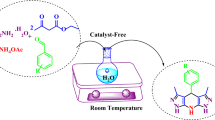
Abstract
A library of quinazolinones was prepared by the one-pot three-component reaction of isatoic anhydride, ammonium salts/amines, and various electrophiles using aqueous hydrotropic solution as an efficient, economical, reusable, and green medium giving good to excellent yields of products in very short time. The method offers a versatile way for the development of diversity-oriented synthesis of quinazolinones.






Similar content being viewed by others
References
Anastas P, Eghbali N (2010) Green chemistry: principles and practice. Chem Soc Rev 39:301–312. doi:10.1039/B918763B
Li CJ, Chen L (2006) Organic chemistry in water. Chem Soc Rev 35:68–82. doi:10.1039/B507207G
Li CJ (2005) Organic reactions in aqueous media with a focus on carbon–carbon bond formations: a decade update. Chem Rev 105:3095–3165. doi:10.1021/cr030009u
Vemula VR, Lagishetty V, Lingala S (2010) Solubility enhancement techniques. Int J Pharm Sci Rev Res 5:41–51
Neuberg C (1916) Hydrotropic phenomena. Biochem Z 76:107–176
McKee RH (1946) Use of hydrotropic solutions in industry. Ind Eng Chem 38:382–384. doi:10.1021/ie50436a012
Kumbhar A, Kamble S, Jadhav S, Rashinkar G, Salunkhe R (2012) Silica tethered Pd-DABCO complex: an efficient and reusable catalyst for Suzuki-Miyaura reaction. Catal Lett 142:1388–1396. doi:10.1007/s10562-012-0912-3
Kurane R, Jadhav J, Khanapure S, Salunkhe R, Rashinkar G (2013) Synergistic catalysis by an aerogel supported ionic liquid phase (ASILP) in the synthesis of 1,5-benzodiazepines. Green Chem 15:1849–1856. doi:10.1039/c3gc40592c
Hayakawa M, Kaizawa H, Moritomo H, Koizumi T, Ohishi T, Okada M, Ohta M, Tsukamoto S, Parker P, Workman P, Waterfield M (2006) Synthesis and biological evaluation of 4-morpholino-2-phenylquinazolines and related derivatives as novel PI3 kinase p110\(\upalpha \) inhibitors. Bioorg Med Chem 14:6847–6858. doi:10.1016/j.bmc.2006.06.046
Noolvi M, Patel H, Bhardwaj V, Chauhan A (2011) Synthesis and in vitro antitumor activity of substituted quinazoline and quinoxaline derivatives : search for anticancer agent. Eur J Med Chem 46:2327–2346. doi:10.1016/j.ejmech.2011.03.015
Ashton W, Hynes J (1973) Synthesis of 5-substituted quinazolines as potential antimalarial agents. J Med Chem 16:1233–1237. doi:10.1021/jm00269a005
Amir M, Ali I, Hasan M (2013) Design and synthesis of some new quinazolin-4-\((3H)\)-ones as anticonvulsant and antidepressant agents. Arch Pharm Res 36:61–68. doi:10.1007/s12272-013-0004-y
Manhas MS, Hoffman WA, Bose AK (2009) Heterocyclic compounds XII. Quinazoline derivatives as potential antifertility agents. J Heterocycl Chem 16:711–715. doi:10.1002/jhet.5570160420
Kung P, Casper M, Cook K, Wilson-Lingardo L, Risen LM, Vickers TA, Ranken R, Blyn L, Wyatt JR, Cook PD, Ecker DJ (1999) Structure-activity relationships of novel 2-substituted quinazoline antibacterial agents. J Med Chem 42:4705–4713. doi:10.1021/jm9903500
Xu G, Song B, Bhadury PS, Yang S, Zhang P, Jin L, Xue W, Hu D, Lu P (2007) Synthesis and antifungal activity of novel \(s\)-substituted 6-fluoro-4-alkyl(aryl)thioquinazoline derivatives. Bioorg Med Chem 15:3768–3774. doi:10.1016/j.bmc.2007.03.037
Potewar TM, Ingale SA, Srinivasan KV (2008) Synthesis of tryptanthrin and deoxyvasicinone by a regioselective lithiation-intramolecular electrophilic reaction approach. ARKIVOC xiv:100–108
Lednicer D, Mitscher LA (1977) The organic chemistry of drug synthesis, vol 1. A Wiley-Interscience Publication, New York
Larksarp C, Alper H (2000) Palladium-catalyzed cyclocarbonylation of \(o\)-iodoanilines with heterocumulenes: regioselective preparation of \(4(3H)\)-quinazolinone derivatives. J Org Chem 65:2773–2777. doi:10.1021/jo991922r
Beccalli EM, Broggini G, Paladino G, Penoni A, Zoni C (2004) Regioselective formation of six- and seven-membered ring by intramolecular Pd-catalyzed amination of \(N\)-allyl-anthranilamides. J Org Chem 69:5627–5630. doi:10.1021/jo0495135
Zhang X, Ye D, Sun H, Guo D, Wang J, Huang H, Zhang X, Jiang H, Liu H (2009) Microwave-assisted synthesis of quinazolinone derivatives by efficient and rapid iron-catalyzed cyclization in water. Green Chem 11:1881–1888. doi:10.1039/b916124b
Broggini G, Borsini E, Fasana A, Poli G, Liron F (2012) Transition-metal-catalyzed hydroamination and carboamination reactions of anthranilic allenamides as a route to 2-vinyl- and 2-(\(\upalpha \)-styryl)quinazolin-4-one derivatives. Eur J Org Chem 19:3617–3624. doi:10.1002/ejoc.201200353
Alizadeh A, Ghanbaripour R, Zhu L (2014) An efficient approach to the synthesis of coumarin-bearing 2,3-dihydro-4\((1H)\)-quinazolinone derivatives using a piperidine and molecular iodine dual-catalyst system. Synlett 25:1596–1600. doi:10.1055/s-0033-1341202
He L, Li H, Chen J, Wu X (2014) Recent advances in \(4(3H)\)-quinazolinone syntheses. RSC Adv 4:12065–12077. doi:10.1039/c4ra00351a
Dabiri M, Salehi P, Otokesh S, Baghbanzadeh M, Kozehgary G, Mohammadi A (2005) Efficient synthesis of mono- and disubstituted 2,3-dihydroquinazolin-4\((1H)\)-ones using KAl\(({\rm SO}_{4})_{2}. {\rm H}_{2}{\rm O}\) as a reusable catalyst in water and ethanol. Tetrahedron Lett 46:6123–6126. doi:10.1016/j.tetlet.2005.06.157
Dabiri M, Salehi P, Baghbanzadeh M, Zolfigol MA, Agheb M, Heydari S (2008) Silica sulfuric acid: An efficient reusable heterogeneous catalyst for the synthesis of 2,3-dihydroquinazolin-4\((1H)\)-ones in water and under solvent-free conditions. Catal Commun 9:785–788. doi:10.1016/j.catcom.2007.08.019
Salehi P, Dabiri M, Baghbanzadeh M, Bahramnejad M (2006) One-pot, three-component synthesis of 2,3-dihydro-4\((1H)\)-quinazolinones by Montmorillonite K-10 as an efficient and reusable catalyst. Synth Commun 36:2287–2292. doi:10.1080/00397910600639752
Dabiri M, Salehi P, Bahramnejad M, Alizadeh M (2010) A practical and versatile approach toward a one-pot synthesis of 2,3-disubstituted \(4(3H)\)-quinazolinones. Monatsh Chem 141:877–881. doi:10.1007/s00706-010-0341-1
Zhang Z, Lu H, Yang S, Gao J (2010) Synthesis of 2,3-dihydroquinazolin-4\((1H)\)-ones by three-component coupling of isatoic anhydride, amines, and aldehydes catalyzed by magnetic \({\rm Fe}_{3}{\rm O}_{4}\) nanoparticles in water. J Comb Chem 12:643–646. doi:10.1021/cc100047j
Yavari I, Beheshti S (2011) ZnO nanoparticles catalyzed efficient one-pot three-component synthesis of 2,3-disubstituted quinazolin-4\((1H)\)-ones under solvent-free conditions. J Iran Chem Soc 8:1030–1035. doi:10.1007/BF03246559
Chen J, Wu D, He F, Liu M, Wu H, Ding J, Su W (2008) Gallium(III) triflate-catalyzed one-pot selective synthesis of 2,3-dihydroquinazolin-4\((1H)\)-ones and quinazolin-4\((3H)\)-ones. Tetrahedron Lett 49:3814–3818. doi:10.1016/j.tetlet.2008.03.127
Surpur MP, Singh PR, Patil SB, Samant SD (2007) Expeditious one-pot and solvent-free synthesis of dihydroquinazolin-4\((1H)\)-ones in the presence of microwaves. Synth Commun 37:1965–1970. doi:10.1080/00397910701354699
Wang L, Hu L, Shao J, Yu J, Zhang L (2008) A novel catalyst zinc(II) perfluorooctanoate \([{\rm Zn(PFO)}_{2}]\)-catalyzed three-component one-pot reaction: Synthesis of quinazolinone derivatives in aqueous micellar media. J Fluorine Chem 129:1139–1145. doi:10.1016/j.jfluchem.2008.08.005
Burke MD, Schreiber SL (2004) A planning strategy for diversity-oriented synthesis. Angew Chem Int Ed 43:46–58. doi:10.1002/anie.200300626
Posner GH (1986) Multicomponent one-pot annulations forming three to six bonds. Chem Rev 86:831–844. doi:10.1021/cr00075a007
Estevez V, Villacampa M, Menendez C (2010) Multicomponent reactions for the synthesis of pyrroles. Chem Soc Rev 39:4402–4421. doi:10.1039/b917644f
Zhu S, Ji S, Su X, Sun C, Liu Y (2008) Facile and efficient synthesis of a new class of bis(3\(^\prime \)-indolyl)pyridine derivatives via one-pot multicomponent reactions. Tetrahedron Lett 49:1777–1781. doi:10.1016/j.tetlet.2008.01.054
Kamble SB, Kumbhar AS, Rashinkar GS, Barge MS, Salunkhe RS (2012) Ultrasound promoted efficient and green synthesis of \(\beta \)-amino carbonyl compounds in aqueous hydrotropic medium. Ultrason Sonochem 19:812–815. doi:10.1016/j.ultsonch.2011.12.001
Barge MS, Kamble SB, Kumbhar AS, Rashinkar GS, Salunkhe RS (2013) Hydrotrope: green and rapid approach for the catalyst-free synthesis of pyrazole derivatives. Monatsh Chem 144:1213–1218. doi:10.1007/s00706-013-0944-4
Saffar-Teluri A, Bolouk S (2010) One-pot, three-component synthesis of 2,3-dihydroquinazolin-4\((1H)\)-ones using \(p\)-toluenesulfonic acid-paraformaldehyde copolymer as an efficient and reusable catalyst. Monatsh Chem 141:1113–1115. doi:10.1007/s00706-010-0376-3
Darvatkar NB, Bhilare SV, Deorukhkar AR, Raut DG, Salunkhe MM (2010) [bmim]\({\rm HSO}_{4}\): an efficient and reusable catalyst for one-pot three-component synthesis of 2,3-dihydro-4\((1H)\)-quinazolinones. Green Chem Lett Rev 3:301–306. doi:10.1080/17518253.2010.485581
Karimi-Jaberi Z, Arjmandi R (2011) Acetic acid-promoted, efficient, one-pot synthesis of 2,3-dihydroquinazolinone-4\((1H)\)-ones. Monatsh Chem 142:631–635. doi:10.1007/s00706-011-0494-6
Mohammadi AA, Rohi H, Soorki AA (2013) Synthesis and In Vitro antibacterial activities of novel 2-aryl-3-(phenylamino)-2,3-dihydroquinazolin-4\((1H)\)-one derivatives. J Heterocycl Chem 50:1129–1133. doi:10.1002/jhet.1075
Baghbanzadeh M, Salehi P, Dabiri M, Kozehgary G (2006) Water-accelerated synthesis of novel bis-2,3-dihydroquinazolin-4\((1H)\)-one. Synthesis 2:344–348. doi:10.1055/s-2005-924766
Dabiri M, Salehi P, Mohammadi AA, Baghbanzadeh M (2005) One-pot synthesis of mono- and disubstituted \((3H)\)-quinazolin-4-ones in dry media under microwave irradiation. Synth Commun 35:279–287. doi:10.1081/SCC-200048462
Khosropour AR, Mohammadpoor-Baltork I, Gharbankhani H (2006) \({\rm Bi(TFA)}_{3}\text{- }{\rm [nbp]}{\rm FeCl}_{4}\): A new, efficient and reusable promoter system for the synthesis of \(4(3H)\)-quinazolinone derivatives. Tetrahedron Lett 47:3561–3564. doi:10.1016/j.tetlet.2006.03.079
Shokrolahi A, Zali A, Zarei M, Esmaeilpour K (2012) Sulfonated porous carbon (SPC): an efficient and recyclable solid acid catalyst for one-pot three-component synthesis of 2,3-dihydroquinazolin-4\((1H)\)-ones under solvent-free conditions. Iran J Catal 2:91–94
Furnis B, Hannaford A, Smith P, Tatchell A (1996) Vogel’s textbook of practical organic chemistry. Prentice Hall, Upper Saddle River
Acknowledgments
Authors R. S. S. and A. A. P. thank UGC, New Delhi for financial assistance [F. No. 41-310/2012 (SR)] and for the research fellowship, respectively.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Patil, A., Barge, M., Rashinkar, G. et al. Aqueous hydrotrope: an efficient and reusable medium for a green one-pot, diversity-oriented synthesis of quinazolinone derivatives. Mol Divers 19, 435–445 (2015). https://doi.org/10.1007/s11030-015-9580-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-015-9580-8