Abstract
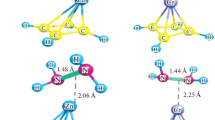
In this study, we explored the reversible hydrogen storage capacity of Ni functionalized C10 carbyne complex through density functional theory (DFT) and molecular dynamic (MD) calculations. ωB97X-D3/def2-TZVP and DLPNO-CCSD(T)/def2-TZVPP methods are used for the estimation of adsorption energies. NiC10 complex is observed to be more sensitive toward hydrogen adsorption compared to isolated C10 carbyne. The nH2-NiC10 complexes are stable when n ≤ 5, and adsorption energies are in the range of − 0.89 to − 0.22 eV/H2 molecule while the hydrogen storage capacity is about 1.11 to 5.33 wt% for hydrogen molecule. For desorption of H2, molecular dynamic calculations are performed at ωB97X-D3 with def2-TZVP O using RCA ABMD package in which the complexes showed stability to desorption up to 2000 steps. This study illustrates the potential of nickel-doped carbyne C10 complex for the storage of hydrogen and applications in fuel cells.










Similar content being viewed by others
References
M. Aneke, M. Wang, Energy storage technologies and real life applications—a state of the art review. Appl. Energy 179, 350–377 (2016). https://doi.org/10.1016/j.apenergy.2016.06.097
S.P.S. Badwal, S. Giddey, C. Munnings, Emerging technologies, markets and commercialization of solid-electrolytic hydrogen production. WIREs Energy Environ. (2018). https://doi.org/10.1002/wene.286
Y. Wu, L. Zhang, Can the development of electric vehicles reduce the emission of air pollutants and greenhouse gases in developing countries? Transp. Res. Part D 51, 129–145 (2017). https://doi.org/10.1016/j.trd.2016.12.007
C. Acar, I. Dincer, The potential role of hydrogen as a sustainable transportation fuel to combat global warming. Int. J. Hydrogen Energy 45(5), 3396–3406 (2020). https://doi.org/10.1016/j.ijhydene.2018.10.149
W.C. Nadaleti, G. Przybyla, Emissions and performance of a spark-ignition gas engine generator operating with hydrogen-rich syngas, methane and biogas blends for application in southern Brazilian rice industries. Energy 154, 38–51 (2018). https://doi.org/10.1016/j.energy.2018.04.046
B. Paul, J. Andrews, PEM unitised reversible/regenerative hydrogen fuel cell systems: state of the art and technical challenges. Renew. Sustain. Energy Rev. 79, 585–599 (2017). https://doi.org/10.1016/j.rser.2017.05.112
A. Gong, D. Verstraete, Fuel cell propulsion in small fixed-wing unmanned aerial vehicles: current status and research needs. Int. J. Hydrogen Energy 42(33), 21311–21333 (2017). https://doi.org/10.1016/j.ijhydene.2017.06.148
F. Dawood, M. Anda, G.M. Shafiullah, Hydrogen production for energy: an overview. Int. J. Hydrogen Energy 45(7), 3847–3869 (2020). https://doi.org/10.1016/j.ijhydene.2019.12.059
S. Singh, S. Jain, A.K. Tiwari, M.R. Nouni, J.K. Pandey, S. Goel, Hydrogen: a sustainable fuel for future of the transport sector. Renew. Sustain. Energy Rev. 51, 623–633 (2015). https://doi.org/10.1016/j.rser.2015.06.040
J. Fischman, P. Godart, D. Hart, Hydrogen generation via the reaction of an activated aluminum slurry with water. Int. J. Hydrogen Energy 45(35), 17118–17130 (2020). https://doi.org/10.1016/j.ijhydene.2020.04.161
K. O’Malley, G. Ordaz, J. Adams, K. Randolph, C.C. Ahn, N.T. Stetson, Applied hydrogen storage research and development: a perspective from the U.S. Department of Energy. J. Alloys Compd. 645, S419–S422 (2015). https://doi.org/10.1016/j.jallcom.2014.12.090
F.J. García de Abajo, Graphene plasmonics: challenges and opportunities. ACS Photon. 1(3), 135–152 (2014). https://doi.org/10.1021/ph400147y
S. Chen, L. Liu, X. Gao, Y. Hua, L. Peng, Y. Zhang, L. Yang, Y. Tan, F. He, H. Xia, Addition of alkynes and osmium carbynes towards functionalized Dπ–Pπ conjugated systems. Nat. Commun. 11(1), 4651 (2020). https://doi.org/10.1038/s41467-020-18498-2
P. Schwerdtfeger, L.N. Wirz, J. Avery, The topology of fullerenes. Wiley Interdiscip. Rev. Comput. Mol. Sci. 5(1), 96–145 (2015). https://doi.org/10.1002/wcms.1207
Y. Hashikawa, T. Fushino, Y. Murata, Double-holed fullerenes. J. Am. Chem. Soc. 142(49), 20572–20576 (2020). https://doi.org/10.1021/jacs.0c10676
V. Mahamiya, A. Shukla, B. Chakraborty, Scandium decorated C24 fullerene as high capacity reversible hydrogen storage material: insights from density functional theory simulations. Appl. Surf. Sci. 573, 151389 (2022). https://doi.org/10.1016/j.apsusc.2021.151389
V. Mahamiya, A. Shukla, B. Chakraborty, Exploring yttrium doped C24 fullerene as a high-capacity reversible hydrogen storage material: DFT investigations. J. Alloys Compd. 897, 162797 (2022). https://doi.org/10.1016/j.jallcom.2021.162797
J. Wang, Y. Du, L. Sun, Ca-decorated novel boron sheet: a potential hydrogen storage medium. Int. J. Hydrogen Energy 41(10), 5276–5283 (2016). https://doi.org/10.1016/j.ijhydene.2016.01.039
R.K. Sahoo, B. Chakraborty, S. Sahu, Reversible hydrogen storage on alkali metal (Li and Na) decorated C20 fullerene: a density functional study. Int. J. Hydrogen Energy 46(80), 40251–40261 (2021). https://doi.org/10.1016/j.ijhydene.2021.09.219
V. Mahamiya, A. Shukla, N. Garg, B. Chakraborty, High-capacity reversible hydrogen storage in scandium decorated holey graphyne: theoretical perspectives. Int. J. Hydrogen Energy 47(12), 7870–7883 (2022). https://doi.org/10.1016/j.ijhydene.2021.12.112
V. Mahamiya, A. Shukla, B. Chakraborty, Ultrahigh reversible hydrogen storage in K and Ca decorated 4–6-8 biphenylene sheet. Int. J. Hydrogen Energy (2022). https://doi.org/10.1016/j.ijhydene.2022.01.216
L.A. Desales-Guzmán, J.H. Pacheco-Sánchez, F.J. Isidro-Ortega, K. De la Mora-Zarco, Hydrogen storage in Ca-decorated carbyne C10-ring on either Dnh or D(n/2)h symmetry. DFT study. Int. J. Hydrogen Energy 45(11), 6780–6792 (2020). https://doi.org/10.1016/j.ijhydene.2019.12.151
L.A. Desales Guzmán, J.H. Pacheco Sánchez, J.S. Arellano Peraza, Carbyne ring activated using ZnCl2 for hydrogen adsorption: DFT study. ACS Omega 7(12), 10100–10114 (2022). https://doi.org/10.1021/acsomega.1c06149
R. Konda, A. Deshmukh, E. Titus, A. Chaudhari, Alkali, alkaline earth and transition metal doped B6H6 complexes for hydrogen storage. Int. J. Hydrogen Energy 42(37), 23723–23730 (2017). https://doi.org/10.1016/j.ijhydene.2017.05.023
L.G. Arellano, F. de Santiago, Á. Miranda, F. Salazar, A. Trejo, L.A. Pérez, M. Cruz-Irisson, Hydrogen storage capacities of alkali and alkaline-earth metal atoms on SiC monolayer: a first-principles study. Int. J. Hydrogen Energy 46(38), 20266–20279 (2021). https://doi.org/10.1016/j.ijhydene.2020.03.078
S. Kumar, T.J. Dhilip Kumar, Hydrogen trapping potential of Ca decorated metal-graphyne framework. Energy 199, 117453 (2020). https://doi.org/10.1016/j.energy.2020.117453
Z.M. Ao, Q. Jiang, R.Q. Zhang, T.T. Tan, S. Li, Al doped graphene: a promising material for hydrogen storage at room temperature. J. Appl. Phys. 105(7), 074307 (2009). https://doi.org/10.1063/1.3103327
W. Liu, Y.H. Zhao, Y. Li, Q. Jiang, E.J. Lavernia, Enhanced hydrogen storage on Li-dispersed carbon nanotubes. J. Phys. Chem. C 113(5), 2028–2033 (2009). https://doi.org/10.1021/jp8091418
E. Beheshti, A. Nojeh, P. Servati, A first-principles study of calcium-decorated, boron-doped graphene for high capacity hydrogen storage. Carbon N. Y. 49(5), 1561–1567 (2011). https://doi.org/10.1016/j.carbon.2010.12.023
K.R.S. Chandrakumar, S.K. Ghosh, Alkali-metal-induced enhancement of hydrogen adsorption in C60 fullerene: an ab initio study. Nano Lett. 8(1), 13–19 (2008). https://doi.org/10.1021/nl071456i
V. Kalamse, N. Wadnerkar, A. Deshmukh, A. Chaudhari, Interaction of molecular hydrogen with Ni doped ethylene and acetylene complex. Int. J. Hydrogen Energy 37(6), 5114–5121 (2012). https://doi.org/10.1016/j.ijhydene.2011.12.100
L. Kavan, J. Kastner, Carbyne forms of carbon: continuation of the story. Carbon N. Y. 32(8), 1533–1536 (1994). https://doi.org/10.1016/0008-6223(94)90149-X
W.A. Chalifoux, M.J. Ferguson, R. McDonald, F. Melin, L. Echegoyen, R.R. Tykwinski, Adamantyl-endcapped polyynes. J. Phys. Org. Chem. 25(1), 69–76 (2012). https://doi.org/10.1002/poc.1874
Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. A.; Nakatsuji, H.; Caricato, M.; Li, X.; Hratchian, H. P.; Izmaylov, A. F.; Bloino, J.; Zheng, G.; Sonnenberg, J. L.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Montgomery, J. A., Jr.; Peralta, J. E.; Ogliaro, F.; Bearpark, M.; Heyd, J. J.; Brothers, E.; Kudin, K. N., Staroverov, V. N., Normand, R. K. J., Raghavachari, K.; Rendell, A.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.; Rega, M. C. N.; Millam, J. M.; Klene, M.; Knox, J. E.; Cross, J. B.; Bakken, V.; Adamo, C.; Gomperts, J. J. R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski, J. W.; Martin, R. L.; Morokuma, K.; Zakrzewski, V. G.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Dapprich, S.; Daniels, A. D.; Farkas, Ö .; Foresman, J. B.; Ortiz, J. V.; Cioslowski, J. D. J. F. Gaussian 09, Revision D.01; Gaussian, Inc.: Wallingford CT, 2009.
R. I. Dennington, T. Keith, J. Millam, GaussView Version 5.0.8, Semichem. Inc., Shawnee Mission, KS, 2008
N. Kosar, T. Mahmood, K. Ayub, Role of dispersion corrected hybrid GGA class in accurately calculating the bond dissociation energy of carbon halogen bond: a benchmark study. J. Mol. Struct. 1150, 447–458 (2017). https://doi.org/10.1016/j.molstruc.2017.08.104
N. Kosar, K. Ayub, T. Mahmood, Accurate theoretical method for homolytic cleavage of C Sn bond: a benchmark approach. Comput. Theor. Chem. 1140, 134–144 (2018). https://doi.org/10.1016/j.comptc.2018.08.003
F. Neese, F. Wennmohs, U. Becker, C. Riplinger, The ORCA quantum chemistry program package. J. Chem. Phys. 152(22), 224108 (2020). https://doi.org/10.1063/5.0004608
T. Lu, F. Chen, Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33(5), 580–592 (2012). https://doi.org/10.1002/jcc.22885
W. Humphrey, A. Dalke, K. Schulten, VMD: visual molecular dynamics. J. Mol. Graph. 14(1), 33–38 (1996). https://doi.org/10.1016/0263-7855(96)00018-5
F. Neese, Software update: the ORCA program system, Version 4.0. WIREs Comput. Mol. Sci. (2018). https://doi.org/10.1002/wcms.1327
T. Torelli, L. Mitas, Electron correlation in C4N+2 carbon rings: aromatic versus dimerized structures. Phys. Rev. Lett. 85(8), 1702–1705 (2000). https://doi.org/10.1103/PhysRevLett.85.1702
F. Ullah, K. Ayub, M.A. Gilani, M. Imran, T. Mahmood, C10F as a potential anode material for alkali-ion batteries; a quantum chemical approach. Comput. Theor. Chem. 1206, 113470 (2021). https://doi.org/10.1016/j.comptc.2021.113470
F. Khaliq, K. Ayub, T. Mahmood, S. Muhammad, S. Tabassum, M.A. Gilani, First example of lanthanum as dopant on Al12N12 and Al12P12 nanocages for improved electronic and nonlinear optical properties with high stability. Mater. Sci. Semicond. Process. 135, 106122 (2021). https://doi.org/10.1016/j.mssp.2021.106122
P. Khan, T. Mahmood, K. Ayub, S. Tabassum, M. Amjad Gilani, Turning diamondoids into nonlinear optical materials by alkali metal substitution: a DFT investigation. Opt. Laser Technol. 142, 107231 (2021). https://doi.org/10.1016/j.optlastec.2021.107231
J. Beheshtian, I. Ravaei, Hydrogen storage by BeO nano-cage: a DFT study. Appl. Surf. Sci. 368, 76–81 (2016). https://doi.org/10.1016/j.apsusc.2016.01.239
A. Catellani, M. Posternak, A. Baldereschi, A.J. Freeman, Bulk and surface electronic structure of hexagonal boron nitride. Phys. Rev. B 36(11), 6105–6111 (1987). https://doi.org/10.1103/PhysRevB.36.6105
A. Kumar, N. Vyas, A.K. Ojha, Hydrogen storage in magnesium decorated boron clusters (Mg2Bn, n = 4–14): a density functional theory study. Int. J. Hydrogen Energy 45(23), 12961–12971 (2020). https://doi.org/10.1016/j.ijhydene.2020.03.018
A.S. Rad, K. Ayub, Ni adsorption on Al12P12 nano-cage: a DFT study. J. Alloys Compd. 678, 317–324 (2016). https://doi.org/10.1016/j.jallcom.2016.03.175
A.S. Rad, S.M. Aghaei, V. Poralijan, M. Peyravi, M. Mirzaei, Application of pristine and Ni-decorated B12P12 nano-clusters as superior media for acetylene and ethylene adsorption: DFT calculations. Comput. Theor. Chem. 1109, 1–9 (2017). https://doi.org/10.1016/j.comptc.2017.03.030
A.S. Rad, K. Ayub, Enhancement in hydrogen molecule adsorption on B12N12 nano-cluster by decoration of nickel. Int. J. Hydrogen Energy 41(47), 22182–22191 (2016). https://doi.org/10.1016/j.ijhydene.2016.08.158
H.J.C. Berendsen, J.P.M. Postma, W.F. van Gunsteren, A. DiNola, J.R. Haak, Molecular dynamics with coupling to an external bath. J. Chem. Phys. 81(8), 3684–3690 (1984). https://doi.org/10.1063/1.448118
C. Liu, Y.Y. Fan, M. Liu, H.T. Cong, H.M. Cheng, M.S. Dresselhaus, Hydrogen storage in single-walled carbon nanotubes at room temperature. Science (80-) 286(5442), 1127–1129 (1999). https://doi.org/10.1126/science.286.5442.1127
A. Arami-Niya, W.M.A.W. Daud, F.S. Mjalli, Using granular activated carbon prepared from oil palm shell by ZnCl2 and physical activation for methane adsorption. J. Anal. Appl. Pyrol. 89(2), 197–203 (2010). https://doi.org/10.1016/j.jaap.2010.08.006
J.M. Blackman, J.W. Patrick, A. Arenillas, W. Shi, C.E. Snape, Activation of carbon nanofibres for hydrogen storage. Carbon N. Y. 44(8), 1376–1385 (2006). https://doi.org/10.1016/j.carbon.2005.11.015
M. Jordá-Beneyto, F. Suárez-García, D. Lozano-Castelló, D. Cazorla-Amorós, A. Linares-Solano, Hydrogen storage on chemically activated carbons and carbon nanomaterials at high pressures. Carbon N. Y. 45(2), 293–303 (2007). https://doi.org/10.1016/j.carbon.2006.09.022
G. Yushin, R. Dash, J. Jagiello, J.E. Fischer, Y. Gogotsi, Carbide-derived carbons: effect of pore size on hydrogen uptake and heat of adsorption. Adv. Funct. Mater. 16(17), 2288–2293 (2006). https://doi.org/10.1002/adfm.200500830
C. Zhang, Z. Geng, M. Cai, J. Zhang, X. Liu, H. Xin, J. Ma, Microstructure regulation of super activated carbon from biomass source corncob with enhanced hydrogen uptake. Int. J. Hydrogen Energy 38(22), 9243–9250 (2013). https://doi.org/10.1016/j.ijhydene.2013.04.163
G. Sethia, A. Sayari, Activated carbon with optimum pore size distribution for hydrogen storage. Carbon N. Y. 99, 289–294 (2016). https://doi.org/10.1016/j.carbon.2015.12.032
M.M. de Castro, M. Martinez-Escandell, M. Molina-Sabio, F. Rodriguez-Reinoso, Hydrogen adsorption on KOH activated carbons from mesophase pitch containing Si, B, Ti or Fe. Carbon N. Y. 48(3), 636–644 (2010). https://doi.org/10.1016/j.carbon.2009.10.005
K. Babeł, K. Jurewicz, KOH activated lignin based nanostructured carbon exhibiting high hydrogen electrosorption. Carbon N. Y. 46(14), 1948–1956 (2008). https://doi.org/10.1016/j.carbon.2008.08.005
D.P. Vargas, L. Giraldo, A. Erto, J.C. Moreno-Piraján, Chemical modification of activated carbon monoliths for CO2 adsorption. J. Therm. Anal. Calorim. 114(3), 1039–1047 (2013). https://doi.org/10.1007/s10973-013-3086-3
Acknowledgements
Authors acknowledge the Higher Education Commission of Pakistan and COMSATS University Islamabad, Abbottabad Campus for their financial and technical support.
Funding
The authors have disclosed all funding sources.
Author information
Authors and Affiliations
Contributions
MB and NK Acquisition and analysis of data; manuscript drafting, Software handling, data handling MAG and FU Software; Study conception and design; funding acquisition MSA Critical revision Resources; TM and KA Supervision; Study conception and design; Critical revision; Resources.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kosar, N., Bibi, M., Ullah, F. et al. Reversible H2 Storage Capacity of Ni Functionalized Carbyne (C10) Complex. J Inorg Organomet Polym 33, 515–528 (2023). https://doi.org/10.1007/s10904-022-02516-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-022-02516-5