Abstract
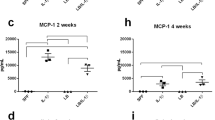
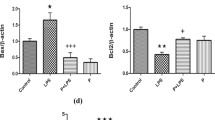
The gut microbiota influences brain development and functioning through the gut-brain axis. This is first study regulate maternal gut microbiota and fetal neurodevelopment processes by using probiotics such as Bifidobacterium bifidum (BIF) and Lactobacillus salivarius (LAC) in the prenatal period. In this study, Wistar Albino female rats were divided into five groups; Control, lipopolysaccharide (LPS, 100 µg/kg), LPS + LAC, LPS + BIF and LPS + LAC + BIF (4 × 109 ml CFU). Maternal rats were given probiotics for 21 days. Inflammation was induced by lipopolysaccharide (LPS), on the 17th day of pregnancy. After birth, the brain tissues of the maternal and neonatal rats were removed and their blood was collected. Fecal calprotectin levels of pregnant rats were measured as an important biomarker in determining intestinal flora disruption. Calprotectin levels were high in LPS group (p < 0.05). Aβ 1–42, APP, γ secretase and β- secretase levels were higher in both maternal and neonatal LPS groups (p < 0.05). These levels were statistically decreased in the probiotic groups compared to the LPS group, as demonstrated in both biochemical and histological analyzes (p < 0.05). While BDNF mRNA expression decreased in LPS groups, APP level increased in the same group. The difference between groups in mRNA expressions in the neonatal brain tissues was similar to maternal brain tissues. What’s more, BDNF/actin and APP/actin rates were proven by western blot and the damage caused by neuroinflammation in the brain tissue and the preservation of the intestinal microbiota were visualized histopathologically on the morphological structures in all groups. It will shed light on new therapeutic strategies for the impact of the use of probiotics on the neurodevelopmental processes of the neonatal against LPS-induced inflammatory responses and impaired gut microbiota in the prenatal period.








Similar content being viewed by others
Data availability
The data support the fndings of this research are available from the corresponding author (fatih.kar@ksbu.edu.tr) upon reasonable request. The authors will take the responsibility for maintaining availability.
References
Asti A, Gioglio L (2014) Can a bacterial endotoxin be a key factor in the kinetics of amyloid fibril formation? J Alzheimers Dis 39(1):169–179. https://doi.org/10.3233/JAD-131394
Borre YE, Keeffe GW, Clarke G, Stanton C, Dinan TG, Cryan JF (2014) Microbiota and neurodevelopmental windows: implications for brain disorders. Trends Mol Med 20(9):509–518. https://doi.org/10.1016/j.molmed.2014.05.002
Braniste V, Al-Asmakh M, Kowal C, Anuar F, Abbaspour A, Tóth M, Korecka A et al (2014) The gut microbiota influences blood-brain barrier permeability in mice. Sci Transl Med 6(263):263ra158. https://doi.org/10.1126/scitranslmed.3009759
Carlino D, De Vanna M, Tongiorgi E (2013) Is altered BDNF biosynthesis a general feature in patients with cognitive dysfunctions? Neuroscientist 19(4):345–353. https://doi.org/10.1177/1073858412469444
Cattaneo A, Cattane N, Galluzzi S, Provasi S, Lopizzo N, Festari C, Group IF (2017) Association of brain amyloidosis with pro-inflammatory gut bacterial taxa and peripheral inflammation markers in cognitively impaired elderly. Neurobiol Aging 49:60–68. https://doi.org/10.1016/j.neurobiolaging.2016.08.019
Clarke G, Grenham S, Scully P, Fitzgerald P, Moloney RD, Shanahan F, Dinan TG, Cryan JF (2013) Themicrobiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol Psychiatry 18(6):666–673. https://doi.org/10.1038/mp.2012.77
Desbonnet L, Clarke G, Shanahan F, Dinan TG, Cryan JF (2014) Microbiota is essential for social development in the mouse. Mol Psychiatry 19(2):146–148. https://doi.org/10.1038/mp.2013.65
Eikelenboom P, Veerhuis R, Exel EV, Hoozemans JJM, Rozemuller AJM, van Gool WA (2011) The early involvement of the innate immunity in the pathogenesis of lateonset Alzheimer’s disease: neuropathological, epidemiological and genetic evidence. Curr Alzheimer Res 8(2):142–150. https://doi.org/10.2174/156720511795256080
Enayati M, Solati J, Hosseini MH, Shahi HR, Saki G, Salari AA (2012) Maternal infection during late pregnancy increases anxiety- and depression-like behaviors with increasing age in male offspring. Brain Res Bull 87(2–3):295–302. https://doi.org/10.1016/j.brainresbull.2011.08.015
Fortier ME, Luheshi GN, Boksa P (2007) Effects of prenatal infection on prepulse inhibition in the rat depend on the nature of the infectious agent and the stage of pregnancy. Behav Brain Res 181(2):270–277. https://doi.org/10.1016/j.bbr.2007.04.016
Gareau MG, Wine E, Rodrigues DM, Cho JH, Whary MT, Philpott DJ, MacQueen G, Sherman PM (2011) Bacterial infection causes stress-induced memory dysfunction in mice. Gut 60(3):307–317. https://doi.org/10.1136/gut.2009.202515
Guslandi M (2015) Role of probiotics in Crohn’s disease and in pouchitis. J Clin Gastroenterol 49(1):46–49. https://doi.org/10.1097/MCG.0000000000000351
Han C, Ding Z, Shi H, Qian W, Hou X, Lin R (2016) The role of probiotics in lipopolysaccharide-induced autophagy in intestinal epithelial cells. Cell Physiol Biochem 38(6):2464–2478. https://doi.org/10.1159/000445597
Hebert JC, Radford-Smith DE, Probert F, Ilott N, Chan KW, Anthony DC, Burnet PW (2021) Mom’s diet matters: Maternal prebiotic intake in mice reduces anxiety and alters brain gene expression and the fecal microbiome in offspring. Brain Behav Immun 91:230–244. https://doi.org/10.1016/j.bbi.2020.09.034
Heijtz RD, Wang S, Anuar F, Qian Y, Björkholm B, Samuelsson A, Pettersson S (2011) Normal gut microbiota modulates brain development and behavior. Proc Natl Acad Sci 108(7):3047–3052. https://doi.org/10.1073/pnas.1010529108
Jakobsson HE, Rodríguez-Piñeiro AM, Schütte A, Ermund A, Boysen P, Bemark M, Johansson ME (2015) The composition of the gut microbiota shapes the colon mucus barrier. EMBO Rep 16(2):164–177. https://doi.org/10.15252/embr.201439263
Kahn MS, Kranjac D, Alonzo CA, Haase JF, Cedillos RO, McLinden KA, Boehm GW, Chumley MJ (2012) Prolonged elevation in hippocampal A beta and cognitive deficits following repeated endotoxin exposure in the mouse. Behav Brain Res 229(1):176–184. https://doi.org/10.1016/j.bbr.2012.01.010
Kesika P, Suganthy N, Sivamaruthi BS, Chaiyasut C (2021) Role of gut-brain axis, gut microbial composition, and probiotic intervention in Alzheimer’s disease. Life Sci 264:118627. https://doi.org/10.1016/j.lfs.2020.118627
Leblhuber F, Geisler S, Steiner K, Fuchs D, Schutz B (2015) Elevated fecal calprotectin in patients with Alzheimer’s dementia indicates leaky gut. J Neural Transm 122(9):1319–1322. https://doi.org/10.1007/s00702-015-1381-9
Lee DC, Rizer J, Selenica MLB, Reid P, Kraft C, Johnson A, Blair D et al (2010) LPS-induced inflammation exacerbates phospho-tau pathology in rTg4510 mice. J Neuroinflammation 7(1):1–16. https://doi.org/10.1186/1742-2094-7-56
Liang S, Wang T, Hu X, Luo J, Li W, Wu X, Duan Y, Jin F (2015) Administration of Lactobacillus helveticus NS8 improves behavioral, cognitive, and biochemical aberrations caused by chronic restraint stress. Neuroscience 310:561–577. https://doi.org/10.1016/j.neuroscience.2015.09.033
Lynch MA (2014) The impact of neuroimmune changes on development of amyloid pathology; relevance to Alzheimer’s disease. Immunology 141(3):292–301. https://doi.org/10.1111/imm.12156
Martínez-Díaz JA, Hernández-Aguilar ME, Rojas-Durán F, Herrera-Covarrubias D, García-Hernández LI, Mestizo-Gutiérrez SL, Aranda-Abreu GE (2020) Expression of proteins linked to Alzheimer’s disease in C6 rat glioma cells under the action of lipopolysaccharide (LPS), nimesulide, resveratrol and citalopram. Turk J Biochem 45(6):793–801. https://doi.org/10.1515/tjb-2020-0091
Mouihate A, Mehdawi H (2016) Toll-like receptor 4-mediated immune stress in pregnant rats activates STAT3 in the fetal brain: role of interleukin-6. Pediatr Res 79(5):781–787. https://doi.org/10.1038/pr.2015.86
Noh H, Jeon J, Seo H (2014) Systemic injection of LPS induces region-specific neuroinflammation and mitochondrial dysfunction in normal mouse brain. Neurochem Int 69:35–40. https://doi.org/10.1016/j.neuint.2014.02.008
O’Mahony L, McCarthy J, Kelly P, Hurley G, Luo F, Chen K, Quigley EM (2005) Lactobacillus and bifidobacterium in irritable bowel syndrome: symptom responses and relationship to cytokine profiles. Gastroenterology 128(3):541–551. https://doi.org/10.1053/j.gastro.2004.11.050
Özpak L, Pazarbaşı A, Keser N (2017) Alzheimer Hastalığının Genetiği ve Epigenetiği. Arşiv Kaynak Tarama Dergisi 26(1):34–49. https://doi.org/10.17827/aktd.280520
Pathak N, Vimal SK, Tandon I, Agrawal L, Hongyi C, Bhattacharyya S (2021) Neurodegenerative Disorders of Alzheimer, Parkinsonism, Amyotrophic Lateral Sclerosis and Multiple Sclerosis: An Early Diagnostic Approach for Precision Treatment. Metabolic Brain Disease, 1-38. https://doi.org/10.1007/s11011-021-00800-w
Prince M, Bryce R, Albanese E, Wimo A, Ribeiro W, Ferri CP (2013) The global prevalence of dementia: a systematic review and metaanalysis. Alzheimer’s Dementia 9(1):63–75. https://doi.org/10.1016/j.jalz.2012.11.007
Qin L, Wu X, Block ML, Liu Y, Breese GR, Hong JS, Knapp DJ, Crews FT (2007) Systemic LPS causes chronic neuroinflammation and progressive neurodegeneration. Glia 55(5):453–462. https://doi.org/10.1002/glia.20467
Sharon G, Sampson TR, Geschwind DH, Mazmanian SK (2016) The central nervous system and the gut microbiome. Cell 167(4):915–932. https://doi.org/10.1016/j.cell.2016.10.027
Shoemark DK, Allen SJ (2015) The microbiome and disease: reviewing the links between the oral microbiome, aging, and Alzheimer’s disease. J Alzheimers Dis 43:725–738. https://doi.org/10.3233/JAD-141170
Sochocka M, Donskow-Łysoniewska K, Diniz BS, Kurpas D, Brzozowska E, Leszek J (2019) The gut microbiome alterations and inflammation-driven pathogenesis of Alzheimer’s disease—a critical review. Mol Neurobiol 56(3):1841–1851. https://doi.org/10.1007/s12035-018-1188-4
Tan ZX, Dong F, Wu LY, Feng YS, Zhang F (2021) The Beneficial Role of Exercise on Treating Alzheimer’s Disease by Inhibiting β-Amyloid Peptide. Mol Neurobiol. https://doi.org/10.1007/s12035-021-02514-7
Wang B, Xu H, Xu F, Zeng Z, Wei H (2016) Efficacy of oral Bifidobacterium bifidum ATCC 29521 on microflora and antioxidant in mice. Can J Microbiol 62(3):249–262. https://doi.org/10.1139/cjm-2015-0685
Wang IK, Wu YY, Yang YF, Ting IW, Lin CC, Yen TH, Chen JH, Wang CH, Huang CC, Lin HC (2015) The effect of probiotics on serum levels of cytokine and endotoxin in peritoneal dialysis patients: a randomised, double-blind, placebo-controlled trial. Benef Microbes 6(4):423–430. https://doi.org/10.3920/BM2014.0088
Woo JY, Gu W, Kim KA, Jang SE, Han MJ, Kim DH (2014) Lactobacillus pentosus var. plantarum C29 ameliorates memory impairment and inflammaging in a D-galactose-induced accelerated aging mouse model. Anaerobe 27:22–26. https://doi.org/10.1016/j.anaerobe.2014.03.003
Wu X, Vallance BA, Boyer L, Bergstrom KS, Walker J, Madsen K, Jacobson K (2008) Saccharomyces boulardii ameliorates Citrobacter rodentium-induced colitis through actions on bacterial virulence factors. American Journal of Physiology-Gastrointestinal and Liver Physiology, 294(1):G295–306. https://doi.org/10.1152/ajpgi.00173.2007.
Zhang R, Miller RG, Gascon R, Champion S, Katz J, Lancero M, Narvaez A, Honrada R, Ruvalcaba D, McGrath MS (2009) Circulating endotoxin and systemic immune activation in sporadic amyotrophic lateral sclerosis (sALS). J Neuroimmunol 206(1–2):121–124. https://doi.org/10.1016/j.jneuroim.2008.09.017
Zhao J, Bi W, Xiao S, Lan X, Cheng X, Zhang J, Zhu L (2019) Neuroinflammation induced by lipopolysaccharide causes cognitive impairment in mice. Sci Rep 9:5790. https://doi.org/10.1038/s41598-019-42286-8
Funding
This study was supported by Eskişehir Osmangazi University Scientific Research Projects Commission. (Project No: 2019–1915).
Author information
Authors and Affiliations
Contributions
F.K. designed, performed, analyzed all the experiments, and wrote the manuscript. C.H. and E.K. assisted in in vivo experiments, ELISA measurements, real-time qPCR and Western Blot methods. D.B.D. helped with the development of histopathological protocol. F.K. and E.K. analyzed behavioral data from maternal behavior. F.K and C.H. performed ELISA analyses to detect calprotectin levels in feces. S.U. and G.K. supervised the design, development, and analyses of molecular experiments. F.K., C.H., and G.K supervised the whole study design, development, and data analyses. F.K. and E.K. also contributed to the fnal version of the paper. All authors made manuscript revisions and approved the submitted version.
Corresponding author
Ethics declarations
Ethics approval
The study was applied to the protocol approved by the ESOGU Animal Experiments Local Ethics Committee (HADYEK) with the decision number 636–1 dated 10.10.2019.
Consent to participate
Not applicable.
Consent for publication
All authors give consent for publication.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kar, F., Hacioglu, C., Kar, E. et al. Probiotics ameliorates LPS induced neuroinflammation injury on Aβ 1–42, APP, γ-β secretase and BDNF levels in maternal gut microbiota and fetal neurodevelopment processes. Metab Brain Dis 37, 1387–1399 (2022). https://doi.org/10.1007/s11011-022-00964-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-00964-z