Abstract
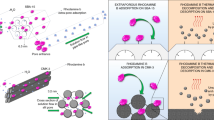
This study describes the surface modification of porous Vycor glass® (PVG®) with ionic species based on 1-methyl-imidazolium groups, and the posterior utilization of the functional porous substrate to synthesize supported gold nanoparticles (Au NPs). The functionalization degree (amount of imidazolium groups per gram of PVG®) was successfully controlled by the concentration of the imidazolium-based alkoxysilane in the reaction media. The ion-exchange properties of the functional groups allowed an effective and homogeneous upload of AuCl4 − throughout the monolithic PVG® structure, with subsequent conversion to Au NPs by thermal treatment at 823 K, which also eliminated the organic groups chemically bonded to the surface. The amount of adsorbed AuCl4 − ions was shown to have influence on loading of the Au NPs while no size variation is observed, indicating that the porous environment of PVG® acts as a nanoreactor where the particles are formed in the confined space.











Similar content being viewed by others
References
Alfaya RVS, Fujiwara ST, Gushikem Y, Kholin YV (2004) Adsorption of metal halides from ethanol solutions by a 3-n-propylpyridiniumsilsesquioxane chloride-coated silica gel surface. J Colloid Interface Sci 269:32–36. doi:10.1016/S0021-9797(03)00611-8
Berg RW (2007) Raman spectroscopy and ab initio model calculations on ionic liquids. Mon Chem 138:1045–1075. doi:10.1007/s00706-007-0760-9
Caiut JMA, Nakagaki S, Friedermann GR, Drechsel SM, Zarbin AJG (2004) Nickel(II) and manganese(III) tetraazaannulenes complexes encapsulated in porous Vycor glass (PVG): investigation of catalytic activity. J Mol Catal A 222:213–222. doi:10.1016/j.molcata.2004.08.011
Cangussu D, Nunes WC, Pereira CLM, Pedroso EF, Mazali IO, Knobel M, Alves OL, Stumpf HO (2008) Slow magnetic relaxation in (CoCuII)-Cu-II coordination oligomer built into mesoporous material. Eur J Inorg Chem 24:3802–3808. doi:10.1002/ejic.200800330
Cangussu D, Nunes WC, Corrêa HLS, Macedo WAA, Knobel M, Alves OL, Filho AGS, Mazali IO (2009) γ-Fe2O3 nanoparticles dispersed in porous Vycor glass: a magnetically diluted integrated system. J Appl Phys 105:013901. doi:10.1063/1.3054173
Carneiro NM, Nunes WC, Borges RP, Godinho M, Fernandez-Outon LE, Macedo WAA, Mazali IO (2010) NiO nanoparticles dispersed in mesoporous silica glass. J Phys Chem C 114:18773–18778. doi:10.1021/jp1046886
Chen H, Kou X, Yang Z, Ni W, Wang J (2008) Shape- and size-dependent refractive index sensitivity of gold nanoparticles. Langmuir 24:5233–5237. doi:10.1021/la800305j
Correa DN, Silva JMDE, Santos EB, Sigoli FA, Souza Filho AG, Mazali IO (2011) TiO2- and CeO2-based biphasic core-shell nanoparticles with tunable core sizes and shell thicknesses. J Phys Chem C 115:10380–10387. doi:10.1021/jp200540g
Corning Glass datasheet (2012) VYCOR® Brand Porous Glass 7930 datasheet. Corning Inc. http://www.corning.com/docs/specialtymaterials/pisheets/Vycor%207930.pdf. Accessed 11 Sep 2012
Daniel MC, Astruc D (2004) Gold nanoparticles: assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem Rev 104:293–346. doi:10.1021/cr030698+
Fattori N, Maroneze CM, Da Costa LP, Strauss M, Sigoli FA, Mazali IO, Gushikem Y (2012) Ion-exchange properties of imidazolium-grafted SBA-15 toward AuCl4 − anions and their conversion into supported gold nanoparticles. Langmuir 28:10281–10288. doi:10.1021/la301374z
Fujiwara ST, Gushikem Y, Alfaya RVS (2001) Adsorption of FeCl3, CuCl2 and ZnCl2 on silsesquioxane 3-n-propylpyridiniumchloride polymer film adsorbed on Al2O3 coated silica gel. Colloids Surf A 178:135–141. doi:10.1016/S0927-7757(00)00685-3
Iamamoto MS, Gushikem Y (1989) Adsorption and pre-concentration of some metal ions from ethanol on silica gel modified with pyridinium ion. Analyst 114:983–985. doi:10.1039/AN9891400983
Jeon Y, Sung J, Seo C, Lim H, Cheong H, Kang M, Moon B, Ouchi Y, Kim D (2008) Structures of ionic liquids with different anions studied by infrared vibration spectroscopy. J Phys Chem B 112:4735–4740. doi:10.1021/jp7120752
Kelly KL, Coronado E, Zhao LL, Schatz GC (2003) The optical properties of metal nanoparticles: the influence of size, shape, and dielectric environment. J Phys Chem B 107:668–677. doi:10.1021/jp026731y
Lowell S, Shields JE, Thomas MA, Thommes M (2006) Characterization of porous solids and powders: surface area. Pore size and density. Springer, Dordrecht
Maroneze CM, Magosso HA, Panteleimonov AV, Kholin YV, Gushikem Y (2011) Surface functionalization of SBA-15 and a nonordered mesoporous silica with a 1,4-diazabicyclo[2.2.2]octane derivative: study of CuCl2 adsorption from ethanol solution. J Colloid Interface Sci 356:248–256. doi:10.1016/j.jcis.2010.12.030
Mazali IO, Souza Filho AG, Viana BC, Mendes Filho J, Alves OL (2006) Size-controllable synthesis of nanosized-TiO2 anatase using porous Vycor glass as template. J Nanopart Res 8:141–148. doi:10.1007/s11051-005-9003-3
Mie GVG (1907) Die optischen Eigenschaften kolloider Goldlösungen. Ann Phys 25:129–133
Rouquerol F, Rouquerol J, Sing KSW (1999) Adsorption by powders and porous solids. Academic Press, San Diego
Santos EB, Sigoli FA, Mazali IO (2012) Structural evolution in crystalline MoO3 nanoparticles with tunable size. J Solid State Chem 190:80–84. doi:10.1016/j.jssc.2012.02.012
Sing KSW, Everett DH, Haul RAW, Moscou L, Pierotti RA, Rouquerol J, Siemieniewska T (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl Chem 57:603–619. doi:10.1351/pac198557040603
Stammreich H, Forneris R (1960) Raman spectra and force constants of planar square molecules of the type AB4. Spectrochim Acta 16(3):363–367. doi:10.1016/0371-1951(60)80097-5
Strauss M, Destefani TA, Sigoli FA, Mazali IO (2011) Crystalline SnO2 nanoparticles size probed by Eu3+ luminescence. Cryst Growth Des 11:4511–4516. doi:10.1021/cg2007292
Usher A, McPhail DC, Brugger J (2009) A spectrophotometric study of aqueous Au(III) halide–hydroxide complexes at 25–80 °C. Geochim Cosmochim Acta 73:3359–3380. doi:10.1016/j.gca.2009.01.036
Wada Y, Kaneko M, Niinobe D, Tsukahara Y (2005) In situ synthesis of Ag2S nanosized particles in porous materials using single-source precursor. Chem Lett 34(12):1618–1619. doi:10.1246/cl.2005.1618
Wada Y, Niinobe D, Kaneko M, Tsukahara Y (2006) Synthesis of Cd x Zn1−x S nanoparticles in porous Vycor glass by reaction of single-source precursors. Chem Lett 35(1):62–63. doi:10.1246/cl.2006.62
Acknowledgments
JPVD, MS, CMM, and LPC are indebted to CNPq and CAPES for fellowships. IOM, FAS and YG are indebted to CNPq and FAPESP for financial support and to Prof. C. H. Collins (IQ-UNICAMP, Brazil) for English revision. Contributions from LNNano (Brazilian Nanotechnology National Laboratory) for TEM imagens and the Multiuser Laboratory of Advanced Optical Spectroscopy (LMEOA/IQ-UNICAMP) for use of its equipment are also gratefully acknowledged. This work is a contribution from the National Institute of Science and Innovation in Complex Functional Materials (INOMAT).
Author information
Authors and Affiliations
Corresponding author
Additional information
Special Issue Editors: Juan Manuel Rojo, Vasileios Koutsos
This article is part of the topical collection on Nanostructured Materials 2012
This paper is dedicated to Professor Fernando Galembeck on the occasion of this 70th anniversary.
Rights and permissions
About this article
Cite this article
Strauss, M., Damasceno, J.P.V., Maroneze, C.M. et al. Exploring the functionalization ratio of mesoporous silica glass with imidazolium entities on the synthesis of supported gold nanoparticles. J Nanopart Res 15, 1441 (2013). https://doi.org/10.1007/s11051-013-1441-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-013-1441-8