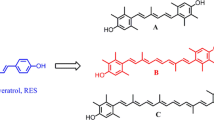
Using 2-thiophenecarbonyl chloride and 2-furoyl chloride to modify the structure of the natural product resveratrol, we synthesized five novel resveratrol derivatives. The target compounds were evaluated for their antitumor activities against cervical cancer HeLa cells by the MTT method. The results indicated that the compound 3a displayed the best antitumor activities, which is higher than the value of resveratrol, and its inhibition ratio was 95.1% against HeLa cells at the concentration of 75 μmol/L.
Similar content being viewed by others
References
P. Min-hsiung, L. Chih-li, T. Jie-heng, H. Chi-tang, and C. Weijen, J. Agric. Food Chem., 58, 226 (2010).
S. Stalin, M. Aarti, N. Shridhar, S. Swaminathan, and M. K. Uma, J. Med. Chem., 56, 970 (2013).
C. U. Chibuike, E. A. Rotimi, and J. H. J. Peter, Nutr. Rev., 66, 445 (2008).
M. Laurent, D. Nicolas, and S. Florence, Biofactors, 36, 377 (2010).
Y. Suwan, Q. Chengxue, and L. W. Owen, Biofactors, 36, 350 (2010).
M. Amarnath, R. Y. Deepthi, K. Chandan, J. R. Vangala, P. R. Surendra, A. Anthony, V. K. Shasi, and S. Bojja J. Med. Chem., 54, 6751 (2011).
Y. J. Lv, N. Zhou, and Q. G. Meng, J. Int. Pharm. Res., 35, 377 (2008).
Z. Xuejing, Z. Jie, X. Xiaoyun, Z. Yong, and L. Huizhen, J. Chin. Pharm. Sci., 13, 10 (2004).
F. Huiting, X. Xiaoyun, C. Wei, L. Xiaoqiang, Z. Yong, Z. Xuejing, and M. Qibing, Chin. J. New Drugs, 15, 874 (2006). 10. L. Huachen, D. Aijun, G. Chunmei, and J. Yuyang, Prog. Chem., 21, 1500 (2009).
Acknowledgment
This work was financially supported by the Innovation Program of Shanghai Municipal Education Commission (No. 12ZZ188) and the Program for Professor of Special Appointment (Eastern Scholar) at Shanghai Institutions of Higher Learning (405ZK110060002).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 4, July–August, 2015, pp. 563–565.
Rights and permissions
About this article
Cite this article
Jin, L., Ren, YJ. & Du, C. Synthesis and Antitumor Activities of Resveratrol Derivatives on Cervical Cancer Hela Cells. Chem Nat Compd 51, 652–655 (2015). https://doi.org/10.1007/s10600-015-1377-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-015-1377-6