Abstract
Introduction
Immune checkpoint inhibitors (ICIs) can induce adverse neurological effects. Due to its rarity as an adverse effect, meningitis has been poorly described. Therefore, meningitis diagnosis and management can be challenging for specialists. Moreover, meningitis can be an obstacle to resuming immunotherapy. Given the lack of alternatives, the possibility of reintroducing immunotherapy should be discussed on an individual basis. Here, we present a comprehensive systematic review of meningitis related to ICIs.
Review
We performed a search for articles regarding immune-related meningitis published in PubMed up to November 2021 with the MeSH terms “meningitis” and “immune checkpoint” using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) method. We summarized the studies not only by category but also based on whether it was a primary article or case report to provide a systematic overview of the subject. We reviewed a total of 38 studies and herein report the clinical experiences, pharmacovigilance data and group knowledge from these studies.
Conclusion
This review summarizes the existing information on immune-related meningitis and the possibility of reintroducing immunotherapy after the development of central neurological side effects. To the best of our knowledge, there is little information in the literature to guide clinicians on decisions regarding whether immunotherapy should be continued after a neurological adverse event occurs, especially meningeal events. This review emphasizes the necessity of systematic examinations, steroid treatment (as a cornerstone of management) and the need for further exploratory studies to obtain a clearer understanding of how to better manage patients who experience these side effects. The findings summarized in this review can help provide guidance to practitioners who face this clinical situation.
Similar content being viewed by others
Introduction
Currently, immune checkpoint inhibitors (ICIs) have become the standard of care for numerous cancers. In 2011, ipilimumab was approved by the Food and Drug Administration (FDA) to treat metastatic melanoma (MM), with an improvement in progression-free survival (PFS) of 4 months [1]. In 2015, nivolumab, an inhibitor of programmed death ligand 1 (PDL1), improved the overall response of MM patients compared to dacarbazine [2]. In 2017, the combination of nivolumab and ipilimumab achieved a median overall survival (OS) of 60 months compared to the 36.9 months achieved with nivolumab alone for the treatment of MM [3]. Consequently, the nivolumab plus ipilimumab combination became the new standard of care for BRAF-negative MM.
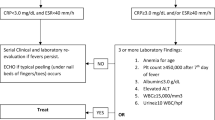
However, ICIs induce unique side effects. Ipilimumab alone and its combination with nivolumab are associated with the highest rates of immune-related adverse effects (irAEs) among other immunotherapies, as 53% of patients treated with such regimens had grade 3–4 irAEs [4]. IrAEs can involve the central nervous system (CNS) and are often severe despite their rarity. Due to the difficulty in diagnosing neurological irAEs, the reported incidence of 1–5% is probably an underestimate [5]. In particular, immune-induced aseptic meningitis is associated with high rates of mortality and/or morbidity [7]. Systematic explorations with at least CNS imaging, lumbar puncture, viral screening and viral serology analysis are recommended by the European Society for Medical Oncology (ESMO) [8]. If meningeal irAEs cause sufficient concern, management typically features high-dose steroid administration for at least 4 to 6 weeks with decreasing doses [8].
Whether ICIs should be resumed thereafter is still debated. After some irAEs develop, because of the lack of an efficient alternative option for metastatic disease treatment, resuming ICIs can be the best choice. The current review attempted to summarize reported knowledge about the management of immune-related meningitis and the reintroduction of ICIs.
Methodology
We searched for articles related to immune-related meningitis published on PubMed with the MesH terms “meningitis” and “immune checkpoint” up to November 19, 2021, using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) method (Fig. 1.). We summarized primary articles and case reports to give a systematic overview of the subject.
Results
In 11 articles, 40 cases of immune-related meningitis or meningoencephalitis (with at least signs of meningitis on lumbar puncture) were reported [10,11,12,13,14,15,16,17,18,19,20,21,22] (Tables 1 and 2). An overview of the results is presented in Fig. 2. In our systematic review, 18 articles were reviews of neuronal irAEs. Their main points are summarized in the following sections in parallel with a description of the case series.
Summary of the 41 cases reported in this review. * = percentages of the symptoms reported in the 41 cases; patients could have more than one symptom; ∫ = percentages from the case reports including MRI results (n = 39); † = percentages from the case reports on treatment initiation (n = 41); ∆ = percentages from the case reports about the patient course after diagnosis of irAEs (n = 41). Abbreviations: CR complete response, PD progressive disease, PR partial response, pt patients, ICI immune checkpoint inhibitor, irAEs immune-related adverse events, IV intravenous, MRI magnetic resonance imaging. The figure was generated with illustrations from smart.servier.com
Population characteristics
Data from 40 patients, including 22 men and 16 women with a median age of 56 years, were collected [range 19–82 years]. Overall, 21 patients (52.5%), 10 patients (25.0%), six patients (15.0%), two patients and one patient presented with melanoma, lung cancer, renal cell carcinoma, Hodgkin lymphoma, and colorectal cancer with microsatellite instability, respectively. Four patients (10.0%) had brain metastasis, and surgery was performed on one patient, but no other data on local treatment were reported for the other patients.
Ipilimumab and nivolumab were the most frequently prescribed ICIs. The combination of both was used in 16 patients (40.0%), ipilimumab alone was used in seven patients (17.5%), and nivolumab alone was used in five patients (12.5%). Pembrolizumab was used in six patients (15.0%), atezolizumab was used in five patients, and spartalizumab was used in one patient.
Clinical outcomes
The most common symptoms were headache, fever, cognitive disturbance and gait instability. The symptoms began after a median of 2 cycles [range 1–14 cycles]. The clinical status of patients deteriorated quickly, occurring within a few days after the beginning of symptoms. All patients except three presented with cerebrospinal fluid (CSF) lymphocytosis. One patient refused lumbar puncture, and one did not have detectable cells in the CSF [11], and their last exam showed only a protein content over 6 g/L [14]. Data on the white blood cell count was available for 17 patients, with a median value of 25 cells/mm3 (0–320 cells/mm3). Proteinorachy was described for 16 patients, with a median value of 0.87 g/L (0.3–3.85 g/L). Cerebral imaging was performed by magnetic resonance imaging (MRI) for 38 patients, with diffuse leptomeningeal enhancement observed in 16 (42.1%). One patient had cerebral edema, which is a sign of encephalitis [19]. No specific signs were reported for 21 patients (55%). Some cases reported specific contrast enhancement of the basal ganglia, pituitary gland, corpus callosum or frontal lobe.
Treatment and evaluation
Proper tapering of high-dose corticosteroids is the cornerstone of treatment [70]. Unfortunately, 20% of patients did not improve with corticosteroids alone, and the addition of an immunosuppressive agent was required [71, 72]. Due to the potential residual benefit of ICIs, multidisciplinary discussions and decisions, particularly about the management of severe cases, are important, especially when the patient is in intensive care [73].
In our case series, the main treatment component was steroids. 30 patients received intravenous (IV) steroids (75.0%), and five received oral steroids (12.5%). The initial dose varied between 1 g/day and 1 mg/kg/day for 3 to 5 days, followed by a dose reduction over a median of 6 weeks after improvement. Katakura et al. reported a patient treated with 30 mg of oral steroids but did not specify the time to complete recovery.
In six patients (15.0%), symptoms did not improve after steroid treatment. IV immunoglobulins were administered to five patients, and infliximab was introduced to two patients. Garcia et al. reported a patient who improved after IV steroid administration but quickly relapsed at the end of the steroid decrease. Consequently, a combination of steroids and immunoglobulins was tried, but the outcome was unsatisfactory. The addition of infliximab finally induced a near complete recovery [23]. Thouvenin et al. reported the case of a 63-year-old man treated with nivolumab for renal cell carcinoma who developed immune-related meningoencephalitis with uncontrolled choreatic movements. Despite steroid and infliximab treatments, the patient deteriorated and died [19].
After the initiation of the treatment, improvement usually occurred in a few days. However, Bompaire et al. reported a case of severe meningoneuritis that required IV steroids and immunoglobulin, which induced symptom improvement within only 1 month. The patient remained in complete remission after 24 months [24]. Sequelae-free complete recovery was observed in 35 patients (87.5%). Only three patients (7.5%) did not achieve complete symptom improvement. All of these patients had clinical signs more related to encephalitis (ataxia and diplopia) [25, 26] or polyradiculoneuropathy [12] than to meningitis. Kopecky et al. and Minami et al. reported two cases of death due to meningitis (4.9%). Both patients died quickly, 1 week after the beginning of deterioration, despite the start of high-dose steroids and/or infliximab [17, 27].
In five cases, the authors did not administer treatment because of low-grade meningitis. Spontaneous improvement was noted at a median time of 10 days (7–65 days) [11, 19].
Follow-up and therapy reintroduction
After recovery, ICI reintroduction was proposed in 14 patients (35.0%). In four patients, the same ICI was prescribed. New irAEs were reported in three patients after reintroduction, all of whom had received the same ICI. One patient developed interstitial lung disease and meningitis relapse, and the other two developed adrenal insufficiency [11, 21, 28]. Takamasu et al. reported that a patient with stage IV renal cell carcinoma achieved a complete response owing to the combination of ipilimumab 1 mg/kg and nivolumab 3 mg/kg, despite irAE reoccurrence [28]. Six of the seven cases reported by Cuzzubbo et al. did not experience irAE reoccurrence, even after ICI continuation, with two of the six cases receiving dual ICI treatment with 1 mg/kg ipilimumab. The patient treated with spartalizumab was diagnosed with interstitial lung disease shortly after reintroduction of the same ICI [11]. Fellner et al. also reported successful outcomes after the reintroduction of ICIs, but only with nivolumab, as irAEs developed with the combination of ipilimumab and nivolumab [81].
Five patients who received therapy reintroduction (35.7%) demonstrated progressive disease, and three of these patients succumbed to disease-related death. Five patients (35.7%) had a complete or partial response, and one other had a dissociated response. No stable disease was reported in the therapy reintroduction population.
At the last follow-up after irAEs were reported, among the patients with reported data, the overall response rate was 51.9%. Five patients achieved a complete response (18.5%), and nine patients achieved a partial response (33.3%). Eight patients experienced disease progression (29.6%), and five patients had stable disease (18.5%). The disease control rate was 70.4%, which is comparable to the rates reported in phase 3 studies of immunotherapy [3, 29].
Pharmacovigilance studies
Three articles analyzed pharmacovigilance data using disproportionality analysis, and the results revealed an association between ICI use and neurotoxicity [7, 30, 31]. Johnson et al. reported 18,518,994 neurological AEs, among which 48,653 were related to ICIs. The researchers concluded that the patients receiving ICIs had a higher incidence of myasthenia gravis (ROR = 16.5), encephalitis (ROR = 10.4), peripheral neuropathy and meningitis compared to those receiving other systemic treatments (ROR = 3.1). Meningitis (0.15% of patients in their cohort) was preferentially associated with the use of anti-CTLA-4 agents [7].
Sato et al. reported data from the Japanese Adverse Drug Event Report database. From a total of 7604 cases of irAEs, they identified 583 (7.67%) neurological AEs related to ICIs. The authors compared the incidences of AEs between nivolumab and other ICI subtypes. They concluded that the use of ipilimumab was associated with a higher incidence of meningitis. The time to the development of meningitis was shorter than the time to the development of other neurological irAEs [31]. In another study of 50,406 irAEs by Mikami et al., they used the FDA reporting system and identified 3619 neurological irAEs (7.2%). This number is similar to that reported by Sato et al., but Mikami et al. showed a higher incidence of neurological complications with the use of ICIs than non-ICI drugs. ICI combinations were associated with a higher incidence of neurological complications, mainly hypophysitis and hypopituitarism. The authors do not report any other risk factors associated with this higher incidence. Dual ICI therapy, older age, melanoma and non-small-cell lung cancer (NSCLC) seemed to be associated with a higher risk of fatal neurological irAEs, including meningeal irAEs [30].
ICI efficacy in brain and leptomeningeal metastasis
Of the studies retrieved by our literature search, five articles focused on the efficacy of ICIs in patients with central nervous system metastasis. Kuske et al. reviewed different treatments for melanoma brain metastasis and reported on phase 2 studies that evaluated ICIs in brain metastasis, which showed an intracranial response of approximately 42 to 55%. No difference in safety data was reported, except for slightly more headaches of any grade with dual ICI treatment [32].
Nguyen et al. focused on leptomeningeal metastasis and reported on the findings of different ongoing studies evaluating ICIs in this context. The researchers provided an interim analysis of the Brastianos et al. study, with 44% of patients alive at 3 months after pembrolizumab treatment for solid tumor leptomeningeal metastasis [33, 34]. The use of ICIs in this setting was also the topic of a review by Kondoh et al. [35].
For NSCLC, Gio et al. reported the efficacy of nivolumab in treating leptomeningeal metastasis and did not report any neurological irAEs [36]. Hendricks et al. reported an analysis of 19 patients with leptomeningeal metastases from NSCLC treated with ICIs. No safety data were reported, but the median overall survival was 3.7 months [37]. Nakashima et al. also reported the case of a 66-year-old woman with meningeal carcinomatosis from NSCLC treated with ICIs in combination with whole brain radiation. She achieved more than 23 months of survival without disease progression. This case introduced the idea of including radiotherapy in the treatment regimen. A higher irAE incidence with radiotherapy has not been reported [38,39,40,41,42].
These articles underline the importance of ICIs for the treatment of metastatic CNS tumors and confirm that there is no obvious increase in the incidence of irAEs after such treatment.
Discussion
Clinical signs and diagnosis
Neurological irAEs can present as various symptoms [43, 44]. In particular, CNS symptoms are easily underestimated because they manifest at a lower intensity than related symptoms. Usually, neurological irAEs are described in three categories: encephalitis, aseptic meningitis and multiple sclerosis. Nonspecific isolated symptoms, such as headaches, are the most frequently reported symptoms (55%) and are usually low intensity [45].
Other than isolated symptoms, encephalitis and encephalopathy are the most frequently reported irAEs. Regardless, they occur in less than 1% of patients treated with ICIs [6]. Medical practitioners must be aware of these complications, especially due to the broad range of symptoms that can occur. Indeed, unexplained paucisymptomatic headache or focal weakness can be manifestations of grade 1 CNS irAEs [10]. Larkin et al. reported 6 cases of encephalitis, and most patients presented with mental disturbance, seizure and fatigue. Five of the six patients required prolonged hospitalization, and one of them died from complications [10]. Encephalitis leads to increased major morbidity and mortality, especially in cases of limbic encephalitis and cerebral inflammation, even with the administration high-dose steroids [46, 47]. Some pharmacovigilance databases have revealed a fatality rate of 19% when the brainstem is involved [48, 49]. The distinction between neurological irAEs and CNS infection can be challenging, particularly due to the lack of specific positive criteria and the presentation of flu-like symptoms in some cases of irAEs [50]. Infection can also probably induce neurological irAEs, as reported in some cases after herpes simplex infection or Epstein–Barr infection [49, 51]. Ultimately, the diagnosis should be based on a systematic approach with MRI, lumbar puncture, electroencephalography (EEG) if clinically indicated, and screening for typical autoimmune antibodies and/or infectious causes is necessary (Herpesviridae, enterovirus, varicella, and/or bacterial culture) [53, 54]. Nonspecific inflammatory signs can be revealed on MRI and can be consistent with the presence of lymphocytic or neutrophilic pleocytosis, leading to the overlapping diagnosis of immune-induced meningoencephalitis. Of note, all of these tests can also yield normal results; ultimately, patient history and symptom resolution with corticosteroid therapy are factors indicative of a diagnosis of immune-related encephalitis [8].
The second most common CNS irAE described in the series was aseptic meningitis, which was more common with ICI combinations, especially combinations with ipilimumab. Immune-related aseptic meningitis occurred earlier than other neurological irAEs, with a median duration of two cycles and a delay of 9 days from the last injection of ICI to the manifestation of clinical signs [7, 45, 55, 56]. Immune-related aseptic meningitis occurs in less than 1% of cases and represents 6 to 15% of all neurological irAEs [5, 45, 57]. The clinical presentation varies from headache with photophobia to complete cranial hypertension with seizure. This variability in symptoms can make it difficult to distinguish aseptic meningitis from encephalitis. MRI results are often normal or reveal leptomeningeal inflammation. Lumbar puncture usually shows lymphocytosis with elevated protein, which is defined according to ESMO as a white blood cell count between 5 and 500/µL [7]. The CSF is sterile and negative for cytopathology. There are several overlapping diagnostic algorithms used to facilitate the differential diagnosis of immune-related meningitis [8, 58,59,60]. When testing for encephalitis, lumbar puncture and MRI with infectious disease screening (in particular, PCR for herpes simplex virus but also typical bacterial screening) are essential [61]. When peripheral symptoms are associated with central clinical signs, screening for thyroid dysfunction and/or vitamin B12/B9 deficiency is recommended [59].
Prevention of irAEs and survival outcomes
Because ICIs are almost universally accepted, the prevention of side effects is key to improving the benefit-risk ratio [65, 66]. The incidence of irAEs depends on the ICI, and different strategies have been explored to limit irAEs [67]. The Checkmate 511 study evaluated two combinations of nivolumab and ipilimumab, comparing treatment with nivolumab 1 mg/kg and ipilimumab 3 mg/kg and treatment with nivolumab 3 mg/kg and ipilimumab 1 mg/kg [68]. After 3 years, the number of grade 3–5 irAEs was significantly lower in the second group (48.3% versus 33.9%), without any difference in OS or PFS [68]. Only the irAEs that occurred in at least 10% of their population were actually reported, so specific data on meningitis are not available.
The prognostic value of irAEs has also been evaluated. Patients who developed side effects seemed to have better survival outcomes than those without any adverse effects [69]. Indini et al. showed improvements in both PFS and OS among patients with MM [9]. Shah et al. analyzed survival data from a cohort of patients who were readministered ICIs after irAEs occurred, and they reported the worst OS and PFS outcomes for patients with a shorter time to the development of initial or post-reintroduction irAEs. On the other hand, patients had a lower risk of disease progression if they completed more than 10 weeks of treatment after the resumption of ICIs.
Reintroduction of ICIs
The reintroduction of ICIs after the resolution of irAEs is still controversial. The National Comprehensive Cancer Network (NCCN), ESMO and the American Society of Clinical Oncology (ASCO) propose reintroducing ICIs only in cases of grade 1 or 2 irAEs [8, 70, 72]. Indeed, some reports have shown that half of the patients with severe irAEs will develop the same or distinct irAEs after the reintroduction of ICIs [74]. However, patients experiencing irAEs could have better OS and PFS outcomes after reintroduction than those who change treatment regimens [75]. A better understanding of the mechanisms of each irAE is clearly required [76,77,78].
The management and follow-up of patients with irAEs should be specific to the system affected. Indeed, patients with immune-related hepatitis as an irAE seem to be amenable to the reintroduction of ICIs, with more than 60% of patients avoiding recurrence of grade 2 or greater hepatitis in the study of Allouchery et al. [79]. In contrast, Simonaggio et al. reported that 55% of their patients experienced irAEs after reintroduction. In these patients, colic, pulmonary, joint and hematological toxicities were most likely to occur [74]. Dolladille et al. also explored the characteristics of irAEs after the reintroduction of ICIs, and the results showed that colitis and pneumonitis had higher recurrence rates than rarer irAEs, such as endocrine irAEs [80]. Although there are more than 400 reported irAEs, the rarity of CNS events complicates their analysis. The severity of irAEs, systems affected by irAEs, alternative therapeutic strategies and patient preference must be considered before the resumption of ICIs.
Regarding immune-related meningitis, case reports tend to show that reintroduction of ICIs is possible and can achieve good outcomes. Different strategies can be used, particularly for dual therapy. The reintroduction of ipilimumab has remained controversial because anti-CTLA4 agents are associated with a higher rate of meningitis and irAEs [7, 67]. Albandar et al. also studied survival outcomes after the reintroduction of ICIs, and they reported a median OS of 38.6 months among patients in whom treatment was reinitiated after interruption versus 24.9 months among patients in whom treatment was discontinued. However, this difference was not significantly different [82]. Only a few studies exploring the possibility of ICI reintroduction have been reported, so further studies are needed to help better understand and manage these meningeal irAEs.
Conclusion
With the emergence of ICIs, AEs have become a new challenge for specialists. In this review, we attempted to describe the variety of clinical signs and consequences of neurological irAEs. Due to their rarity, particularly meningitis, the guidelines recommend systematic biological and clinical examinations to avoid misdiagnosis. Steroids remain the principal treatment for neurological irAEs and successfully resolve the majority of cases. However, whether ICIs should be reintroduced remains to be determined. The answer seems to depend on the system involved, kinetics of improvement and clinical severity, but good outcomes have been achieved after reintroduction in some patients with immune-related meningitis. The collection of additional data in the near future will help to personalize the management strategy and follow-up schedule for patients with such irAEs. In conclusion, our review provides a comprehensive summary of the real-world knowledge on immune-related aseptic meningitis, which we hope will provide guidance for physicians who manage these patients.
Decalartions
Conflict of interest
None declared.
Data availability
All data analyzed during this study are included in this published article and its supplementary information files.
Abbreviations
- ASCO:
-
American Society of Clinical Oncology
- CNS:
-
Central nervous system
- CSF:
-
Cerebrospinal fluid
- CTLA-4:
-
Cytotoxic T-lymphocyte-associated protein 4
- EEG:
-
Electroencephalography
- ESMO:
-
European Society of Medical Oncology
- FDA:
-
Food and Drug Association
- ICI:
-
Immune checkpoint inhibitor
- IrAEs:
-
Immune-related adverse events
- MM:
-
Metastatic melanoma
- MRI:
-
Magnetic resonance imaging
- NCCN:
-
National Comprehensive Cancer Network
- NMDA:
-
N-methyl-D-aspartate
- NSCLC:
-
Non-small-cell lung cancer
- OS:
-
Overall survival
- PD(L)1:
-
Programmed death (ligand) 1
- PET-CT:
-
Positron emission tomography-computed tomography
- PFS:
-
Progression-free survival
References:
Alexander W (2016) The checkpoint immunotherapy revolution. P T mars 41(3):185–191
Robert C, Long GV, Brady B, Dutriaux C, Maio M, Mortier L et al (2015) nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med 372(4):320–30
Larkin J, Chiarion-Sileni V, Gonzalez R, Grob J-J, Rutkowski P, Lao CD et al (2019) Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 381(16):1535–46
Xu C, Chen Y-P, Du X-J, Liu J-Q, Huang C-L, Chen L et al (2018) Comparative safety of immune checkpoint inhibitors in cancer: systematic review and network meta-analysis. BMJ. 363:k4226
Duong SL, Barbiero FJ, Nowak RJ, Baehring JM (2021) Neurotoxicities associated with immune checkpoint inhibitor therapy. J Neurooncol avr 152(2):265–277
Galmiche S, Lheure C, Kramkimel N, Franck N, Boitier F, Dupin N et al (2019) Encephalitis induced by immune checkpoint inhibitors in metastatic melanoma: a monocentric retrospective study. J Eur Acad Dermatol Venereol 33(12):e440–e443
Johnson DB, Manouchehri A, Haugh AM, Quach HT, Balko JM, Lebrun-Vignes B et al (2019) Neurologic toxicity associated with immune checkpoint inhibitors: a pharmacovigilance study. J Immunother Cancer 7(1):134
Haanen JBAG, Carbonnel F, Robert C, Kerr KM, Peters S, Larkin J et al (2017) Management of toxicities from immunotherapy: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 28:iv119-42
Indini A, Di Guardo L, Cimminiello C, Prisciandaro M, Randon G, De Braud F et al (2019) Immune-related adverse events correlate with improved survival in patients undergoing anti-PD1 immunotherapy for metastatic melanoma. J Cancer Res Clin Oncol févr 145(2):511–521
Larkin J, Chmielowski B, Lao CD, Hodi FS, Sharfman W, Weber J et al (2017) Neurologic serious adverse events associated with nivolumab plus ipilimumab or nivolumab alone in advanced melanoma, including a case series of encephalitis. Oncologist juin 22(6):709–718
Cuzzubbo S, Tetu P, Guegan S, Ursu R, Belin C, Sirven Villaros L et al (2020) Reintroduction of immune-checkpoint inhibitors after immune-related meningitis: a case series of melanoma patients. J Immunother Cancer 8(2):e001034
Ohno N, Sugimoto T, Giga M, Naito H, Kono T, Nomura E (2021) A case of meningoencephalitis and polyradiculoneuropathy induced by combination therapy with ipilimumab and nivolumab. Rinsho Shinkeigaku. 61(10):658–662
Bello-Chavolla OY, Cortes-Arroyo JE, Vargas-Vazquez A, Quiroz-Compean F, Leal-Gutierrez G, Barragan-Dessavre M et al (2018) Meningeal syndrome in a patient treated with a combination of immune checkpoint inhibitors for a metastatic melanoma. Rev Neurol 67(7):279–80
Laserna A, Tummala S, Patel N, El Hamouda DEM, Gutiérrez C (2018) Atezolizumab-related encephalitis in the intensive care unit: Case report and review of the literature. SAGE Open Med Case Rep. https://doi.org/10.1177/2050313X18792422
Yonenobu Y, Ishijima M, Toyooka K, Fujimura H (2019) A case of meningoencephalitis associated with pembrolizumab treated for squamous cell lung cancer. Rinsho Shinkeigaku 59(2):105–8
Shields LBE, Alsorogi MS, Mar N, Rezazadeh Kalebasty A (2021) Immune-related meningoencephalitis following Nivolumab in metastatic renal cell carcinoma. Case Rep Oncol 14(2):1051–8
Minami S, Okada H, Ihara S, Tsuji H, Yamadera M, Yasuoka H (2021) Pembrolizumab-induced meningoencephalitis: a brain autopsy case. J Med Cases sept 12(9):359–365
Ogawa K, Kaneda H, Kawamoto T, Tani Y, Izumi M, Matsumoto Y et al (2020) Early-onset meningitis associated with atezolizumab treatment for non-small cell lung cancer: case report and literature review. Invest New Drugs déc 38(6):1901–1905
Thouvenin L, Olivier T, Banna G, Addeo A, Friedlaender A (2021) Immune checkpoint inhibitor-induced aseptic meningitis and encephalitis: a case-series and narrative review. Ther Adv Drug Saf 12:20420986211004744
Lima G, Kahn A, Sama S, Savage J (2019) Aseptic meningitis as an immune-related adverse event after pembrolizumab. Case Rep Oncol Med 2019:7183747
Katakura Y, Kimura T, Kusano T, Tatsumi F, Iwamoto Y, Sanada J et al (2021) Case report: a variety of immune-related adverse events triggered by immune checkpoint inhibitors in a subject with malignant melanoma: destructive thyroiditis, aseptic meningitis and isolated ACTH deficiency. Front Endocrinol (Lausanne) 12:722586
Toyozawa R, Haratake N, Toyokawa G, Matsubara T, Takamori S, Miura N, Yamaguchi M, Takenoyama M, Seto T (2020) Atezolizumab-induced aseptic meningitis in patients with NSCLC. JTO Clin Res Reports 1(1):100012. https://doi.org/10.1016/j.jtocrr.2020.100012
Garcia CA, El-Ali A, Rath TJ, Contis LC, Gorantla V, Drappatz J et al (2018) Neurologic immune-related adverse events associated with adjuvant ipilimumab: report of two cases. J Immunother Cancer 6:83
Bompaire F, Mateus C, Taillia H, De Greslan T, Lahutte M, Sallansonnet-Froment M et al (2012) Severe meningo-radiculo-neuritis associated with ipilimumab. Invest New Drugs déc 30(6):2407–2410
Quach HT, Robbins CJ, Balko JM, Chiu CY, Miller S, Wilson MR et al (2019) Severe epididymo-orchitis and encephalitis complicating anti-PD-1 therapy. Oncologist juill 24(7):872–876
Zurko J, Mehta A (2018) Association of immune-mediated cerebellitis with immune checkpoint inhibitor therapy. Mayo Clin Proc Innov Qual Outcomes 2(1):74–7
Kopecký J, Kubeček O, Geryk T, Slováčková B, Hoffmann P, Žiaran M et al (2018) Nivolumab induced encephalopathy in a man with metastatic renal cell cancer: a case report. J Med Case Rep 12:262
Takamatsu D, Furubayashi N, Negishi T, Ieiri K, Inoue T, Tsukino K et al (2019) Relapse of aseptic meningitis induced by ipilimumab and nivolumab therapy for metastatic renal cell carcinoma: a case report. Mol Clin Oncol déc 11(6):590–594
Garon EB, Hellmann MD, Rizvi NA, Carcereny E, Leighl NB, Ahn M-J et al (2019) Five-year overall survival for patients with advanced non-small-cell lung cancer treated with pembrolizumab: results from the phase I KEYNOTE-001 study. J Clin Oncol 37(28):2518–27
Mikami T, Liaw B, Asada M, Niimura T, Zamami Y, Green-LaRoche D et al (2021) Neuroimmunological adverse events associated with immune checkpoint inhibitor: a retrospective, pharmacovigilance study using FAERS database. J Neurooncol mars 152(1):135–144
Sato K, Mano T, Iwata A, Toda T (2019) Neurological and related adverse events in immune checkpoint inhibitors: a pharmacovigilance study from the Japanese adverse drug event report database. J Neurooncol 145(1):1–9
Kuske M, Rauschenberg R, Garzarolli M, Meredyth-Stewart M, Beissert S, Troost EGC et al (2018) Melanoma brain metastases: local therapies, targeted therapies, immune checkpoint inhibitors and their combinations—chances and challenges. Am J Clin Dermatol 19(4):529–541
An overview of leptomeningeal disease - Nguyen - Annals of Palliative Medicine [Internet]. [cité 10 oct 2021]. Disponible sur: https://apm.amegroups.com/article/view/51419/html
Pembrolizumab In Central Nervous System Metastases - Full Text View - ClinicalTrials.gov [Internet]. [cité 10 oct 2021]. Disponible sur: https://clinicaltrials.gov/ct2/show/NCT02886585
Kondoh T, Sonoda T (2021) Treatment options for leptomeningeal metastases of solid cancers: literature review and personal experience. Acta Neurochir Suppl 128:71–84
Gion M, Remon J, Caramella C, Soria J-C, Besse B (2017) Symptomatic leptomeningeal metastasis improvement with nivolumab in advanced non-small cell lung cancer patient. Lung Cancer juin 108:72–74
Survival of patients with non-small cell lung cancer having leptomeningeal metastases treated with immune checkpoint inhibitors - European Journal of Cancer [Internet]. [cité 10 oct 2021]. Disponible sur: https://www.ejcancer.com/article/S0959-8049(19)30322-3/fulltext
Goldberg SB, Schalper KA, Gettinger SN, Mahajan A, Herbst RS, Chiang AC et al (2020) Pembrolizumab for management of patients with NSCLC and brain metastases: long-term results and biomarker analysis from a non-randomised, open-label, phase 2 trial. Lancet Oncol mai 21(5):655–663
Shaverdian N, Lisberg AE, Bornazyan K, Veruttipong D, Goldman JW, Formenti SC et al (2017) Previous radiotherapy and the clinical activity and toxicity of pembrolizumab in the treatment of non-small-cell lung cancer: a secondary analysis of the KEYNOTE-001 phase 1 trial. Lancet Oncol juill 18(7):895–903
Alomari AK, Cohen J, Vortmeyer AO, Chiang A, Gettinger S, Goldberg S et al (2016) Possible interaction of Anti-PD-1 therapy with the effects of radiosurgery on brain metastases. Cancer Immunol Res juin 4(6):481–487
Pike LRG, Bang A, Ott P, Balboni T, Taylor A, Catalano P et al (2017) Radiation and PD-1 inhibition: favorable outcomes after brain-directed radiation. Radiother Oncol juill 124(1):98–103
Hubbeling HG, Schapira EF, Horick NK, Goodwin KEH, Lin JJ, Oh KS et al (2018) Safety of combined PD-1 pathway inhibition and intracranial radiation therapy in non-small cell lung cancer. J Thorac Oncol avr 13(4):550–558
Hottinger AF (2016) Neurologic complications of immune checkpoint inhibitors. Curr Opin Neurol déc 29(6):806–812
Seki M, Suzuki S (2021) Distinctive disease entity of neurological adverse events associated with immune checkpoint inhibitors. Brain Nerve janv 73(1):35–46
Cuzzubbo S, Javeri F, Tissier M, Roumi A, Barlog C, Doridam J et al (2017) Neurological adverse events associated with immune checkpoint inhibitors: review of the literature. European Journal of Cancer 73:1–8
Salam S, Lavin T, Turan A (2016) Limbic encephalitis following immunotherapy against metastatic malignant melanoma. BMJ Case Rep. https://doi.org/10.1136/bcr-2016-215012
Vitt JR, Kreple C, Mahmood N, Dickerson E, Lopez GY, Richie MB (2018) Autoimmune pancerebellitis associated with pembrolizumab therapy. Neurology 91(2):91
Bossart S, Thurneysen S, Rushing E, Frontzek K, Leske H, Mihic-Probst D et al (2017) Case report: encephalitis, with brainstem involvement, following checkpoint inhibitor therapy in metastatic melanoma. Oncologist juin 22(6):749–753
Johnson DB, McDonnell WJ, Gonzalez-Ericsson PI, Al-Rohil RN, Mobley BC, Salem J-E et al (2019) A case report of clonal EBV-like memory CD4+ T cell activation in fatal checkpoint inhibitor-induced encephalitis. Nat Med août 25(8):1243–1250
Burke M, Hardesty M, Downs W (2018) A case of severe encephalitis while on PD-1 immunotherapy for recurrent clear cell ovarian cancer. Gynecol Oncol Rep mai 24:51–53
Armangue T, Spatola M, Vlagea A, Mattozzi S, Cárceles-Cordon M, Martinez-Heras E et al (2018) Frequency, symptoms, risk factors, and outcomes of autoimmune encephalitis after herpes simplex encephalitis: a prospective observational study and retrospective analysis. Lancet Neurol sept 17(9):760–772
Williams TJ, Benavides DR, Patrice K-A, Dalmau JO, de Ávila ALR, Le DT et al (2016) Association of autoimmune encephalitis with combined immune checkpoint inhibitor treatment for metastatic cancer. JAMA Neurol. 73(8):928–33
Brown MP, Hissaria P, Hsieh AH, Kneebone C, Vallat W (2017) Autoimmune limbic encephalitis with anti-contactin-associated protein-like 2 antibody secondary to pembrolizumab therapy. J Neuroimmunol 305:16–8
Dutra LA, Abrantes F, Toso FF, Pedroso JL, Barsottini OGP, Hoftberger R (2018) Autoimmune encephalitis: a review of diagnosis and treatment. Arq Neuro-Psiquiatr janv 76:41–49
Dubey D, David WS, Reynolds KL, Chute DF, Clement NF, Cohen JV et al (2020) Severe neurological toxicity of immune checkpoint inhibitors: growing spectrum. Ann Neurol 87(5):659–669
Bruna J, Argyriou AA, Anastopoulou GG, Alemany M, Nadal E, Kalofonou F et al (2020) Incidence and characteristics of neurotoxicity in immune checkpoint inhibitors with focus on neuromuscular events: experience beyond the clinical trials. J Peripher Nerv Syst juin 25(2):171–177
Dalakas MC (2018) Neurological complications of immune checkpoint inhibitors: what happens when you ‘take the brakes off’ the immune system. Ther Adv Neurol Disord 11:1756286418799864
Haugh AM, Probasco JC, Johnson DB (2020) Neurologic complications of immune checkpoint inhibitors. Expert Opin Drug Saf avr 19(4):479–488
Touat M, Talmasov D, Ricard D, Psimaras D (2017) Neurological toxicities associated with immune-checkpoint inhibitors. Curr Opin Neurol déc 30(6):659–668
Shi J, Niu J, Shen D, Liu M, Tan Y, Li Y et al (2020) Clinical diagnosis and treatment recommendations for immune checkpoint inhibitor-related adverse reactions in the nervous system. Thorac Cancer févr 11(2):481–487
Astaras C, de Micheli R, Moura B, Hundsberger T, Hottinger AF (2018) Neurological adverse events associated with immune checkpoint inhibitors: diagnosis and management. Curr Neurol Neurosci Rep 18(1):3
Garcia CR, Jayswal R, Adams V, Anthony LB, Villano JL (2019) Multiple sclerosis outcomes after cancer immunotherapy. Clin Transl Oncol 21(10):1336–1342
Yshii LM, Hohlfeld R, Liblau RS (2017) Inflammatory CNS disease caused by immune checkpoint inhibitors: status and perspectives. Nat Rev Neurol déc 13(12):755–763
Cao Y, Nylander A, Ramanan S, Goods BA, Ponath G, Zabad R et al (2016) CNS demyelination and enhanced myelin-reactive responses after ipilimumab treatment. Neurology 86(16):1553–6
Thompson JA (2018) New NCCN Guidelines: Recognition and Management of Immunotherapy-Related Toxicity. J Natl Compr Canc Netw mai 16(5S):594–596
Pan PC-W, Haggiagi A (2019) Neurologic immune-related adverse events associated with immune checkpoint inhibition. Curr Oncol Rep 21(12):108
Marini A, Bernardini A, Gigli GL, Valente M, Muñiz-Castrillo S, Honnorat J et al (2021) Neurologic adverse events of immune checkpoint inhibitors: a systematic review. Neurology 96(16):754–66
Lebbé C, Meyer N, Mortier L, Marquez-Rodas I, Robert C, Rutkowski P et al (2019) Evaluation of two dosing regimens for nivolumab in combination with ipilimumab in patients with advanced melanoma: results from the phase IIIb/IV Checkmate 511 trial. J Clin Oncol 37(11):867–75
Das S, Johnson DB (2019) Immune-related adverse events and anti-tumor efficacy of immune checkpoint inhibitors. J Immunother Cancer 7:306
Brahmer JR, Lacchetti C, Thompson JA (2018) Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: american society of clinical oncology clinical practice guideline summary. J Oncol Pract avr 14(4):247–249
Pollack MH, Betof A, Dearden H, Rapazzo K, Valentine I, Brohl AS et al (2018) Safety of resuming anti-PD-1 in patients with immune-related adverse events (irAEs) during combined anti-CTLA-4 and anti-PD1 in metastatic melanoma. Ann Oncol 29(1):250–5
Thompson JA, Schneider BJ, Brahmer J, Andrews S, Armand P, Bhatia S et al (2019) Management of immunotherapy-related toxicities version 1.2019. J Natl Compr Canc Netw 17(3):255–89
Joseph A, Simonaggio A, Stoclin A, Vieillard-Baron A, Geri G, Oudard S et al (2020) Immune-related adverse events: a retrospective look into the future of oncology in the intensive care unit. Ann Intensive Care 10:143
Simonaggio A, Michot JM, Voisin AL, Le Pavec J, Collins M, Lallart A et al (2019) Evaluation of readministration of immune checkpoint inhibitors after immune-related adverse events in patients with cancer. JAMA Oncol 5(9):1310–7
Santini FC, Rizvi H, Plodkowski AJ, Ni A, Lacouture ME, Gambarin-Gelwan M et al (2018) Safety and efficacy of re-treating with Immunotherapy after immune-related adverse events in patients with NSCLC. Cancer Immunol Res 6(9):1093–9
Zhao Z, Zhang C, Zhou L, Dong P, Shi L (2021) Immune checkpoint inhibitors and neurotoxicity. Curr Neuropharmacol 19(8):1246–1263
Seki M, Kitano S, Suzuki S (2021) Neurological disorders associated with immune checkpoint inhibitors: an association with autoantibodies. Cancer Immunol Immunother 71(4):769–775
Wesley SF, Haggiagi A, Thakur KT, De Jager PL (2021) Neurological immunotoxicity from cancer treatment. Int J Mol Sci 22(13):6716
Correction: Safety of immune checkpoint inhibitor rechallenge after discontinuation for grade ≥2 immune-related adverse events in patients with cancer (2021). J Immunother Cancer. 9(2):1. https://doi.org/10.1136/jitc-2020-001622
Dolladille C, Ederhy S, Sassier M, Cautela J, Thuny F, Cohen AA et al (2020) Immune checkpoint inhibitor rechallenge after immune-related adverse events in patients with cancer. JAMA Oncol juin 6(6):1–7
Fellner A, Makranz C, Lotem M, Bokstein F, Taliansky A, Rosenberg S et al (2018) Neurologic complications of immune checkpoint inhibitors. J Neurooncol 137(3):601–9
Albandar HJ, Fuqua J, Albandar JM, Safi S, Merrill SA, Ma PC (2021) Immune-Related Adverse Events (irAE) in Cancer Immune Checkpoint Inhibitors (ICI) and survival outcomes correlation: to rechallenge or not? Cancers (Basel) 13(5):989
Funding
we don't have any funding to declare.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by SN and RS. The first draft of the manuscript was written by SN, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nannini, S., Koshenkova, L., Baloglu, S. et al. Immune-related aseptic meningitis and strategies to manage immune checkpoint inhibitor therapy: a systematic review. J Neurooncol 157, 533–550 (2022). https://doi.org/10.1007/s11060-022-03997-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-022-03997-7