Abstract
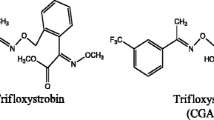
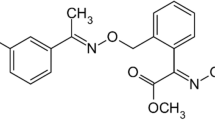
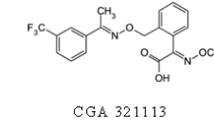
The dissipation of trifloxystrobin and its metabolite trifloxystrobin acid in apples and soil was studied, and the half-life (DT50) was estimated in a field study carried out at three different locations for apples and four different locations for soil. Trifloxystrobin was sprayed on apples at 127 g a.i./ha for the dissipation study. Samples of apple and soil for the dissipation experiment were collected at time intervals of 0, 1, 3, 7, 14, 21, 30, and 45 days after treatment. The quantification of residues was done by liquid chromatography-tandem mass spectrometry (LC-MS/MS). The DT50 of trifloxystrobin ranged from 0.54 to 8.8 and 4.8 to 9.5 days in soil and apples at different latitude sites. Photolysis may be the main dissipation pathway for trifloxystrobin, and the number of sunshine hours may be the main factor affecting the trifloxystrobin dissipation rate in the field. For trifloxystrobin acid residues in soil and apples, it first increased and then began decreasing. It was indicated that the risk of trifloxystrobin application in shorter sunshine hour area should be considered.



Similar content being viewed by others
References
Anke, T., Oberwinkler, F., Steglich, W., & Schramm, G. (1977). The strobilurins—new antifungal antibiotics from the basidiomycete Strobilurus tenacellus (Pers. ex Fr.) Sing. Journal of Antibiotics, 30, 806–810.
Balba, H. (2007). Review of strobilurin fungicide chemicals. Journal of Environmental Science and Health, Part B Pesticides, 42, 441–451.
Banerjee, K., Ligon, A. P., & Spiteller, M. (2006). Environmental fate of trifloxystrobin in soils of different geographical origins and photolytic degradation in water. Journal of Agricultural and Food Chemistry, 54, 9479–9487.
Banerjee, K., Ligon, A. P., & Spiteller, M. (2007). Spectral elucidation of the acid metabolites of the four geometric isomers of trifloxystrobin. Analytical and Bioanalytical Chemistry, 388, 1831–1838.
Bartlett, D. W., Clough, J. M., Godwin, J. R., Hall, A. A., Hamer, M., & Parr-Dobrzanski, B. (2002). The strobilurine fungicides. Pest Management Science, 58(7), 649–662.
Boudina, A., Emmelin, C., Baaliouamer, A., Paisse, O., & Chovelon, J. M. (2007). Photochemical transformation of azoxystrobin in aqueous solutions. Chemosphere, 68, 1280–1288.
DG SANCO. (2014). EU pesticides database. http://ec.europa.eu/sanco_pesticides/public/index.cfm. Accessed 15 Aug 2012.
FAO, & WHO. (2013). Codex pesticides residues in food online database. http://www.codexalimentarius.net/pestres/data/index.html. Accessed 15 Aug 2014.
Food Standards Australia New Zealand. (2014). Food standards code. http://www.foodstandards.gov.au/code/Pages/default.aspx. Accessed 15 Aug 2014.
Garau, V. L., Angioni, A., Del Real, A. A., Russo, M., & Cabras, P. (2002). Disappearance of azoxystrobin, pyrimethanil, cyprodinil, and fludioxonil on tomatoes in a greenhouse. Journal of Agricultural and Food Chemistry, 50, 1929–1932.
Ghosh, R. K., & Singh, N. (2009a). Effect of organic manure on sorption and degradation of azoxystrobin in soil. Journal of Agricultural and Food Chemistry, 57, 632–636.
Ghosh, R. K., & Singh, N. (2009b). Leaching behaviour of azoxystrobin and metabolites in soil columns. Pest Management Science, 65, 1009–1014.
Health Canada. (2012). Maximum residue limits for pesticides. http://pr-rp.hc-sc.gc.ca/mrl-lrm/index-eng.php. Accessed 15 Aug 2014.
Isenring, H. P., & Weiss, B. (1991). Preparation of [(iminooxy)tolyl]acrylates and analogs as fungicides. Europe Patent EP0460575.
Japan Food Chemical Research Foundation. (2014). The Japanese positive list system for agricultural chemical residues in foods. http://www.ffcr.or.jp/zaidan/FFCRHOME.nsf/pages/MRLs-p. Accessed 15 Aug 2014.
Joint FAO/WHO Meeting On Pesticide Residues. (2004). JMPR evaluation: trifloxystrobin. http://www.fao.org/fileadmin/templates/agphome/documents/Pests_Pesticides/JMPR/Evaluation04/TRIFLOXYSTROBIN.pdf. Accessed 27 Dec 2013.
Myung, K., Williams, D. A., Xiong, Q. B., & Thornburgh, S. (2013). Metabolism of strobilurins by wheat cell suspension cultures. Journal of Agricultural and Food Chemistry, 61, 47–52.
Patyal, S. K., Sharma, I. D., Chandel, R. S., & Dubey, J. K. (2013). Dissipation kinetics of trifloxystrobin and tebuconazole on apple (Malus domestica) and soil—a multi location study from north western Himalayan region. Chemosphere, 92, 949–954.
Singh, N., Singh, S. B., Mukerjee, I., Gupta, S., Gajbhiye, V. T., Sharma, P. K., Goel, M., & Dureja, P. (2010). Metabolism of 14C-azoxystrobin in water at different pH. Journal of Environmental Science and Health, Part B Pesticides, 45, 123–127.
Tomlin, C.D.S. (2006). The pesticide manual: a world compendium. Alton, Hampshire, UK: British Crop Protection Council 1349
US Government Printing Office. (2014). Tolerances and exemptions for pesticide chemical residues in food. http://www.ecfr.gov/cgi-bin/text-idx?c=ecfr&sid=5d35d354cc838eb105a733f5dff13ab8&tpl=/ecfrbrowse/Title40/40cfr180_main_02.tpl. Accessed 15 Aug 2014.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, C., Wu, J., Zhang, Y. et al. Field dissipation of trifloxystrobin and its metabolite trifloxystrobin acid in soil and apples. Environ Monit Assess 187, 4100 (2015). https://doi.org/10.1007/s10661-014-4100-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-014-4100-3