Abstract
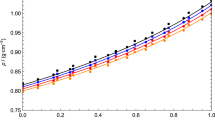
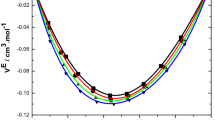
Densities, viscosities, and refractive indices of binary mixtures of n-butyl acetate (1) +1-chlorobutane (2), +1-chloropentane (2), +1-chlorohexane (2), +1-chloroheptane (2), and +1-chlorooctane (2) were measured at 298.15 K for the liquid region and at ambient pressure for the whole composition range. The excess molar volumes V E were calculated from experimental densities. McAllister’s three-body interaction, and Hind and Grunberg–Nissan models are used for correlating the viscosity of binary mixtures. The experimental data of binaries are analyzed to discuss the nature and strength of intermolecular interactions in these mixtures.
Similar content being viewed by others
References
Iloukhani H., Rezaei-Sameti M.: Monatsh Chem. 140, 451 (2009)
Iloukhani H., Parsa J.B., Hatami M.: Phys. Chem. Liq. 46, 495 (2008)
Iloukhani H., Samiey B.: J. Chem. Thermodyn. 39, 206 (2007)
Iloukhani H., Khanlarzadeh K.: J. Chem. Eng. Data 51, 1226 (2006)
Iloukhani H., Rezaei-Sameti M., Parsa J.B.: J. Chem. Thermodyn. 38, 975 (2006)
McAllister R.A.: AIChE J. 6, 42 (1960)
Hind R.K., McLaughlin E., Ubbelohde A.R.: Trans. Faraday. Soc. 56, 328 (1960)
Grunberg L., Nissan A.H.: Nature 164, 799 (1949)
Riddick J.A., Bunger W.B., Sakano T.: Organic Solvents, Physical Properties and Methods of Purification, 4th edn. Wiley, New York (1986)
Moravkova L., Link L.J.: J. Chem. Thermodyn. 34, 1407 (2002)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iloukhani, H., Khanlarzadeh, K. & Rakhshi, M. Densities, Excess Molar Volumes, Viscosities, and Refractive Indices of Binary Mixtures of n-Butyl Acetate with 1-Chloroalkanes (C4–C8) at 298.15 K. Int J Thermophys 32, 614–621 (2011). https://doi.org/10.1007/s10765-010-0902-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10765-010-0902-x