Abstract
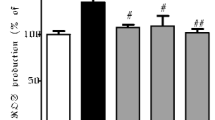
Parkinson's disease (PD) is a neurodegenerative disorder characterized by both motor and non-motor features. The current treatment regimen for PD are dopamine enhancers which have been reported to worsen the disease prognosis after long term treatment, thus, the need for better treatment options. This study sought to investigate the protective action of Double Stem Cell® (DSC), a blend of stem cells extracts from Swiss apples (Malus Domestica) and Burgundy grapes (Vitis vinifera) on 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced Parkinsonism in mice and genetic model of PD in Drosophila melanogaster. Male albino mice were pretreated with MPTP (4 × 20 mg/kg, i.p., two hourly in 8 h), twelve hours before administration of DSC (8, 40, or 200 mg/kg, p.o.). Thereafter, behavioural, biochemical and immunohistochemical assays were carried out. The impact of vehicle or DSC supplementation on α-synuclein aggregation was evaluated in Drosophila melanogaster using the UAS-Gal4 system, female DDC-Gal4 flies were crossed with male UAS-α-synuclein, the progenies were examined for fecundity, locomotion, memory, and lifespan. MPTP-induced motor deficits in open field test (OFT), working memory impairment (Y-maze test (YMT)) and muscle incoordination (rotarod test) were ameliorated by DSC (8, 40 or 200 mg/kg) through dose-dependent and significant improvements in motor, cognitive and motor coordination. Moreso, MPTP exposure caused significant increase in lipid peroxidation and decrease in antioxidant enzymes activities (glutathione, catalase and superoxide dismutase) in the midbrain which were attenuated by DSC. MPTP-induced expression of microglia (iba-1), astrocytes (glia fibrillary acidic protein; GFAP) as well as degeneration of dopamine neurons (tyrosine hydroxylase positive neurons) in the substantia nigra (SN) were reversed by DSC. Supplementation of flies feed with graded concentration of DSC (0.8, 4 or 20 mg/ml) did not affect fecundity but improved climbing activity and lifespan. Findings from this study showed that Double Stem Cell improved motor and cognitive functions in both mice and Drosophila through attenuation of neurotoxin-induced oxidative stress and neuroinflammation.














Similar content being viewed by others
Data availability
Data will be made available on request.
Code availability
Not applicable.
References
Ali YO, Escala W, Ruan K, Zhai RG (2011) Assaying locomotor, learning, and memory deficits in Drosophila models of neurodegeneration. J Vis Exp 49:2504. https://doi.org/10.3791/2504
Aryal B, Lee Y (2019) Disease model organism for Parkinson disease: Drosophila melanogaster. BMB Rep 52(4):250–258. https://doi.org/10.5483/BMBRep.2019.52.4.204
Bellomo G, Mirabelli F, DiMonte D, Richelmi P, Thor H, Orrenius C, Orrenius S (1987) Formation and reduction of glutathione-protein mixed disulfides during oxidative stress. A study with isolated hepatocytes and menadione (2-methyl-1,4-naphthoquinone). Biochem Pharmacol 36(8):1313–20. https://doi.org/10.1016/0006-2952(87)90087-6
Blesa J, Phani S, Jackson-Lewis V, Przedborski S (2012) Classic and new animal models of Parkinson’s disease. J Biomed Biotechnol 2012:845618. https://doi.org/10.1155/2012/845618
Bohnen NI, Frey KA, Studenski S, Kotagal V, Koeppe RA, Scott PJ, Albin RL, Müller ML (2013) Gait speed in Parkinson disease correlates with cholinergic degeneration. Neurology 81(18):1611–1616. https://doi.org/10.1212/WNL.0b013e3182a9f558
Busson D, Pret AM (2007) GAL4/UAS targeted gene expression for studying Drosophila Hedgehog signaling. Methods Mol Biol 397:161–201. https://doi.org/10.1007/978-1-59745-516-9_13
Chagniel L, Robitaille C, Lacharité-Mueller C, Bureau G, Cyr M (2012) Partial dopamine depletion in MPTP-treated mice differentially altered motor skill learning and action control. Behav Brain Res 228(1):9–15. https://doi.org/10.1016/j.bbr.2011.11.019
Coetzee SG, Pierce S, Brundin P, Brundin L, Hazelett DJ, Coetzee GA (2016) Enrichment of risk SNPs in regulatory regions implicate diverse tissues in Parkinson’s disease etiology. Sci Rep 27(6):30509. https://doi.org/10.1038/srep30509
Ellman GL, Courtney KD, Andres V Jr, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Feany MB, Bender WW (2000) A Drosophila model of Parkinson’s disease. Nature 404(6776):394–398. https://doi.org/10.1038/35006074
Fridovich I (1974) Superoxide dismutases. Adv Enzymol Relat Areas Mol Biol 41(0):35–97. https://doi.org/10.1002/9780470122860.ch2
Gao HM, Kotzbauer PT, Uryu K, Leight S, Trojanowski JQ, Lee VM (2008) Neuroinflammation and oxidation/nitration of alpha-synuclein linked to dopaminergic neurodegeneration. J Neurosci 28(30):7687–7698. https://doi.org/10.1523/JNEUROSCI.0143-07.2008
Garcia-Ptacek S, Kramberger MG (2016) Parkinson disease and dementia. J Geriatr Psychiatry Neurol 29(5):261–270. https://doi.org/10.1177/0891988716654985
Harris JP, Burrell JC, Struzyna LA, Chen HI, Serruya MD, Wolf JA, Duda JE, Cullen DK (2020) Emerging regenerative medicine and tissue engineering strategies for Parkinson’s disease. NPJ Parkinsons Dis 6:4. https://doi.org/10.1038/s41531-019-0105-5
Ishola IO, Chatterjee M, Tota S, Tadigopulla N, Adeyemi OO, Palit G, Shukla R (2012) Antidepressant and anxiolytic effects of amentoflavone isolated from Cnestis ferruginea in mice. Pharmacol Biochem Behav 103(2):322–331. https://doi.org/10.1016/j.pbb.2012.08.017
Ishola IO, Agbaje EO, Akinleye MO, Ibeh CO, Adeyemi OO (2014a) Antidepressant-like effect of the hydroethanolic leaf extract of Alchornea cordifolia (Schumach. & Thonn.) Mull. Arg. (Euphorbiaceae) in mice: involvement of monoaminergic system. J Ethnopharmacol 158 Pt A:364–72. https://doi.org/10.1016/j.jep.2014.10.008
Ishola IO, Ochieng CO, Olayemi SO, Jimoh MO, Lawal SM (2014b) Potential of novel phytoecdysteroids isolated from Vitex doniana in the treatment depression: involvement of monoaminergic systems. Pharmacol Biochem Behav 127:90–100. https://doi.org/10.1016/j.pbb.2014.11.005
Ishola IO, Olayemi SO, Yemitan OK, Umeh EA (2015) Antidepressant and anxiolytic effects of the methanol root extract of Capparis thonningii: involvement of monoaminergic, cholinergic and GABAergic systems. Drug Res (Stuttg) 65(4):205–213. https://doi.org/10.1055/s-0034-1376963
Ishola IO, Akinleye MO, Oduola MD, Adeyemi OO (2016) Roles of monoaminergic, antioxidant defense and neuroendocrine systems in antidepressant-like effect of Cnestis ferruginea Vahl ex DC (Connaraceae) in rats. Biomed Pharmacother 83:340–348. https://doi.org/10.1016/j.biopha.2016.06.054
Ishola IO, Akataobi OE, Alade AA, Adeyemi OO (2019) Glimepiride prevents paraquat-induced Parkinsonism in mice: involvement of oxidative stress and neuroinflammation. Fundam Clin Pharmacol 33(3):277–285. https://doi.org/10.1111/fcp.12434
Ishola IO, Akinyede AA, Eloke JE, Chaturvedi JP, Narender T (2020) Diastereomeric mixture of calophyllic and isocalophyllic acid ameliorates scopolamine-induced memory impairment in mice: involvement of antioxidant defense and cholinergic systems. Neurotox Res 37(1):58–66. https://doi.org/10.1007/s12640-019-00117-8
Ishola IO, Afolayan O, Akinleye MO, Nwajei WI, Adeyemi OO, Mishra RK (2021a) Ameliorative influence of vildagliptin on genetic model of neurodegenerative diseases in Drosophila melanogaster. Nig J Pharm 55(2):91–97
Ishola IO, Ben-Azu B, Adebayo OA, Ajayi AM, Omorodion IL, Edje KE, Adeyemi OO (2021b) Prevention and reversal of ketamine-induced experimental psychosis in mice by the neuroactive flavonoid, hesperidin: The role of oxidative and cholinergic mechanisms. Brain Res Bull 177:239–251. https://doi.org/10.1016/j.brainresbull.2021.10.007
Ishola IO, Olubodun-Obadun TG, Ochieng CO, Nwajuwe MC, Mukoro RU, Adeyemi OO (2022a) Monoaminergic system involvement in the antidepressant-like and anxiolytic-like properties of novel β-dihydroagarofuran sesquiterpene alkaloid and triterpenes isolated from Gymnosporia heterophylla aerial parts in mice. Neurochem Int 158:105379. https://doi.org/10.1016/j.neuint.2022.105379
Ishola IO, Afolayan O, Badru A, Olubodun-Obadun T, John N, Adeyemi O (2022b) Angiotensin converting enzyme inhibitor captopril prevents neuronal overexpression of amyloid-beta and alpha-synuclein in Drosophila melanogaster genetic models of neurodegenerative diseases. Niger J Physiol Sci 37(1):21–28. https://doi.org/10.54548/njps.v37i1.3
Kozioł K, Zebrowski J, Betlej G, Bator E, Czarny W, Bajorek W, Czarnota B, Czaja R, Król P, Kwiatkowska A (2020) Changes in γH2AX and H4K16ac levels are involved in the biochemical response to a competitive soccer match in adolescent players. Sci Rep 10(1):14481. https://doi.org/10.1038/s41598-020-71436-6
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951)Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–75
M’Angale PG, Staveley BE (2016) The Bcl-2 homologue Buffy rescues α-synuclein-induced Parkinson disease-like phenotypes in Drosophila. BMC Neurosci 17(1):24. https://doi.org/10.1186/s12868-016-0261-z
Niyomrat K, Cheaha D, Nukitram J, Kumarnsit E (2021) Locomotor activity and resting local field potential oscillatory rhythms of 6-OHDA mouse model of Parkinson's disease in response to acute and repeated treatments with Ldopa. Neurosci Lett 759:136007. https://doi.org/10.1016/j.neulet.2021.136007
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95(2):351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Qi C, Varga S, Oh SJ, Lee CJ, Lee D (2017) Optogenetic rescue of locomotor dysfunction and dopaminergic degeneration caused by Alpha-Synuclein and EKO genes. Exp Neurobiol 26(2):97–103. https://doi.org/10.5607/en.2017.26.2.97
Ryan M, Eatmon CV, Slevin JT (2019) Drug treatment strategies for depression in Parkinson disease. Expert Opin Pharmacother 20(11):1351–1363. https://doi.org/10.1080/14656566.2019.1612877
Shiotsuki H, Yoshimi K, Shimo Y, Funayama M, Takamatsu Y, Ikeda K, Takahashi R, Kitazawa S, Hattori N (2010) A rotarod test for evaluation of motor skill learning. J Neurosci Methods 189(2):180–185. https://doi.org/10.1016/j.jneumeth.2010.03.026
Sinha AM, Haldar S, Chatterjee GC (1982) Studies on alkaline phosphatase, catalase and z-galactosidase in Vibrio el tor under normal and rifampicin resistant conditions. Acta Microbiol Pol 31(3–4):271–278
Taylor JM, Main BS, Crack PJ (2013) Neuroinflammation and oxidative stress: co-conspirators in the pathology of Parkinson’s disease. Neurochem Int 62(5):803–819. https://doi.org/10.1016/j.neuint.2012.12.016
Terada T, Yokokura M, Yoshikawa E, Futatsubashi M, Kono S, Konishi T, Miyajima H, Hashizume T, Ouchi Y (2016) Extrastriatal spreading of microglial activation in Parkinson’s disease: a positron emission tomography study. Ann Nucl Med 30(8):579–587. https://doi.org/10.1007/s12149-016-1099-2
Trist BG, Hare DJ, Double KL (2019) Oxidative stress in the aging substantia nigra and the etiology of Parkinson’s disease. Aging Cell 18(6):e13031. https://doi.org/10.1111/acel.13031
Umeda M, Ikeuchi M, Ishikawa M, Ito T, Nishihama R, Kyozuka J, Torii KU, Satake A, Goshima G, Sakakibara H (2021) Plant stem cell research is uncovering the secrets of longevity and persistent growth. Plant J 106(2):326–335. https://doi.org/10.1111/tpj.15184
Varga SJ, Qi C, Podolsky E, Lee D (2014) A new Drosophila model to study the interaction between genetic and environmental factors in Parkinson's disease. Brain Res 1583:277–86. https://doi.org/10.1016/j.brainres.2014.08.021
Yan T, He B, Wan S, Xu M, Yang H, Xiao F, Bi K, Jia Y (2017) Antidepressant-like effects and cognitive enhancement of Schisandra chinensis in chronic unpredictable mild stress mice and its related mechanism. Sci Rep 7(1):6903. https://doi.org/10.1038/s41598-017-07407-1
Zang LY, Misra HP (2003) Inactivation of acetylcholinesterase by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine hydrochloride. Mol Cell Biochem 254(1–2):131–136. https://doi.org/10.1023/a:1027376303043
Zeng XS, Geng WS, Jia JJ (2018) Neurotoxin-induced animal models of parkinson disease: pathogenic mechanism and assessment. ASN Neuro 10:1759091418777438. https://doi.org/10.1177/1759091418777438
Zhao X, Sun X, Cai S, Ran D, Yan Y, Pei Z (2015) Role of α-synuclein in cognitive dysfunction: Studies in Drosophila melanogaster. Mol Med Rep 12(2):2683–2688. https://doi.org/10.3892/mmr.2015.3763
Acknowledgements
The authors are very grateful to the University of Lagos, Central Research Committee for the Grant awarded to IOI, OAK, OSA, AOO (CRC/2018/22).
Funding
This work was supported by the University of Lagos, Central Research Committee for the Grant awarded number CRC/2018/22 to IOI, OAK, OSA, AOO.
Author information
Authors and Affiliations
Contributions
IOI, OAK, OTG and GOD are involved in laboratory bench work while IOI, OAK, OSA and AOO are involved in study conceptualization and manuscript writing.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by Health Research Ethics Committee of the College of Medicine, University of Lagos, Lagos State, Nigeria with the approval number CMUL/HREC/0961/19.
Conflict of interest
We do not have any financial or conflict of interests to declare.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ishola, I.O., Oloyo, A.K., Olubodun-Obadun, T.G. et al. Neuroprotective potential of plant derived parenchymal stem cells extract on environmental and genetic models of Parkinson disease through attenuation of oxidative stress and neuroinflammation. Metab Brain Dis 38, 557–571 (2023). https://doi.org/10.1007/s11011-022-01120-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-01120-3