Abstract
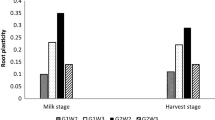
A greenhouse experiment was conducted to evaluate root plasticity and drought responses of Catalpa bungei under partial root-zone drought as affected by nitrogen (N) forms. A two-factor experimental design was used in which two nitrogen forms (ammonium nitrogen (AN) and nitrate nitrogen (NN)) were combined with four water treatments (well-watered condition (WW), vertical partial root-zone drought (V-PRD), horizontal partial root-zone drought (H-PRD), full root-zone drought (FRD)). Root parameters were interactively affected by water conditions and N forms. Root development was sharply inhibited by FRD, and nitrate (NN) was more beneficial than ammonium (AN) for lateral root elongation under FRD. Root responded to PRDs with changing of their morphological plasticity, depending on the supplied N forms. Moreover, root system exhibited contrasting strategy for water foraging under V-PRD and H-PRD. Under V-PRD, root forage for water by hydrotropism, which was indicated by active root proliferation in moist soil zone. This strategy contributed to the efficient carbohydrate production and dominant xylem development under V-PRD. In contrast, root forage for water under H-PRD by significant root proliferation in the drought soil zone. The absence of horizontal hydrotropism under H-PRD condition and the presence of vertical hydrotropism under V-PRD was related to the gravitropism action. Physiological plasticity under PRDs might be preliminary disclosed by the specific activation of antioxidant defense in the drought root zone. Specifically, APX, CAT and SOD were induced in the drought root zone (top zone) of V-PRD, and APX was induced in the drought root zone (right zone) of H-PRD. Root physiological plasticity under PRDs was related to N forms and it displayed contrasting patterns in NN and AN treatment.







Similar content being viewed by others
References
Ahmad S, Raza MAS, Saleem FM, Iqbal R, Zaheer MS, Haider I, Aslam MU, Ali M, Khan H (2020) Significance of partial root zone drying and mulches for water saving and weed suppression in wheat. J Anim Sci 30:154–162
Albert B, Cahérec FL, Niogret MF, Faes P, Avice JC, Leport L, Bouchereau A (2012) Nitrogen availability impacts oilseed rape (Brassica napus L.) plant water status and proline production efficiency under water-limited conditions. Planta 236:659–676
Alt DS, Doyle JW, Malladi A (2017) Nitrogen-source preference in blueberry (vaccinium sp.): Enhanced shoot nitrogen assimilation in response to direct supply of nitrate. J Plant Physiol 216:79–87
Battal P, Erez ME, Turker M, Berber I (2008) Molecular and physiological changes in maize (Zea mays) induced by exogenous NAA, ABA and MeJa during cold stress. Ann Bot Fenn 45:173–185
Cantó CF, Simonin M, King E, Moulin L, Bennett MJ, Castrillo G, Laplaze L (2020) An extended root phenotype: the rhizosphere, its formation and impacts on plant fitness. Plant J 103:951–964
Cao X, Jia J, Zhang C, Li H, Liu T, Jiang X, Luo ZB (2014) Anatomical, physiological and transcriptional responses of two contrasting poplar genotypes to drought and re-watering. Physiol Plant 151:480–494
Chen LH, Han Y, Jiang H, Korpelainen H, Li C (2011) Nitrogen nutrient status induces sexual differences in responses to cadmium in Populus yunnanensis. J Exp Bot 62:5037–5050
Cheng LB, Zhao MR, Hu ZB, Liu HY, Li SY (2020) Comparative transcriptome analysis revealed the cooperative regulation of sucrose and IAA on adventitious root formation in lotus (Nelumbo nucifera Gaertn). BMC Genomics 21:653
Dekker LW, Doerr SH, Oostindie K, Ziogas AK, Ritsema (2001) Water repellency and critical soil water content in a dune sand. Soil Sci Soc Am J 65:1667–1674
Demirkol G (2021) Popw enhances drought stress tolerance of alfalfa via activating antioxidative enzymes, endogenous hormones, drought related genes and inhibiting senescence genes. Plant Physio Bioch 166:540–548
Dietrich D (2018) Hydrotropism: how roots search for water. J Exp Bot 69:11
Dietrich D, Pang L, Kobayashi A, Fozard JA, Boudolf V, Bhosale R, Antoni R, Nguyen T, Hiratsuka S, Fujii N, Miyazawa Y, Bae TW, Wells DM, Owen MR, Band LR, Dyson RJ, Jensen OE, King JR, Tracy SR, Sturrock CJ, Mooney SJ, Roberts JA, Bhalerao RP, Dinneny JR, Rodriguez PL, Nagatani A, Hosokawa Y, Baskin TI, Pridmore TP, Veylder LD, Takahashi H, Bennett MJ (2017) Root hydrotropism is controlled via a cortex-specific growth mechanism. Nat Plants 3:17057
Du H, Wu N, Chang Y, Li XH, Xiao JH, Xiong LZ (2013) Carotenoid deficiency impairs ABA and IAA biosynthesis and differentially affects drought and cold tolerance in rice. Plant Mol Biol 83:475–488
Escobar MA, Geisler DA, Rasmusson AG (2010) Reorganization of the alternative pathways of the arabidopsis respiratory chain by nitrogen supply: opposing effects of ammonium and nitrate. Plant J 45:775–788
Farooqi AHA, Fatima S, Khan A, Sharma S (2005) Ameliorative effect of chlormequat chloride and IAA on drought stressed plants of Cymbopogon martinii and C. winterianus. Plant Growth Regul 46:277–284
Ghafari H, Hassanpour H, Jafari M, Besharat S (2020) Physiological, biochemical and gene-expressional responses to water deficit in apple subjected to partial root-zone drying (PRD). Plant Physiol Bioch 148:333–346
Gruber BD, Giehl RFH, Swetlana F, Nicolaus VW (2013) Plasticity of the Arabidopsis root system under nutrient deficiencies. Plant Physiol 163:161–179
Guo S, Zhou Y, Shen Q, Zhang F (2005) Effect of ammonium and nitrate nutrition on some physiological processes in higher plants-growth, photosynthesis, photorespiration, and water relations. Plant Biol 9:21–29
He JL, Qin JJ, Long LY, Ma YL, Li H, Li K, Jiang XN, Liu TX, Polle A, Liang ZS, Luo ZB (2011) Net cadmium flux and accumulation reveal tissue-specific oxidative stress and detoxification in Populus × canescens. Physiol Plant 143:50–63
Hideki Y, Eiichi T, Takaaki H, Yohei M, Rie M, Mayuko K, Mitsuhiro T, Sayaka T, Ko H, Masatsune KI, Takashi A, Makoto M, Miyako UT (2018) Evolution and diversification of the plant gibberellin receptor GID1. Proc Natl Acad Sci USA 115:E7844–E7853
Horváth E, Szalai G, Janda T (2007) Induction of abiotic stress tolerance by salicylic acid signaling. J Plant Growth Regul 26:290–300
Iqbal R, Raza MAS, Saleem MF, Khan IH, Ahmad S, Zaheer MS, Aslam MU, Haider I (2019) Physiological and biochemical appraisal for mulching and partial rhizosphere drying of cotton. J Arid Land 11:785–794
Kagenishi T, Yokawa K, Baluška F (2016) Mes buffer affects arabidopsis root apex zonation and root growth by suppressing superoxide generation in root apex. Front Plant Sci 7:79
Kang SZ, Hu XT, Goodwin I, Jerie P (2002) Soil water distribution, water use, and yield response to partial root zone drying under a shallow groundwater table condition in a pear orchard. Sci Hortic-Amsterdam 92:277–291
Kang SZ, Liang ZS, Pan YH, Shi PZ, Zhang JH (2000) Alternate furrow irrigation for maize production in arid area. Agric Water Manage 45:267–274
Kang SZ, Zhang J (2001) Controlled alternate partial root-zone irrigation: its physiological consequences and impact on water use efficiency. Acta Botan Boreali-Occiden Sin 55:2437–2446
Kang SZ, Zhang L, Hu XT, Li ZJ, Jerie P (2001) An improved water use efficiency for hot pepper grown under controlled alternate drip irrigation on partial roots. Sci Hortic-Amsterdam 89:257–267
Kaushik D, Aryadeep R (2014) Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front Environ Sci 2
Kunert KJ, Vorster BJ, Tsholofelo K, Giuseppe D, Foyer CH (2016) Drought stress responses in soybean roots and nodules. Front Plant Sci 7:1015
Lecube ML, Noriega GO, Cruz DMS, Tomaro ML, Balestrasse KB (2014) Indole acetic acid is responsible for protection against oxidative stress caused by drought in soybean plants: the role of heme oxygenase induction. Redox Rep 19:242–250
Li Y, Yuan W, Li LC, Dai H, Dang XL, Miao R, Baluška F, Kronzucker HJ, Lu CM, Zhang JH, Xu WF (2020) Comparative analysis reveals gravity is involved in the MIZ1-regulated root hydrotropism. J Exp Bot 22
Lima JE, Kojima S, Takahashi H, Wirén NV (2010) Ammonium triggers lateral root branching in Arabidopsis in an ammonium transporter1;3-dependent manner. Plant Cell 22:3621–3633
Liu XX, Zhang HH, Zhu QY, YeJY ZhuYX, Jiang XT, Du WX, Zhou M, Lin XY, Zheng SJ, Jin CW (2022) Phloem iron remodels root development in response to ammonium as the major nitrogen source. Nat Commun 13:561
Luo ZB, Li K, Jiang XN, Polle A (2009) Ectomycorrhizal fungus (paxillus involutus) and hydrogels affect performance of populus euphratica exposed to drought stress. Ann Forest Sci 66:106
McLean EH, Ludwing M, Grierson PF (2011) Root hydraulic conductance and aquaporin abundance respond rapidly to partial root-zone drying events in a riparian melaleuca species. New Phytol 192:664–675
Miller AJ, Fan X, Orsel M, Smith SJ, Wells DM (2007) Nitrate transport and signaling. J Exp Bot 58:2297–2306
Miyazawa Y, Sakashita T, Funayama T, Hamada N, Negishi H, Kobayashi A, Kaneyasu T, Ooba A, Morohashi K, Kakizaki T, Wada S, Kobayashi Y, Fujii N, Takahashi H (2008) Effects of locally targeted heavy-ion and laser microbeam on root hydrotropism in Arabidopsis thaliana. J Radiat Res 49:373–379
Morohashi K, Okamoto M, Yamazaki C, Fujii N, Miyazawa Y, Kamada M, Kasahara H, Osada I, Shimazu T, Fusejima Y, Higashibata A, Yamazaki T, Ishioka N, Kobayashi A, Takahashi H (2017) Gravitropism interferes with hydrotropism via counteracting auxin dynamics in cucumber roots: clinorotation and spaceflight experiments. New Phytol 215:1476–1489
Müller LM (2020) NIT proteins regulate rice root plasticity in response to nitrate and ammonium. Plant Physiol 183:25–26
Nakajima Y, Nara Y, Kobayashi A, Sugita T, Miyazawa Y, Fujii N, Takahashi H (2017) Auxin transport and response requirements for root hydrotropism differ between plant species. J Exp Bot 68:3441–3456
Paz H, Pineda-García F, Pinzón-Pérez LF (2015) Root depth and morphology in response to soil drought: comparing ecological groups along the secondary succession in a tropical dry forest. Oecologia 179:551–561
Pierik R, Testerink C (2014) The art of being flexible: how to escape from shade, salt, and drought. Plant Physiol 166:5–22
Prince SJ, Song L, Qiu D, Santos JVM, Chai CL, Joshi T, Patil G, Valliyodan B, Vuong TD, Murphy M, Krampis K, Tucker DM, Biyashev R, Dorrance AE, Maroof MAS, Xu D, Shannon JG, Nguyen HT (2015) Genetic variants in root architecturerelated genes in a Glycine soja accession, a potential resource to improve cultivated soybea. BMC Genomics 16:132
Pustovoitova TN, Drozdova IS, Zhdanova NE, Zholkevich VN (2003) Leaf growth, photosynthetic rate, and phytohormone contents in cucumis sativus plants under progressive soil drought. Russ J Plant Physiol 50:441–443
Ravazzolo L, Trevisan S, Forestan C, Varotto S, Sut S, Accqua SD, Malagoli M, Quaggiotti S (2020) Nitrate and ammonium affect the overall maize response to nitrogen availability by triggering specific and common transcriptional signatures in roots. Int J Mol Sci 21
Remans T, Nacry P, Pervent M, Filleur S, Diatloff E, Mounier E, Tillard P, Forde BG, Gojon A (2006) The Arabidopsis NRT1.1 transporter participates in the signaling pathway triggering root colonization of nitrate-rich patches. Proc Natl Acad USA 103:19206–19211
Rizza A, Tang B, Stanley CE, Guido G, Owen MR, Band LR, Jones AM (2021) Differential biosynthesis and cellular permeability explain longitudinal gibberellin gradients in growing roots. Proc Natl Acad Sci USA 118
Roychoudhury A, Aftab T (2021) Phytohormones, plant growth regulators and signaling molecules: cross-talk and stress responses. Plant Cell Rep 40:1301–1303
Sannwald EH, Pyke DA, Caldwell MM (2011) Perception of neighbouring plants by rhizomes and roots: morphological manifestations of a clonal plant. Plant Cell Environ 75:2146–2157
Sarma RK, Saikia R (2014) Alleviation of drought stress in mung bean by strain Pseudomonas aeruginosa GGRJ21. Plant Soil 377:111–126
Schlesinger WH, Dietze MC, Jackson RB, Phillips RP, Rhoades CC, Rustad LE, Vose JM (2015) Forest biogeochemistry in response to drought. Global Change Biol 22:2318–2328
Schroeder JI, Kwak JM, Allen GJ (2001) Guard cell abscisic acid signaling and engineering drought hardiness in plants. Nature 410:327–330
Shi HL, Ma WJ, Song JY, Lu M, Rahman SU, Bui TTX, Vu DD, Zheng HF, Wang JH, Zhang Y (2017) Physiological and transcriptional responses of Catalpa bungei to drought stress under sufficient- and deficient-nitrogen conditions. Tree Physiol 37:1457–1468
Song JY, Wang Y, Pan YH, Pang JY, Zhang X, Fan JF, Zhang Y (2019) The influence of nitrogen availability on anatomical and physiological responses of Populus alba × P. glandulosa to drought stress. BMC Plant Biol 19:63
Song MQ, Fan XR, Chen JG, Qu HY, LuoL, Xu GH (2020) OsNAR2.1 Interaction with OsNIT1 and OsNIT2 functions in root-growth responses to nitrate and ammonium. Plant Physiol 183
Trevisan S, Trentin AR, Ghisi R, Masi A, Quaggiotti S (2018) Nitrate affects transcriptional regulation of upbeat1 and ros localisation in roots of zea mays L. Physiol Plantarum 166:794–811
Verslues PE, Zhu JK (2005) Before and beyond ABA: upstream sensing and internal signals that determine ABA accumulation and response under abiotic stress. Biochem Soc T 33:375–379
Wang JC, Qin XM, Xu SQ, Zhao MY, Shu PQ, Xu FJ, Ma JY, Sun Y, Dong H, Guo ZY, Long DP, Ma WJ, Lu YZ, Xie XM, Chen YL, Chu J, Wang JH, Zhang Y (2021) Nitrogen availability affects stem development and response to differential root-zone drought stress in Catalpa bungei. Environ Exp Bot 186
Wang Y, Liu F, Jensen CR (2012) Comparative effects of deficit irrigation and alternate partial root-zone irrigation on xylem pH, ABA and ionic concentrations in tomatoes. J Exp Bot 63:1907–1917
Xu N, Yu B, Chen RR, Li ST, Zhang GH, Huang JL (2020) OsNAR2.2 plays a vital role in the root growth and development by promoting nitrate uptake and signaling in rice. Plant Physiol Bioch 149:159–169
Yang WQ, Goulart BL, Demchak K, Li Y (2002) Interactive effects of mycorrhizal inoculation and organic soil amendments on nitrogen acquisition and growth of highbush blueberry. J Am Soc Hortic Sci 127:742–748
Yildirm K, Yağci A, Sucu S, Tunc S (2018) Responses of grapevine rootstocks to drought through altered root system architecture and root transcriptomic regulations. Plant Physiol Bioch 127:256–268
Zheng HF, Zhang X, Ma WJ, Song JY, Rahman SU, Wang JH, Yi Z (2017) Morphological and physiological responses to cyclic drought in two contrasting genotypes of Catalpa bungei. Environ Exp Bot 138:77–87
Zhao MR, Han YY, Feng YN, Li F, Wang W (2012) Expansins are involved in cell growth mediated by abscisic acid and indole-3-acetic acid under drought stress in wheat. Plant Cell Rep 31:671–685
Zou N, Huang L, Chen HJ, Huang XF, Song QN, Yang QP, Wang TC (2020) Nitrogen form plays an important role in the growth of moso bamboo (phyllostachys edulis) seedlings. PeerJ 8:e9938
Acknowledgements
This project was financially supported by The National Key Research and Development Program of China (Grant No. 2021YFD2200301), The Subject of Key R & D Plan of Shandong Province (2021LZGC023), the Key R&D Program of Shaanxi province (Grant No. 2020NY-005), and the Key R&D Program of Shaanxi province (Grant No. 2020NY- -120).
Author information
Authors and Affiliations
Contributions
TS, WM and YL cultivated plants. TS, RR, SZ, DH, MW, QH, WC, TX, LK and KL investigated and measured physiological parameters. TS, MX, WY and YF analyzed data. TS and QD prepared original draft. YC and JW reviewed and edited manuscript. YZ and JW supervised the experiment. YZ revised the manuscript. MX, LZ, WY and YF contributed to the revision of manuscript. WZ supervised the revision of the paper and revised the Discussion part.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of financial and non-financial interests, funding.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, T., Ren, R., Xing, M. et al. Morphological and physiological plasticity of Catalpa bungei roots under partial root-zone drought as affected by nitrogen forms. New Forests 55, 383–402 (2024). https://doi.org/10.1007/s11056-023-09983-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11056-023-09983-9