Abstract
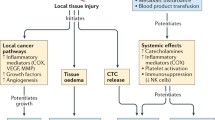
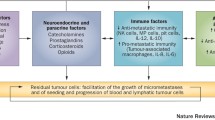
Cancer, currently the leading cause of death in the population aged less than 85 years, poses a significant global disease burden and is anticipated to continue to increase in incidence in both developed and developing nations. A substantial proportion of cancers are amenable to surgery, with more than 60% of patients undergoing tumour resection. Up to 80% of patients will receive anaesthesia for diagnostic, therapeutic or palliative intervention. Alarmingly, retrospective studies have implicated surgical stress in disease progression that is predominantly characterised by metastatic disease—the primary cause of cancer-associated mortality. Our understanding of the mechanisms of surgical stress and impact of perioperative interventions is, however, far from complete. Accumulating evidence from preclinical studies suggests that adrenergic-inflammatory pathways may contribute to cancer progression. Importantly, these pathways are amenable to modulation by adapting surgical (e.g. minimally invasive surgery) and anaesthetic technique (e.g. general vs. neuraxial anaesthesia). Disturbingly, drugs used for general anaesthesia (e.g. inhalational vs. intravenous anaesthesia and potentially opioid analgesia) may also affect behaviour of tumour cells and immune cells, suggesting that choice of anaesthetic agent may also be linked to adverse long-term cancer outcomes. Critically, current clinical practice guidelines on the use of anaesthetic techniques, anaesthetic agents and perioperative adjuvants (e.g. anti-inflammatory drugs) during cancer surgery do not take into account their potential effect on cancer outcomes due to a lack of robust prospective data. To help address this gap, we provide an up-to-date review of current clinical evidence supporting or refuting the role of perioperative stress, anaesthetic techniques and anaesthetic agents in cancer progression and review pre-clinical studies that provide insights into biological mechanisms.
Similar content being viewed by others
References
Ferlay JSI, Ervik M (2013) Cancer incidence and mortality worldwide. GLOBOCAN. IARC CancerBase No. 11 International Agency for Research on Cancer, Lyon
Sullivan R, Peppercorn J, Sikora K, Zalcberg J, Meropol NJ, Amir E, Khayat D, Boyle P, Autier P, Tannock I, Fojo T, Siderov J, Williamson S, Camporesi S, McVie J, Purushotham A, Naredi P, Eggermont A, Brennan M, Steinberg M, De Ridder M, McCloskey S, Verellen D, Roberts T, Storme G, Hicks R, Ell P, Hirsch B, Carbone D, Schulman K, Catchpole P, Taylor D, Geissler J, Brinker NG, Meltzer D, Kerr D, Aapro M (2011) Delivering affordable cancer care in high-income countries. Lancet Oncol 12(10):933–980. doi:10.1016/s1470-2045(11)70141-3
Alkire B, Raykar N, Shrime M, Weiser T, Bickler S, Rose J, Nutt C, Greenberg S, Kotagal M, Riesel J, Esquivel M, Uribe-Leitz T, Molina G, Roy N, Meara J, Farmer P (2015) Global access to surgical care: a modelling study. Lancet Glob Health 3(6):e316–e323. doi:10.1016/S2214-109X(15)70115-4
Mehlen P, Puisieux A (2006) Metastasis: a question of life or death. Nat Rev Cancer 6(6):449–458. doi:10.1038/nrc1886
Newton P, Mason J, Venkatappa N, Jochelson M, Hurt B, Nieva J, Comen E, Norton L, Kuhn P (2015) Spatiotemporal progression of metastatic breast cancer: a Markov chain model highlighting the role of early metastatic sites. NPJ Breast Cancer 1:15018. doi:10.1038/npjbcancer.2015.18
Zellmer V, Zhang S (2014) Evolving concepts of tumor heterogeneity. Cell Biosci 4:69. doi:10.1186/2045-3701-4-69
Horowitz M, Neeman E, Sharon E, Ben-Eliyahu S (2015) Exploiting the critical perioperative period to improve long-term cancer outcomes. Nat Rev Clin Oncol 12(4):213–226. doi:10.1038/nrclinonc.2014.224
Sloan EK, Priceman SJ, Cox BF, Yu S, Pimentel MA, Tangkanangnukul V, Arevalo JM, Morizono K, Karanikolas BD, Wu L, Sood AK, Cole SW (2010) The sympathetic nervous system induces a metastatic switch in primary breast cancer. Cancer Res 70(18):7042–7052. doi:10.1158/0008-5472.can-10-0522
Thaker P (2006) Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nat Med 12:939–944
Le C, Nowell C, Kim-Fuchs C, Botteri E, Hiller J, Ismail H, Pimentel M, Chai M, Karnezis T, Rotmensz N, Renne G, Gandini S, Pouton C, Ferrari D, Möller A, Stacker S, Sloan E (2016) Chronic stress in mice remodels lymph vasculature to promote tumour cell dissemination. Nat Commun 7:10634. doi:10.1038/ncomms10634
Chang A, Le C, Walker A, Creed S, Pon C, Albold S, Carroll D, Halls M, Lane J, Riedel B, Ferrari D, Sloan E (2016) Beta2-Adrenoceptors on tumor cells play a critical role in stress-enhanced metastasis in a mouse model of breast cancer. Brain Behav Immun 57:106–115. doi:10.1016/j.bbi.2016.06.011
Creed S, Le C, Hassan M, Pon C, Albold S, Chan K, Berginski M, Huang Z, Bear J, Lane J, Halls M, Ferrari D, Nowell C, Sloan E (2015) Beta2-adrenoceptor signaling regulates invadopodia formation to enhance tumor cell invasion. Breast Cancer Res 17(1):145. doi:10.1186/s13058-015-0655-3
Hiller J, Hacking M, Link E, Wessels K, Riedel B (2014) Perioperative epidural analgesia reduces cancer recurrence after gastro-oesophageal surgery. Acta Anaesthesiol Scand 58(3):281–290. doi: 10.1111/aas.12255 doi
Wigmore T, Mohammed K, Jhanji S (2016) Long-term survival for patients undergoing volatile versus IV anaesthesia for cancer surgery a retrospective analysis. Anesthesiology 124(1):69–79. doi:10.1097/ALN.0000000000000936
Amato A, Pescatori M (2006) Perioperative blood transfusions for the recurrence of colorectal cancer. Cochrane Database Syst Rev 1:Cd005033. doi:10.1002/14651858.CD005033.pub2
Aloia T, Zimmitti G, Conrad C, Gottumukalla V, Kopetz S, Vauthey J (2014) Return to intended oncologic treatment (RIOT): a novel metric for evaluating the quality of oncosurgical therapy for malignancy. J Surg Oncol 110(2):107–114. doi:10.1002/jso.23626
Cata J, Bugada D, Marchesini M, De Gregori M, Allegri M (2016) Opioids and cancer recurrence: a brief review of the literature. Cancer Cell Microenviron. doi:10.14800/ccm.1159
Wigmore T, Farquhar-Smith P (2016) Opioids and cancer: friend or foe? Curr Opin Supportive Palliat Care 10(2):109–118. doi:10.1097/spc.0000000000000208
Singleton P, Moss J (2010) Effect of perioperative opioids on cancer recurrence: a hypothesis. Future Oncol 6(8):1237–1242. doi:10.2217/fon.10.99
Sekandarzad M, van Zundert A, Lirk P, Doornebal C, Hollmann M (2017) Perioperative anaesthesia care and tumor progression. Anesth Analg 124(5):1697–1708. doi:10.1213/ane.0000000000001652
Wigmore T, Gottumukkala V, Riedel B (2016) Making the case for the subspecialty of onco-anaesthesia. Int Anesthesiol Clin 54(4):19–28. doi:10.1097/aia.0000000000000117
Schlagenhauff B, Ellwanger U, Breuninger H, Stroebel W, Rassner G, Garbe C (2000) Prognostic impact of the type of anaesthesia used during the excision of primary cutaneous melanoma. Melanoma Res 10(2):165–169
Lee J, Kang S, Kim Y, Kim H, Kim B (2016) Effects of propofol-based total intravenous anaesthesia on recurrence and overall survival in patients after modified radical mastectomy: a retrospective study. Korean J Anesthesiol 69(2):126–132. doi:10.4097/kjae.2016.69.2.126
Enlund M, Berglund A, Andreasson K, Cicek C, Enlund A, Bergkvist L (2014) The choice of anaesthetic—sevoflurane or propofol—and outcome from cancer surgery: a retrospective analysis. Ups J Med Sci 119(3):251–261
Cata J, Hagan K, Bhavsar S, Arunkumar R, Grasu R, Dang A, Carlson R, Arnold B, Potylchansky Y, Lipski I, McHugh T, Jimenez F, Nguyen A, Feng L, Rahlfs T (2017) The use of isoflurane and desflurane as inhalational agents for glioblastoma surgery. A survival analysis. J Clin Neurosci 35:82–87. doi:10.1016/j.jocn.2016.10.006
Enlund M, Bergkvist L (2016) Cancer and anaesthesia: survival after radical surgery—a comparison between propofol or sevoflurane anaesthesia (CAN). Avaliable via ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01975064?term=NCT01975064&rank=1. Accessed 28 Aug 2017
Wang D, Zhang Y (2016) Impact of anaesthesia maintenance methods on long-term survival rate. Available via ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT02660411?term=NCT02660411&rank=1. Accessed 28 Aug 2017
Demaria S, Afonso A, Bennett-Guerrero E (2017) General anesthetics in cancer resection surgery (GA-CARES) Trial (GA-CARES). Available via ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT03034096?term=NCT03034096&rank=1. Accessed 28 Aug 2017
Riedel B (2017) Volatile anaesthesia and perioperative outcomes related to cancer (VAPOR-C): a feasibility study. Available via Australian New Zealand Cliical Trials Registry. https://www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=373249&isReview=true. Accessed 28 Aug 2017
Shapiro J, Jersky J, Katzav S, Feldman M, Segal S (1981) Anesthetic drugs accelerate the progression of postoperative metastases of mouse tumors. J Clin Invest 68(3):678
Moudgil G, Singal D (1997) Halothane and isoflurane enhance melanoma tumour metastasis in mice. Can J Anaesth 44(1):90–94. doi:10.1007/BF03014331
Melamed R, Bar-Yosef S, Shakhar G, Shakhar K, Ben-Eliyahu S (2003) Suppression of natural killer cell activity and promotion of tumor metastasis by ketamine, thiopental, and halothane, but not by propofol: mediating mechanisms and prophylactic measures. Anesth Analg 97(5):1331–1339
Mammoto T, Mukai M, Mammoto A, Yamanaka Y, Hayashi Y, Mashimo T, Kishi Y, Nakamura H (2002) Intravenous anesthetic, propofol inhibits invasion of cancer cells. Cancer Lett 184(2):165–170
Hanahan D, Weinberg R (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674. doi:10.1016/j.cell.2011.02.013
Hanahan D, Coussens L (2012) Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 21(3):309–322. doi:10.1016/j.ccr.2012.02.022
Kalluri R, Weinberg R (2009) The basics of epithelial-mesenchymal transition. J Clin Investig 119(6):1420–1428. doi:10.1172/JCI39104
Reymond N, d’Agua B, Ridley A (2013) Crossing the endothelial barrier during metastasis. Nat Rev Cancer 13(12):858–870. doi:10.1038/nrc3628
Chiang S, Cabrera R, Segall J (2016) Tumor cell intravasation. Am J Physiol Cell Physiol 311(1):C1–C14. doi:10.1152/ajpcell.00238.2015
Wu G, Chen W, Sung C, Jean Y, Hung C, Chen F, Hsieh M, Wen Z (2009) Isoflurane attenuates dynorphin-induced cytotoxicity and downregulation of Bcl-2 expression in differentiated neuroblastoma SH-SY5Y cells. Acta Anaesthesiol Scand 53(1):55–60. doi:10.1111/j.1399-6576.2008.01828.x
Kawaraguchi Y, Horikawa Y, Murphy A, Murray F, Miyanohara A, Ali S, Head B, Patel P, Roth D, Patel H (2011) Volatile anesthetics protect cancer cells against tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis via caveolins. J Am Soc Anesthesiol 115(3):499–508. doi:10.1097/ALN.0b013e3182276d42
Ecimovic P, McHugh B, Murray D, Doran P, Buggy D (2013) Effects of sevoflurane on breast cancer cell function in vitro. Anticancer Res 33(10):4255–4260
Huang H, Benzonana L, Zhao H, Watts H, Perry N, Bevan C, Brown R, Ma D (2014) Prostate cancer cell malignancy via modulation of HIF-1α pathway with isoflurane and propofol alone and in combination. Br J Cancer 111(7):1338–1349. doi:10.1038/bjc.2014.426
Zhang D, Zhou X, Zhang J, Zhou Y, Ying J, Wu G, Qian J (2015) Propofol promotes cell apoptosis via inhibiting HOTAIR mediated mTOR pathway in cervical cancer. Biochem Biophys Res Commun 468(4):561–567. doi:10.1016/j.bbrc.2015.10.129
Wu K, Yang S, Hsu S, Chiang J, Hsia T, Yang J, Liu K, Wu R, Chung J (2013) Propofol induces DNA damage in mouse leukemic monocyte macrophage RAW264.7 cells. Oncol Rep 30(5):2304–2310. doi:10.3892/or.2013.2722
Ye Z, Jingzhong L, Yangbo L, Lei C, Jiandong Y (2013) Propofol inhibits proliferation and invasion of osteosarcoma cells by regulation of microRNA-143 expression. Oncol Res Featur Preclin Clin Cancer Ther 21(4):201–207. doi:10.3727/096504014X13890370410203
Wu K, Yang S, Hsia T, Yang J, Chiou S, Lu C, Wu R, Chung J (2012) Suppression of cell invasion and migration by propofol are involved in down-regulating matrix metalloproteinase-2 and p38 MAPK signaling in A549 human lung adenocarcinoma epithelial cells. Anticancer Res 32(11):4833–4842
Deegan C, Murray D, Doran P, Ecimovic P, Moriarty D, Buggy D (2009) Effect of anaesthetic technique on oestrogen receptor-negative breast cancer cell function in vitro. Br J Anaesth 103:685–690. doi:10.1093/bja/aep261
Jaura A, Flood G, Gallagher H, Buggy D (2014) Differential effects of serum from patients administered distinct anaesthetic techniques on apoptosis in breast cancer cells in vitro: a pilot study. Br J Anaesth 113(suppl 1):i63–i67. doi:10.1093/bja/aet581
Saeidnia S, Manayi A, Abdollahi M (2015) From in vitro experiments to in vivo and clinical studies; pros and cons. Curr Drug Discov Technol 12(4):218–224
Mak I, Evaniew N, Ghert M (2014) Lost in translation: animal models and clinical trials in cancer treatment. Am J Transl Res 6(2):114–118
Engquist A, Brandt M, Fernandes A, Kehlet H (1977) The blocking effect of epidural analgesia on the adrenocortical and hyperglycemic responses to surgery. Acta Anaesthesiol Scand 21(4):330–335
Myles P, Peyton P, Silbert B, Hunt J, Rigg J, Sessler D (2011) Perioperative epidural analgesia for major abdominal surgery for cancer and recurrence-free survival: randomised trial. BMJ 342:d1491. doi:10.1136/bmj.d1491
Jang D, Lim C, Shin Y, Ko Y, Park S, Song S, Kim B (2016) A comparison of regional and general anesthesia effects on 5 year survival and cancer recurrence after transurethral resection of the bladder tumor: a retrospective analysis. BMC Anesthesiol 16(1):1. doi:10.1186/s12871-016-0181-6
Kairaluoma P, Mattson J, Heikkila P, Pere P, Leidenius M (2016) Perioperative paravertebral regional anaesthesia and breast cancer recurrence. Anticancer Res 36(1):415–418
Zimmitti G, Soliz J, Aloia T, Gottumukkala V, Cata J, Tzeng C, Vauthey J (2016) Positive impact of epidural analgesia on oncologic outcomes in patients undergoing resection of colorectal liver metastases. Ann Surg Oncol 23(3):1003–1011. doi:10.1245/s10434-015-4933-1
Elias K, Kang S, Liu X, Horowitz N, Berkowitz R, Frendl G (2015) Anesthetic selection and disease-free survival following optimal primary cytoreductive surgery for stage III epithelial ovarian cancer. Ann Surg Oncol 22(4):1341–1348. doi:10.1245/s10434-014-4112-9
Merquiol F, Montelimard A, Nourissat A, Molliex S, Zufferey P (2013) Cervical epidural anesthesia is associated with increased cancer-free survival in laryngeal and hypopharyngeal cancer surgery: a retrospective propensity-matched analysis. Reg Anesth Pain Med 38(5):398–402. doi:10.1097/AAP.0b013e31829cc3fb
Gupta A, Björnsson A, Fredriksson M, Hallböök O, Eintrei C (2011) Reduction in mortality after epidural anaesthesia and analgesia in patients undergoing rectal but not colonic cancer surgery: a retrospective analysis of data from 655 patients in central Sweden. Br J Anaesth 107(2):164–170
Lin L, Liu C, Tan H, Ouyang H, Zhang Y, Zeng W (2011) Anaesthetic technique may affect prognosis for ovarian serous adenocarcinoma: a retrospective analysis. Br J Anaesth 106(6):814–822. doi:10.1093/bja/aer055
Gottschalk A, Brodner G, Van Aken H, Ellger B, Althaus S, Schulze H (2012) Can regional anaesthesia for lymph-node dissection improve the prognosis in malignant melanoma? Br J Anaesth 109(2):253–259. doi:10.1093/bja/aes176
de Oliveira Jr G, Ahmad S, Schink J, Singh D, Fitzgerald P, McCarthy R (2011) Intraoperative neuraxial anesthesia but not postoperative neuraxial analgesia is associated with increased relapse-free survival in ovarian cancer patients after primary cytoreductive surgery. Reg Anesth Pain Med 36(3):271–277. doi:10.1097/AAP.0b013e318217aada
Tsigonis A, Al-Hamadani M, Linebarger J, Vang C, Krause F, Johnson J, Marchese E, Marcou K, Hudak J, Landercasper J (2016) Are cure rates for breast cancer improved by local and regional anesthesia? Reg Anesth Pain Med 41(3):339–347. doi:10.1097/AAP.0000000000000379
Sun Y, Li T, Gan T (2015) The effects of perioperative regional anesthesia and analgesia on cancer recurrence and survival after oncology surgery: a systematic review and meta-analysis. Reg Anesth Pain Med 40(5):589–598. doi:10.1097/AAP.0000000000000273
Sprung J, Scavonetto F, Yeoh T, Kramer J, Karnes R, Eisenach J, Schroeder D, Weingarten T (2014) Outcomes after radical prostatectomy for cancer: a comparison between general anesthesia and epidural anesthesia with fentanyl analgesia: a matched cohort study. Anesth Analg 119(4):859–866. doi:10.1213/ANE.0000000000000320
Roiss M, Schiffmann J, Tennstedt P, Kessler T, Blanc I, Goetz A, Schlomm T, Graefen M, Reuter D (2014) Oncological long-term outcome of 4772 patients with prostate cancer undergoing radical prostatectomy: does the anaesthetic technique matter? Eur J Surg Oncol 40(12):1686–1692. doi:10.1016/j.ejso.2014.02.223
Cata J, Gottumukkala V, Thakar D, Keerty D, Gebhardt R, Liu D (2014) Effects of postoperative epidural analgesia on recurrence-free and overall survival in patients with nonsmall cell lung cancer. J Clin Anesth 26(1):3–17. doi:10.1016/j.jclinane.2013.06.007
Lacassie H, Cartagena J, Brañes J, Assel M, Echevarría G (2013) The relationship between neuraxial anesthesia and advanced ovarian cancer-related outcomes in the Chilean population. Anesth Analg 117(3):653–660. doi:10.1213/ANE.0b013e3182a07046
Wuethrich P, Thalmann G, Studer U, Burkhard F (2013) Epidural analgesia during open radical prostatectomy does not improve long-term cancer-related outcome: a retrospective study in patients with advanced prostate cancer. PloS ONE 8(8):e72873. doi:10.1371/journal.pone.0072873
Ismail H, Ho K, Narayan K, Kondalsamy-Chennakesavan S (2010) Effect of neuraxial anaesthesia on tumour progression in cervical cancer patients treated with brachytherapy: a retrospective cohort study. Br J Anaesth 105(2):145–149. doi:10.1093/bja/aeq156
Cakmakkaya O, Kolodzie K, Apfel C, Pace N (2014) Anaesthetic techniques for risk of malignant tumour recurrence. The Cochrane Library. doi:10.1002/14651858.CD008877
Christopherson R, James K, Tableman M, Marshall P, Johnson F (2008) Long-term survival after colon cancer surgery: a variation associated with choice of anesthesia. Anesth Analg 107(1):325–332. doi:10.1213/ane.0b013e3181770f55
Binczak M, Tournay E, Billard V, Rey A, Jayr C (2013) Major abdominal surgery for cancer: does epidural analgesia have a long-term effect on recurrence-free and overall survival? Annales francaises d’anesthesie et de reanimation 32(5):e81–e88. doi:10.1016/j.annfar.2013.02.027
Tsui B, Rashiq S, Schopflocher D, Murtha A, Broemling S, Pillay J, Finucane B (2010) Epidural anaesthesia and cancer recurrence rates after radical prostatectomy. Can J Anaesth 57(2):107–112. doi:10.1007/s12630-009-9214-7
Hiller J, Ismail H, Riedel B (2014) Improved quality of anaesthesia and cancer recurrence studies. Anesth Analg 119(3):751–752. doi:10.1213/ane.0000000000000290
Wada H, Seki S, Takahashi T, Kawarabayashi N, Higuchi H, Habu Y, Sugahara S, Kazama T (2007) Combined spinal and general anesthesia attenuates liver metastasis by preserving TH1/TH2 cytokine balance. J Am Soc Anesthesiol 106(3):499–506
Bar-Yosef S, Melamed R, Page G, Shakhar G, Shakhar K, Ben-Eliyahu S (2001) Attenuation of the tumor-promoting effect of surgery by spinal blockade in rats. J Am Soc Anesthesiol 94(6):1066–1073
Brackenbury W (2012) Voltage-gated sodium channels and metastatic disease. Channels 6(5):352–361. doi:10.4161/chan.21910
Roger S, Guennec J, Besson P (2004) Particular sensitivity to calcium channel blockers of the fast inward voltage-dependent sodium current involved in the invasive properties of a metastastic breast cancer cell line. Br J Pharmacol 141(4):610–615. doi:10.1038/sj.bjp.0705649
Roger S, Rollin J, Barascu A, Besson P, Raynal P, Iochmann S, Lei M, Bougnoux P, Gruel Y, Le Guennec J (2007) Voltage-gated sodium channels potentiate the invasive capacities of human non-small-cell lung cancer cell lines. Int J Biochem Cell Biol 39(4):774–786. doi:10.1016/j.biocel.2006.12.007
Lirk P, Hollmann M, Fleischer M, Weber N, Fiegl H (2014) Lidocaine and ropivacaine, but not bupivacaine, demethylate deoxyribonucleic acid in breast cancer cells in vitro. Br J Anaesth 113(suppl 1):i32–i38. doi:10.1093/bja/aeu201
Lirk P, Berger R, Hollmann M, Fiegl H (2012) Lidocaine time-and dose-dependently demethylates deoxyribonucleic acid in breast cancer cell lines in vitro. Br J Anaesth 109(2):200–207. doi:10.1093/bja/aes128
Li K, Yang J, Han X (2014) Lidocaine sensitizes the cytotoxicity of cisplatin in breast cancer cells via up-regulation of RARβ2 and RASSF1A demethylation. Int J Mol Sci 15(12):23519–23536. doi:10.3390/ijms151223519
Lazo J, Braun I, Meandžija B, Kennedy K, Pham E, Smaldone L (1985) Lidocaine potentiation of bleomycin A2 cytotoxicity and DNA strand breakage in L1210 and human A-253 cells. Cancer Res 45(5):2103–2109
Mizuno S, Ishida A (1982) Selective enhancement of the cytotoxicity of the bleomycin derivative, peoplomycin, by local anesthetics alone and combined with hyperthermia. Cancer Res 42(11):4726–4729
Chlebowski R, Block J, Cundiff D, Dietrich M (1982) Doxorubicin cytotoxicity enhanced by local anesthetics in a human melanoma cell line. Cancer Treat Rep 66(1):121–125
Robins H, Dennis W, Slattery J, Lange T, Yatvin M (1983) Systemic lidocaine enhancement of hyperthermia-induced tumor regression in transplantable murine tumor models. Cancer Res 43(7):3187–3191
Yatvin M, Clifton K, Dennis W (1979) Hyperthermia and local anesthetics: potentiation of survival of tumor-bearing mice. Science 205(4402):195–196
Piegeler T, Votta-Velis E, Liu G, Place A, Schwartz D, Beck-Schimmer B, Minshall R, Borgeat A (2012) Antimetastatic potential of amide-linked local anesthetics inhibition of lung adenocarcinoma cell migration and inflammatory src signaling independent of sodium channel blockade. J Am Soc Anesthesiol 117(3):548–559. doi:10.1097/ALN.0b013e3182661977
Lennon F, Mirzapoiazova T, Mambetsariev B, Poroyko V, Salgia R, Moss J, Singleton P (2014) The mu opioid receptor promotes opioid and growth factor-induced proliferation, migration and epithelial mesenchymal transition (EMT) in human lung cancer. PLoS ONE 9(3):e91577. doi:10.1371/journal.pone.0091577
Karniel M, Beitner R (2000) Local anesthetics induce a decrease in the levels of glucose 1, 6-bisphosphate, fructose 1, 6-bisphosphate, and ATP, and in the viability of melanoma cells. Mol Genet Metab 69(1):40–45. doi:10.1006/mgme.1999.2954
Martin O, Anderson R, Narayan K, MacManus M (2016) Does the mobilization of circulating tumour cells during cancer therapy cause metastasis? Nat Rev Clin Oncol (Adv Online Publ). doi:10.1038/nrclinonc.2016.128
Hardingham J, Grover P, Winter M, Hewett P, Price T, Thierry B (2015) Detection and clinical significance of circulating tumor cells in colorectal cancer—20 years of progress. Mol Med 21(Suppl 1):S25–S31. doi:10.2119/molmed.2015.00149
Yuki K, Eckenhoff R (2016) Mechanisms of the immunological effects of volatile anesthetics: a review. Anesth Analg 123(2):326–335. doi:10.1213/ANE.0000000000001403
Woo J, Baik H, Kim C, Chung R, Kim D, Lee G, Chun E (2015) Effect of propofol and desflurane on immune cell populations in breast cancer patients: a randomized trial. J Korean Med Sci 30(10):1503–1508. doi:10.3346/jkms.2015.30.10.1503
Buckley A, McQuaid S, Johnson P, Buggy D (2014) Effect of anaesthetic technique on the natural killer cell anti-tumour activity of serum from women undergoing breast cancer surgery: a pilot study. Br J Anaesth 113(Suppl 1):i56–i62. doi:10.1093/bja/aeu200
Desmond F, Mccormack J, Mulligan N, Stokes M, Buggy D (2015) Effect of anaesthetic technique on immune cell infiltration in breast cancer: a follow-up pilot analysis of a prospective, randomised, investigator-masked study. Anticancer Res 35(3):1311–1319
Ren X, Li W, Meng F, Lin C (2010) Differential effects of propofol and isoflurane on the activation of T-helper cells in lung cancer patients. Anaesthesia 65(5):478–482. doi:10.1111/j.1365-2044.2010.06304
Tsui B, Green J (2011) Type of anaesthesia during cancer surgery and cancer recurrence. BMJ 342:d1605. doi:10.1136/bmj.d1605
Inada T, Kubo K, Ueshima H, Shingu K (2011) Intravenous anesthetic propofol suppresses prostaglandin E2 production in murine dendritic cells. J Immunotoxicol 8(4):359–366. doi:10.3109/1547691x.2011.620036
Elena G, Amerio N, Ferrero P, Bay M, Valenti J, Colucci D, Puig N (2003) Effects of repetitive sevoflurane anaesthesia on immune response, select biochemical parameters and organ histology in mice. Lab Anim 37(3):193–203. doi:10.1258/002367703766453038
Ben-Eliyahu S, Shakhar G, Rosenne E, Levinson Y, Beilin B (1999) Hypothermia in barbiturate-anesthetized rats suppresses natural killer cell activity and compromises resistance to tumor metastasis a role for adrenergic mechanisms. J Am Soc Anesthesiol 91(3):732–740
Kalimeris K, Christodoulaki K, Karakitsos P, Batistatou A, Lekka M, Bai M, Kitsiouli E, Nakos G, Kostopanagiotou G (2011) Influence of propofol and volatile anaesthetics on the inflammatory response in the ventilated lung. Acta Anaesthesiol Scand 55(6):740–748. doi:10.1111/j.1399-6576.2011.02461.x
Kostopanagiotou G, Kalimeris K, Christodoulaki K, Nastos C, Papoutsidakis N, Dima C, Chrelias C, Pandazi A, Mourouzis I, Pantos C (2010) The differential impact of volatile and intravenous anaesthetics on stress response in the swine. Hormones 9(1):67–75
Miyata T, Kodama T, Honma R, Nezu Y, Harada Y, Yogo T, Hara Y, Tagawa M (2013) Influence of general anesthesia with isoflurane following propofol-induction on natural killer cell cytotoxic activities of peripheral blood lymphocytes in dogs. J Vet Med Sci 75(7):917–921
Stollings L, Jia L, Tang P, Dou H, Lu B, Xu Y (2016) Immune modulation by volatile anesthetics. Anesthesiology. doi:10.1097/ALN.0000000000001195
Chen R, Chen T, Chen T, Lin L, Chang C, Chang H, Wu C (2005) Anti-inflammatory and antioxidative effects of propofol on lipopolysaccharide-activated macrophages. Ann N Y Acad Sci 1042:262–271. doi:10.1196/annals.1338.030
Yuki K, Soriano S, Shimaoka M (2011) Sedative drug modulates T-cell and lymphocyte function-associated antigen-1 function. Anesth Analg 112(4):830. doi:10.1213/ANE.0000000000001403
Desborough J (2000) The stress response to trauma and surgery. Br J Anaesth 85(1):109–117
Phillips S (2000) Physiology of wound healing and surgical wound care. ASAIO J 46(6):S2–S5
Neeman E, Zmora O, Ben-Eliyahu S (2012) A new approach to reducing postsurgical cancer recurrence: perioperative targeting of catecholamines and prostaglandins. Clin Cancer Res 18(18):4895–4902. doi:10.1158/1078-0432.ccr-12-1087
DeKeyser F, Leker R, Weidenfeld J (2000) Activation of the adrenocortical axis by surgical stress: involvement of central norepinephrine and interleukin-1. Neuroimmunomodulation 7(4):182–188
Naito Y, Tamai S, Shingu K, Shindo K, Matsui T, Segawa H, Nakai Y, Mori K (1992) Responses of plasma adrenocorticotropic hormone, cortisol, and cytokines during and after upper abdominal surgery. Anesthesiology 77(3):426–431
Kruimel J, Pesman GJ, Sweep C, van der Vliet J, Liem T, Jansen J, van der Meer J, Naber A (1999) Depression of plasma levels of cytokines and ex-vivo cytokine production in relation to the activity of the pituitary-adrenal axis, in patients undergoing major vascular surgery. Cytokine 11(5):382–388. doi:10.1006/cyto.1999.0440
Roth-Isigkeit A, Schmucker P (1997) Postoperative dissociation of blood levels of cortisol and adrenocorticotropin after coronary artery bypass grafting surgery. Steroids 62(11):695–699
Coutinho A, Chapman K (2011) The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol Cell Endocrinol 335(1):2–13. doi:10.1016/j.mce.2010.04.005
Vukelic S, Stojadinovic O, Pastar I, Rabach M, Krzyzanowska A, Lebrun E, Davis S, Resnik S, Brem H, Tomic-Canic M (2011) Cortisol synthesis in epidermis is induced by IL-1 and tissue injury. J Biol Chem 286(12):10265–10275. doi:10.1074/jbc.M110.188268
Brochner A, Toft P (2009) Pathophysiology of the systemic inflammatory response after major accidental trauma. Scand J Trauma Resusc Emerg Med 17:43. doi:10.1186/1757-7241-17-43
Palucka A, Coussens L (2016) The basis of oncoimmunology. Cell 164(6):1233–1247. doi:10.1016/j.cell.2016.01.049
Gaudilliere B, Fragiadakis G, Bruggner R, Nicolau M, Finck R, Tingle M, Silva J, Ganio E, Yeh C, Maloney W, Huddleston J, Goodman S, Davis M, Bendall S, Fantl W, Angst M, Nolan G (2014) Clinical recovery from surgery correlates with single-cell immune signatures. Sci Transl Med 6(255):255ra131. doi:10.1126/scitranslmed.3009701
Tsimogiannis K, Tellis C, Tselepis A, Pappas-Gogos G, Tsimoyiannis E, Basdanis G (2012) Toll-like receptors in the inflammatory response during open and laparoscopic colectomy for colorectal cancer. Surg Endosc 26(2):330–336. doi:10.1007/s00464-011-1871-2
Veenhof A, Sietses C, Von Blomberg B, Van Hoogstraten I, Vd Pas M, Meijerink W, vd Peet D, Vd Tol M, Bonjer H, Cuesta M (2011) The surgical stress response and postoperative immune function after laparoscopic or conventional total mesorectal excision in rectal cancer: a randomized trial. Int J Colorectal Dis 26(1):53–59
Ogawa K, Hirai M, Katsube T, Murayama M, Hamaguchi K, Shimakawa T, Naritake Y, Hosokawa T, Kajiwara T (2000) Suppression of cellular immunity by surgical stress. Surgery 127(3):329–336
Ogawa K, Hirai M, Katsube T, Murayama M, Hamaguchi K, Shimakawa T, Naritake Y, Hosokawa T, Kajiwara T (2000) Suppression of cellular immunity by surgical stress. Surgery 127(3):329–336. doi:10.1067/msy.2000.103498
Kondo E, Koda K, Takiguchi N, Oda K, Seike K, Ishizuka M, Miyazaki M (2003) Preoperative natural killer cell activity as a prognostic factor for distant metastasis following surgery for colon cancer. Dig Surg 20(5):445–451
Tartter P, Steinberg B, Barron D, Martinelli G (1987) The prognostic significance of natural killer cytotoxicity in patients with colorectal cancer. Arch Surg 122(11):1264–1268
Zhou L, Li Y, Li X, Chen G, Liang H, Wu Y, Tong J, Ouyang W (2016) Propranolol attenuates surgical stress-induced elevation of the regulatory T cell response in patients undergoing radical mastectomy. J Immunol 196(8):3460–3469. doi:10.4049/jimmunol.1501677
Ma W, Wang K, Du J, Luan J, Lou G (2015) Multi-dose parecoxib provides an immunoprotective effect by balancing T helper 1. Th2, Th17 and regulatory T cytokines following laparoscopy in patients with cervical cancer. Mol Med Rep 11(4)(Th1):2999–3008. doi:10.3892/mmr.2014.3003
Shaashua L, Shabat-Simon M, Haldar R, Matzner P, Zmora O, Shabtai M, Sharon E, Allweis T, Barshack I, Hayman L, Arevalo J, Ma J, Horowitz M, Cole S, Ben-Eliyahu S (2017) Perioperative COX-2 and β-adrenergic blockade improves metastatic biomarkers in breast cancer patients in a phase-II randomized trial. Clin Cancer Res. doi:10.1158/1078-0432.ccr-17-0152
Lacy A, Garcia-Valdecasas J, Delgado S, Castells A, Taura P, Pique J, Visa J (2002) Laparoscopy-assisted colectomy versus open colectomy for treatment of non-metastatic colon cancer: a randomised trial. The Lancet 359(9325):2224–2229. doi:10.1016/s0140-6736(02)09290-5
Mirnezami A, Mirnezami R, Chandrakumaran K, Sasapu K, Sagar P, Finan P (2011) Increased local recurrence and reduced survival from colorectal cancer following anastomotic leak: systematic review and meta-analysis. Ann Surg 253(5):890–899. doi:10.1097/SLA.0b013e3182128929
Tsuchiya Y, Sawada S, Yoshioka I, Ohashi Y, Matsuo M, Harimaya Y, Tsukada K, Saiki I (2003) Increased surgical stress promotes tumor metastasis. Surgery 133(5):547–555. doi:10.1067/msy.2003.141
Shiromizu A, Suematsu T, Yamaguchi K, Shiraishi N, Adachi Y, Kitano S (2000) Effect of laparotomy and laparoscopy on the establishment of lung metastasis in a murine model. Surgery 128(5):799–805. doi:10.1067/msy.2000.108047
Sorski L, Levi B, Shaashua L, Neeman E, Benish M, Matzner P, Hoffman A, Ben-Eliyahu S (2014) The impact of surgical extent and sex on the hepatic metastasis of colon cancer. Surg Today 44(10):1925–1934. doi:10.1007/s00595-013-0768-1
Ben-Eliyahu S, Page G, Yirmiya R, Shakhar G (1999) Evidence that stress and surgical interventions promote tumor development by suppressing natural killer cell activity. Int J Cancer 80(6):880–888
Tohme S, Yazdani H, Al-Khafaji A, Chidi A, Loughran P, Mowen K, Wang Y, Simmons R, Huang H, Tsung A (2016) Neutrophil extracellular traps promote the development and progression of liver metastases after surgical stress. Cancer Res 76(6):1367–1380. doi:10.1158/0008-5472.can-15-1591
Glasner A, Avraham R, Rosenne E, Benish M, Zmora O, Shemer S, Meiboom H, Ben-Eliyahu S (2010) Improving survival rates in two models of spontaneous postoperative metastasis in mice by combined administration of a beta-adrenergic antagonist and a cyclooxygenase-2 inhibitor. J Immunol 184(5):2449–2457. doi:10.4049/jimmunol.0903301
Matzner P, Sorski L, Shaashua L, Elbaz E, Lavon H, Melamed R, Rosenne E, Gotlieb N, Benbenishty A, Reed S, Ben-Eliyahu S (2016) Perioperative treatment with the new synthetic TLR-4 agonist GLA-SE reduces cancer metastasis without adverse effects. Int J Cancer 138(7):1754–1764. doi:10.1002/ijc.29885
Seth R, Tai L, Falls T, de Souza C, Bell J, Carrier M, Atkins H, Boushey R, Auer R (2013) Surgical stress promotes the development of cancer metastases by a coagulation-dependent mechanism involving natural killer cells in a murine model. Ann Surg 258(1):158–168. doi:10.1097/SLA.0b013e31826fcbdb
Wrobel L, Bod L, Lengagne R, Kato M, Prevost-Blondel A, Le Gal F (2016) Propanolol induces a favourable shift of anti-tumour immunity in a murine spontaneous model melanoma. Oncotarget 7:77825–77837. doi:10.18632/oncotarget.12833
Gargiulo S, Greco A, Gramanzini M, Esposito S, Affuso A, Brunetti A, Vesce G (2012) Mice anesthesia, analgesia, and care, part I: anesthetic considerations in preclinical research. ILAR J 53(1). doi:10.1093/ilar.53.1.55
Lipsky M, Sharp L (2001) From idea to market: the drug approval process. Journal Am Board Fam Pract 14(5):362–367
Fleming E, Perkins J, Easa D, Conde J, Baker R, Southerland W, Dottin R, Benabe J, Ofili E, Bond V, McClure S, Sayre M, Beanan M, Norris K (2008) The role of translational research in addressing health disparities: a conceptual framework. Ethn Dis 18(2 Suppl 2):S2-155–S2-160
Acknowledgements
This work was supported by the David and Lorelle Skewes Foundation, the Peter Mac Foundation, the Australian and New Zealand College of Anaesthetists and the National Cancer Institute (Grant No. CA160890).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dubowitz, J.A., Sloan, E.K. & Riedel, B.J. Implicating anaesthesia and the perioperative period in cancer recurrence and metastasis. Clin Exp Metastasis 35, 347–358 (2018). https://doi.org/10.1007/s10585-017-9862-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-017-9862-x