Abstract
A palladium-catalyzed selective synthesis of cis- or trans-chloro-substituted-\(\alpha \)-methylene-\(\gamma \)-butyrolactones from single substrate (propiolic acid) has been realized by controlling cis- or trans-chloropalladation. This method is highly effective for building C–Cl, C–O, and C–C bonds into a one-pot procedure because of its good atom and step efficiency.
Graphical Abstract





Similar content being viewed by others
References
Seechurn CCCJ, Kitching MO, Colacot TJ, Snieckus V (2012) Palladium-catalyzed cross-coupling: a historical contextual perspective to the 2010 Nobel Prize. Angew Chem Int Ed 51:5062–5085. doi:10.1002/anie.201107017
Cacchi S, Fabrizi G (2011) Update 1 of: Synthesis and functionalization of indoles through palladium-catalyzed reactions. Chem Rev 111: PR215–PR283. doi:10.1021/cr100403z
Lyons TW, Sanford MS (2010) Palladium-catalyzed ligand-directed C–H functionalization reactions. Chem Rev 110:1147–1169. doi:10.1021/cr900184e
Muñiz K (2009) High-oxidation-state palladium catalysis: new reactivity for organic synthesis. Angew Chem Int Ed 48:9412–9423. doi:10.1002/anie.200903671
Beccalli EM, Broggini G, Martinelli M, Sottocornola S (2007) C–C, C–O, C–N bond formation on \(\text{ sp}^{2}\) carbon by Pd(II)-catalyzed reactions involving oxidant agents. Chem Rev 107:5318–5365. doi:10.1021/cr068006f
Nicolaou KC, Bulger PG, Sarlah D (2005) Palladium-catalyzed cross-coupling reactions in total synthesis. Angew Chem Int Ed 44:4442–4489. doi:10.1002/anie.200500368
Chemler SR, Trauner D, Danishefsky SJ (2001) The \(B\)-alkyl suzuki-miyaura cross-coupling reaction: development, mechanistic study, and applications in natural product synthesis. Angew Chem Int Ed 40:4544–4568. doi:10.1002/1521-3773(20011217)
Lu X (2002) Handbook of organopalladium chemistry for organic synthesis, Vol 2, Negishi E (ed). Wiley, New York, pp 2267–2287
Kitson RRA, Millemaggi A, Taylor RJK (2009) The renaissance of \(\alpha \)-methylene-\(\gamma \)-butyrolactones: New synthetic approaches. Angew Chem Int Ed 48:9426–9451 and references therein. doi:10.1002/anie.200903108
Takenaka K, Hashimoto S, Takizawa S, Sasai H (2011) Chlorinative cyclization of 1,6-enynes by enantioselective palladium(II)/palladium(IV) catalysis. Adv Synth Catal 353: 1067–1070. doi:10.1002/adsc.201000926
Li Y, Jardine KJ, Tan R, Song D, Dong VM (2009) Palladium-catalyzed intramolecular carboesterification of olefins. Angew Chem Int Ed 48:9690–9692. doi:10.1002/anie.200905478
Yin G, Liu G (2008) Palladium-catalyzed oxidative cyclization of enynes with hydrogen peroxide as the oxidant. Angew Chem Int Ed 47:5442–5445. doi:10.1002/anie.200801438
Li W, shi M (2008) An efficient method for the synthesis of vinylbromohydrin and vinylbromoalkoxy derivatives and cyclocarbonylation of \(\alpha \)-allenic alcohols catalyzed by palladium chloride. J Org Chem 73:6698–6705. doi: 10.1021/jo8009352
Song J, Shen Q, Xu F, Lu X (2007) Cationic Pd(II)-catalyzed enantioselective cyclization of aroylmethyl 2-alkynoates initiated by carbopalladation of alkynes with arylboronic acids. Org Lett 9:2947–2950. doi:10.1021/ol0711772
Yonehara K, Miyoshi Y, Tsukajima A, Akatsuka T, Saito M (2011) Palladium(II)-catalyzed aerobic intermolecular cyclization of acrylic acid with alkenes to \(\alpha \)-methylene-\(\gamma \)-butyrolactones. Adv Synth Catal 353:1071–1075. doi:10.1002/adsc.201100006
Arcadi A, Cacchi S, Fabrizi G, Moro L (1999) Palladium-catalyzed cyclocarbonylation of \(o\)-ethynylphenols and vinyl triflates to form 3-alkylidene-2-coumaranones. Eur J Org Chem 5:1137–1141. doi:10.1002/(SICI)1099-0690(199905)
Huang L, Wang Q, Liu X, Jiang H (2012) Switch of selectivity in the synthesis of \(\alpha \)-methylene-\(\gamma \)-lactones: palladium-catalyzed intermolecular carboesterification of alkenes with alkynes. Angew Chem Int Ed 51:5696–5700. doi: 10.1002/anie.201109141
Borsini E, Broggini G, Fasana A, Galli S, Khansaa M, Piarulli U, Rigamonti M (2011) Intramolecular palladium-catalyzed aminocarboxylation of olefins as a direct route to bicyclic oxazolidinones. Adv Synth Catal 353:985–994. doi:10.1002/adsc.201000889
Broggini G, Barbera V, Beccalli E, Borsini E, Galli S, Lanza G, Zecchi G (2012) Palladium(II)/copper halide/solvent combination for selective intramolecular domino reactions of indolecarboxylic acid allylamides: An Unprecedented arylation/esterification sequence. Adv Synth Catal 354:159–170. doi:10.1002/adsc.201100614
Kubizna P, Špánik I, Kožíšek J, Szolcsányi P (2010) Synthesis of 2,6-disubstituted piperidine alkaloids from ladybird beetles Calvia 10-guttata and Calvia 14-guttata. Tetrahedron 66: 2351–2355. doi:10.1016/j.tet.2010.01.106
Szolcsányi P, Gracza T, Špánik I (2008) Short racemic syntheses of calvine and epicalvine. Tetrahedron Lett 49:1357–1360. doi:10.1016/j.tetlet.2007.12.099
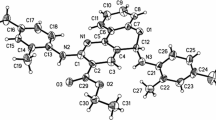
The structures of these two isomers were identified with \(^{1}\text{ H}\) NMR, \(^{13}\text{ C}\) NMR and HRMs data
Bäckvall J-E, Nilsson YIM, Gatti RGP (1995) Stereochemistry and Mechanism of Chloropalladation of Acetylenes. Organometallics 14:4242–4246. doi:10.1021/om00009a029
Chang S-T, Wang DS-Y, Wu C-L, Shiah S-G, Kuo Y-H, Chang C-J (2000) Cytotoxicity of extractives from Taiwania cryptomerioides heartwood. Phytochemistry 55:227–232. doi:10.1016/S0031-9422(00)00275-2
Novák P, Correa A, Gallardo-Donaire J, Martin R (2011) Synergistic palladium-catalyzed C(sp3)-H activation/C(sp3)-O bond formation: a direct, step-economical route to benzolactones. Angew Chem Int Ed 50:12236–12239. doi:10.1002/anie.201105894
Acknowledgments
We acknowledge Bo Liu and Tongxiang Cao for their assistance in preparation of intermediates. This research is supported financially by the joint grant of National Natural Science Foundation of China (NSFC) and National Institute of Health (NIH) of USA (No. 81161120402) and the National 863 Program of China, No. 2012AA020303.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dong, Y., Guo, X., Yu, Y. et al. Highly stereoselective synthesis of (Z)- and (E)-chloro-substituted-\(\alpha \)-methylene-\(\gamma \)-butyrolactone by possibly controlling cis- and trans-chloropalladation. Mol Divers 17, 1–7 (2013). https://doi.org/10.1007/s11030-012-9419-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-012-9419-5