose. Curr Opin Biotechnol 27C:176–184. doi:10.1016/j.copbio.2014.02.002
Georgelis N, Yennawar NH, Cosgrove DJ (2012) Structural basis for entropy-driven cellulose binding by a type-A cellulose-binding module (CBM) and bacterial expansin. Proc Natl Acad Sci 109:14830–14835. doi:10.1073/pnas.1213200109
Gomes TCF, Skaf MS (2012) Cellulose-builder: a toolkit for building crystalline structures of cellulose. J Comput Chem 33:1338–1346. doi:10.1002/jcc.22959
Gross AS, Chu J-W (2010) On the molecular origins of biomass recalcitrance: the interaction network and solvation structures of cellulose microfibrils. J Phys Chem B 114:13333–13341. doi:10.1021/jp106452m
Grossfield A An implementation of WHAM: the weighted histogram analysis method. http://membrane.urmc.rochester.edu/Software/WHAM/WHAM.html.
Guvench O, Greene SN, Kamath G et al (2008) Additive empirical force field for hexopyranose monosaccharides. J Comput Chem 29:2543–2564. doi:10.1002/jcc
Ha MA, Apperley DC, Evans BW et al (1998) Fine structure in cellulose microfibrils: NMR evidence from onion and quince. Plant J 16:183–190. doi:10.1046/j.1365-313x.1998.00291.x
Haigler CH, Brown RM (1986) Transport of rosettes from the golgi apparatus to the plasma membrane in isolated mesophyll cells of Zinnia elegans during differentiation to tracheary elements in suspension culture. Protoplasma 134:111–120. doi:10.1007/BF01275709
Hanus J, Mazeau K (2006) The xyloglucan–cellulose assembly at the atomic scale. Biopolymers 82:59–73. doi:10.1002/bip
Harris D, Bulone V, Ding S-Y, DeBolt S (2010) Tools for cellulose analysis in plant cell walls. Plant Physiol 153:420–426. doi:10.1104/pp.110.154203
Hayashi T (1989) Xyloglucans in the primary cell wall. Annu Rev Plant Physiol Plant Mol Biol 40:139–168. doi:10.1146/annurev.pp.40.060189.001035
Herth W (1983) Arrays of plasma-membrane “rosettes” involved in cellulose microfibril formation of Spirogyra. Planta 159:347–356. doi:10.1007/BF00393174
Hung S-W, Hsiao P-Y, Chieng C-C (2011) Dynamic information for cardiotoxin protein desorption from a methyl-terminated self-assembled monolayer using steered molecular dynamics simulation. J Chem Phys 134:194705. doi:10.1063/1.3592559
Jarzynski C (1997) Nonequilibrium equality for free energy differences. Phys Rev Lett 78:2690–2693. doi:10.1103/PhysRevLett.78.2690
Kalé L, Skeel R, Bhandarkar M et al (1999) NAMD2: greater scalability for parallel molecular dynamics. J Comput Phys 151:283–312. doi:10.1006/jcph.1999.6201
Keegstra K, Talmadge KW, Bauer WD, Albersheim P (1973) The structure of plant cell walls: III. A model of the walls of suspension-cultured sycamore cells based on the interconnections of the macromolecular components. Plant Physiol 51:188–197. doi:10.1104/pp.51.1.188
Kumar S, Bouzida D, Swendsen RH et al (1992) The weighted histogram analysis method for free-energy calculations on biomolecules. I. The method. J Comput Chem 13:1011–1021. doi:10.1002/jcc.540130812
Larsson PT, Hult E, Wickholm K et al (1999) CP/MAS-NMR spectroscopy applied to structure and interaction studies on cellulose I. Solid State Nucl Magn Reson 15:31–40. doi:10.1016/S0926-2040(99)00044-2
Lehtiö J, Sugiyama J, Gustavsson M et al (2003) The binding specificity and affinity determinants of family 1 and family 3 cellulose binding modules. Proc Natl Acad Sci USA 100:484–489. doi:10.1073/pnas.212651999
Li D-C, Ji B-H (2012) Free energy calculation of single molecular interaction using Jarzynski’s identity method: the case of HIV-1 protease inhibitor system. Acta Mech Sin 28:891–903. doi:10.1007/s10409-012-0112-9
Marga F, Grandbois M, Cosgrove DJ, Baskin TI (2005) Cell wall extension results in the coordinate separation of parallel microfibrils: Evidence from scanning electron microscopy and atomic force microscopy. Plant J 43:181–190. doi:10.1111/j.1365-313X.2005.02447.x
Martínez-Sanz M, Gidley MJ, Gilbert EP (2015a) Application of X-ray and neutron small angle scattering techniques to study the hierarchical structure of plant cell walls: a review. Carbohydr Polym 125:120–134. doi:10.1016/j.carbpol.2015.02.010
Martínez-Sanz M, Lopez-Sanchez P, Gidley MJ, Gilbert EP (2015b) Evidence for differential interaction mechanism of plant cell wall matrix polysaccharides in hierarchically-structured bacterial cellulose. Cellulose. doi:10.1007/s10570-015-0614-2
Matthews JF, Skopec CE, Mason PE et al (2006) Computer simulation studies of microcrystalline cellulose Ibeta. Carbohydr Res 341:138–152. doi:10.1016/j.carres.2005.09.028
Matthews JF, Bergenstråhle M, Beckham GT et al (2011a) High-temperature behavior of cellulose I. J Phys Chem B 115:2155–2166. doi:10.1021/jp1106839
Matthews JF, Himmel ME, Crowley MF (2011b) Conversion of cellulose Iα to Iβ via a high temperature intermediate (I-HT) and other cellulose phase transformations. Cellulose 19:297–306. doi:10.1007/s10570-011-9608-x
Matthews JF, Beckham GT, Bergenstråhle-Wohlert M et al (2012) Comparison of cellulose Iβ simulations with three carbohydrate force fields. J Chem Theory Comput 8:735–748. doi:10.1021/ct2007692
Mazeau K (2005) Structural micro-heterogeneities of crystalline Iβ-cellulose. Cellulose 12:339–349. doi:10.1007/s10570-005-2200-5
Mazeau K, Vergelati C (2002) Atomistic modeling of the adsorption of benzophenone onto cellulosic surfaces. Langmuir 18:1919–1927. doi:10.1021/la010792q
McFarlane HE, Döring A, Persson S (2014) The cell biology of cellulose synthesis. Annu Rev Plant Biol 65:69–94. doi:10.1146/annurev-arplant-050213-040240
Miyamoto S, Kollman PA (1992) Settle: an analytical version of the SHAKE and RATTLE algorithm for rigid water models. J Comput Chem 13:952–962. doi:10.1002/jcc.540130805
Miyamoto H, Schnupf U, Brady JW (2014) Water structuring over the hydrophobic surface of cellulose. J Agric Food Chem 62:11017–11023. doi:10.1021/jf501763r
Moon RJ, Martini A, Nairn J et al (2011) Cellulose nanomaterials review: structure, properties and nanocomposites. Chem Soc Rev 40:3941. doi:10.1039/c0cs00108b
Muthukumar L, Khare R (2013) Molecular dynamics simulation of free energy of desorption of cellohexaose from a cellulose crystal surface. In: Fitzgerald G, Govind N (eds) Applications of molecular modeling to challenges in clean energy. American Chemical Society, Washington, DC, pp 1–17
Newman RH, Hill SJ, Harris PJ (2013) Wide-angle x-ray scattering and solid-state nuclear magnetic resonance data combined to test models for cellulose microfibrils in mung bean cell walls. Plant Physiol 163:1558–1567. doi:10.1104/pp.113.228262
Nishiyama Y, Langan P, Chanzy H (2002) Crystal structure and hydrogen-bonding system in cellulose Iβ from synchrotron X-ray and neutron fiber diffraction. J Am Chem Soc 124:9074–9082. doi:10.1021/ja0257319
Oehme DP, Downton MT, Doblin MS et al (2015) Unique aspects of the structure and dynamics of Iβ elementary cellulose microfibrils revealed by computational simulations. Plant Physiol 168:3–17. doi:10.1104/pp.114.254664
Park YB, Cosgrove DJ (2012) A revised architecture of primary cell walls based on biomechanical changes induced by substrate-specific endoglucanases. Plant Physiol 158:1933–1943. doi:10.1104/pp.111.192880
Park S, Khalili-Araghi F, Tajkhorshid E, Schulten K (2003) Free energy calculation from steered molecular dynamics simulations using Jarzynski’s equality. J Chem Phys 119:3559. doi:10.1063/1.1590311
Pauly M, Albersheim P, Darvill A, York WS (1999) Molecular domains of the cellulose/xyloglucan network in the cell walls of higher plants. Plant J 20:629–639. doi:10.1046/j.1365-313X.1999.00630.x
Pauly M, Gille S, Liu L et al (2013) Hemicellulose biosynthesis. Planta 238:627–642. doi:10.1007/s00425-013-1921-1
Payne CM, Himmel ME, Crowley MF, Beckham GT (2011) Decrystallization of oligosaccharides from the cellulose Iβ surface with molecular simulation. J Phys Chem Lett 2:1546–1550. doi:10.1021/jz2005122
Peri S, Muthukumar L, Nazmul Karim M, Khare R (2012) Dynamics of cello-oligosaccharides on a cellulose crystal surface. Cellulose 19:1791–1806. doi:10.1007/s10570-012-9771-8
Ryckaert J-P, Ciccotti G, Berendsen HJC (1977) Numerical integration of the cartesian equations of motion of a system with constraints: molecular dynamics of n-alkanes. J Comput Phys 23:327–341. doi:10.1016/0021-9991(77)90098-5
Somerville C (2006) Cellulose synthesis in higher plants. Annu Rev Cell Dev Biol 22:53–78. doi:10.1146/annurev.cellbio.22.022206.160206
Sorek N, Sorek H, Kijac A et al (2014) The Arabidopsis COBRA protein facilitates cellulose crystallization at the plasma membrane. J Biol Chem 289:34911–34920
Sotomayor M, Schulten K (2007) Single-molecule experiments in vitro and in silico. Science 316:1144–1148. doi:10.1126/science.1137591
Talbott LD, Ray PM (1992) Molecular size and separability features of pea cell wall polysaccharides: implications for models of primary wall structure. Plant Physiol 98:357–368. doi:10.1104/pp.98.1.357
Thomas LH, Forsyth VT, Sturcová A et al (2013) Structure of cellulose microfibrils in primary cell walls from collenchyma. Plant Physiol 161:465–476. doi:10.1104/pp.112.206359
Vain T, Crowell EF, Timpano H et al (2014) The cellulase KORRIGAN is part of the cellulose synthase complex. Plant Physiol. doi:10.1104/pp.114.241216
Wickholm K, Larsson PT, Iversen T (1998) Assignment of non-crystalline forms in cellulose I by CP/MAS 13C NMR spectroscopy. Carbohydr Res 312:123–129. doi:10.1016/S0008-6215(98)00236-5
Ytreberg FM, Zuckerman DM (2004) Efficient use of nonequilibrium measurement to estimate free energy differences for molecular systems. J Comput Chem 25:1749–1759. doi:10.1002/jcc.20103
Zhang Q, Brumer H, Ågren H, Tu Y (2011) The adsorption of xyloglucan on cellulose: effects of explicit water and side chain variation. Carbohydr Res 346:2595–2602. doi:10.1016/j.carres.2011.09.007
Zhao Z, Shklyaev OE, Nili A et al (2013) Cellulose microfibril twist, mechanics, and implication for cellulose biosynthesis. J Phys Chem A 117:2580–2589
Zhao Z, Crespi VH, Kubicki JD et al (2014) Molecular dynamics simulation study of xyloglucan adsorption on cellulose surfaces: effects of surface hydrophobicity and side-chain variation. Cellulose 21(1025–1039):1041–1042. doi:10.1007/s10570-013-0041-1
Acknowledgments
This work was funded by a grant from the Australia Research Council to the ARC Centre of Excellence in Plant Cell Walls (MJG, MSD and AB) (CE110001007); and the Victorian Life Sciences Computation Initiative (VLSCI) grant numbers “VR0319” on its Peak Computing Facility at the University of Melbourne, an initiative of the Victorian State Government. We thank Daniel Weber (University of Melbourne) for his assistance in developing scripts to setup and analyse umbrella sampling simulations.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Plots of the movement of the centre of mass of the freeM in the xy plane over the length of the simulation, coloured by squared displacement of the centre of mass averaged over 20 snapshots. Each row details the movement of the freeM for each replicate of that surface-surface unbiased adsorption. From top to bottom the surface-surface adsorptions are: 200 vs 200; 200 vs 110; 200 vs 1-10; 200 vs 100; 100 vs 110; 100 vs 1-10; 100 vs 100; 110 vs 110; 110 vs 1-10; 1-10 vs 1-10. Replicates that contained the snapshot with most favourable interaction energy have a thickened border (TIFF 3579 kb)
Fig. S2
110 to 110 unbiased adsorption simulation (A) force and (B) work curves for all 10 replicates of the SMD simulations (TIFF 929 kb)
Rights and permissions
About this article
Cite this article
Oehme, D.P., Doblin, M.S., Wagner, J. et al. Gaining insight into cell wall cellulose macrofibril organisation by simulating microfibril adsorption. Cellulose 22, 3501–3520 (2015). https://doi.org/10.1007/s10570-015-0778-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-015-0778-9