Abstract
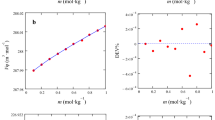
Partial molar volumes (\(V_\phi^0\)) and viscosity B-coefficients of sodium molybdate in 1,3-dioxolane + water mixtures have been determined from solution density and viscosity measurements at 303.15, 313.15, and 323.15 K and at various electrolyte concentrations. Also, the adiabatic compressibility of different solutions has been determined from the measurement of sound speeds at 303.15 K. The experimental density data were evaluated by the Masson equation, and the derived parameters were interpreted in terms of ion–solvent and ion–ion interactions. The viscosity data have been analyzed using the Jones–Dole equation, and the derived parameters, B and A, have also been interpreted in terms of ion–solvent and ion–ion interactions, respectively. The structure- making or breaking capacity of the electrolyte under investigation has been discussed in terms of the sign of \((\delta ^{2}V_\phi^0/\delta T^{2})_{P}\) . The compressibility data obtained from sound speeds of different solutions indicate the electrostriction of the solvent molecules around the ions.
Similar content being viewed by others
References
McDowali J.M., Vincent C.A. (1974). J. Chem. Soc. Faraday Trans. 1: 1862
Deck M.R.J., Bird K.J., Parker A.J. (1975) Aust. J. Chem. 28: 955
Roy M.N., Sinha B., Dey R., Sinha A. (2005) . Int. J. Thermophys. 26: 1549
Janz C.G., Tomkins R.P.T. (1973). Non-aqueous Electrolytes Handbook, vol. 2 Academic Press, New York
Jasinki R. (1967). High Energy Batteries. Plenum Press, New York
Roy M.N., Sinha A. (2006). Fluid Phase Equilib. 243: 133
Roy M.N., Sinha A., Sinha B. (2005). J. Solution Chem. 34: 1311
Gascon I., Mainar A.M., Royo F.M., Urieta J.S. (2000). J. Chem. Eng. Data 45: 751
Tamura K., Ohomura K., Murakami S. (1983). J. Chem. Thermodyn. 15: 859
Roy M.N., Hazra D.K. (1994). Indian J. Chem. Technol. 1: 93
Roy M.N., Sinha B., Dakua V.K. (2006). J. Chem. Eng. Data 51: 590
Peralta R.D., Infante R., Cortez G., Cadenas G., Wisniak J. (2003) . Int. J. Thermophys. 24: 1061
Shoemaker D.P., Garland C.W.S. (1967). Experiments in Physical Chemistry McGraw Hill, New York, p. 131
Masson D.O. (1929) . Philos. Mag. 8: 218
Parmar M.L., Banyal D.S. (2005) . Indian J. Chem. 44A: 1582
Millero F.J. (1972). Structure and Transport Process in Water and Aqueous Solutions. R.A. Horne, New York
Hepler L.G. (1969). Can. J. Chem. 47: 4617
Millero F.J. (1971). Chem. Rev. 71: 147
Jones G., Dole M. (1929). J. Am. Chem. Soc. 51: 2950
Millero F.J., Losurdo F.J., Shin C. (1978) . J. Phys. Chem. 82: 784
Gopal R., Siddique M.A. (1969). J. Phys. Chem. 72: 1814
Saha N., Das B. (1997). J. Chem. Eng. Data 42: 277
Martin Contreras S. (2001). J. Chem. Eng. Data 46: 1149
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Roy, M.N., Dakua, V.K. & Sinha, B. Partial Molar Volumes, Viscosity B-Coefficients, and Adiabatic Compressibilities of Sodium Molybdate in Aqueous 1,3-Dioxolane Mixtures from 303.15 to 323.15 K. Int J Thermophys 28, 1275–1284 (2007). https://doi.org/10.1007/s10765-007-0220-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10765-007-0220-0