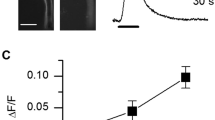
Behavioral and histochemical studies demonstrated that endogenous tryptophan metabolites – kynurenines – play a role in the long-term retention of memory traces and the functioning of key components in the (GluR–LIMK1– F-actin) signaling cascade, which mediates these functions. Kynurenine deficiency induced by injections of allopurinol (a tryptophan oxygenase inhibitor) inhibited long-term memory, decreased the level of expression of LIMK1, and produced a paradoxical increase in the F-actin level in the cerebral ganglion in bees. These results are consistent with our previous data obtained in Drosophila with a mutation in the structural gene for tryptophan oxygenase, i.e., the vermilion mutation.
Similar content being viewed by others
References
I. P. Lapin, “The kynurenine pathway of tryptophan metabolism and its role in nervous system function and in the actions of psychotropic drugs,” Zh. Khim. Vses. Obshch. D. I. Mendeleeva, 21, No. 2, 151–157 (1976).
N. G. Lopatina, T. G. Zachepilo, E. G. Chesnokova, and E. V. Savvateeva- Popova, “Mutations in the structural genes for enzymes involved in the kynurenine pathway of tryptophan metabolism in the modulation of components of the signal cascade – glutamate receptors and cytoskeletal actin,” Genetika, 43, No. 10, 1396–1401 (2007).
N. G. Lopatina, T. G. Zachepilo, and E. V. Savvateeva-Popova, “LIM kinase 1 in the cerebral ganglion in Drosophila in condition of genetic lesions to the balance of kynurenines,” Dokl. Ros. Akad. Nauk., 418, No. 1, 125–128 (2008).
N. G. Lopatina, E. G. Chesnokova,V. B. Smirnov, I. V. Ryzhova, and V. V. Ponomarenko, “The kynurenine pathway of tryptophan metabolism and its importance in the neurophysiology of insects,” Entomol. Obozr., 83, No. 1, 3–22 (2004).
I. V. Ryzhova, N. G. Lopatina, and E. G. Chesnokova, “Excitatory amino acid receptors in associative learning in the honeybee Apis mellifera L.,” Tr. Russk. Entomol. Obshch., 74, 17–32 (2003).
M. Alkondon, E. Pereira, and P. Yu, “Targeted deletion of the kynurenine aminotransferase II gene reveals a critical role of endogenous kynurenic acid in the regulation of synaptic transmission via α7 nicotinic receptors in the hippocampus,” J. Neurosci., 24, No. 19, 4635–4648 (2004).
N. Cairns, V. Lee, and J. Trojanowski, “The cytoskeleton in neurodegenerative diseases,” J. Pathol., 204, 438–449 (2004).
L. Chen, C. Rex, and M. Casale, “Changes in synaptic morphology accompany actin signaling during LTP,” J. Neurosci., 27, No. 20, 5563–5372 (2007).
L. Cingolani and Y. Goda, “Actin in action: the interplay between the actin cytoskeleton and synaptic efficacy,” Neurosci., 9, 344–356 (2008).
C. Dillon and Y. Goda, “The actin cytoskeleton: integrating form and function at the synapse,” Ann. Rev. Neurosci., 28, 25–56 (2005).
I. Frambach,W. Rossler, M. Winkler, and F. Schurmann, “F-actin at identified synapses in the mushroom body neuropil of the insect brain,” J. Comp. Neurol., 475, No. 3, 303–314 (2004).
Y. Meng,Y. Zhang, and V. Tregoubov, “Abnormal spine morphology and enhanced LTP in LIMK/knockout mice,” Neuron, 35, 121–133 (2002).
H. Nemeth, J. Toidi, and L. Veesei, “Role of kynurenines in the central and peripheral nervous systems,” Curr. Neurovasc. Res., 2, No. 3, 249–260 (2005).
R. H. Osborne, “Insect neurotransmission: neurotransmitters and their receptors,” Pharmacol. Ther., 69, 117–142 (1996).
M. Sapko, P. Guidetti, and P. Yu, “Endogenous kynurenate controls the vulnerability of striatal neurons to quinolinate: implications for Huntington’s disease,” Exptl. Neurol., 197, No. 1, 31–40 (2006).
R. Schwarcz, P. Rassoulpour, and H.-Q. Wu, “Increased cortical kynurenate content in schizophrenia,” Biol. Psych., 50, 521–530 (2001).
T. Tada and M. Sheng, “Molecular mechanisms of dendritic spine morphogenesis,” Curr. Opin. Neurobiol., 16, No. 1, 95–101 (2006).
P. Yu, N. Di Prospero, and M. Sapko, “Biochemical and phenotypic abnormalities in kynurenine aminotransferase II-deficient mice,” Mol. Cell. Biol., 24, No. 16, 6919–6930 (2004).
P. Yu, N. Mosbrook, and D. Tagle, “Genomic organization and expression analysis of mouse kynurenine aminotransferase II, a possible factor in the pathophysiology of Huntington’s disease,” Mamm. Genome, 10, 845–852 (1999).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Vysshei Nervnoi Deyatel’nosti imeni I. P. Pavlova, Vol. 60, No. 2, pp. 229–235, March–April, 2010.
Rights and permissions
About this article
Cite this article
Lopatina, N.G., Zachepilo, T.G., Chesnokova, E.G. et al. Behavioral and Molecular Consequences of Deficiency of Endogenous Kynurenines in Honeybees (Apis mellifera L.). Neurosci Behav Physi 41, 626–631 (2011). https://doi.org/10.1007/s11055-011-9465-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-011-9465-y